Amarin Corp. (AMRN)Website link: www.amarincorp.com
Archive of AMRN related updates and articles: Blue font denotes premium content for Chimera Research subscribers only Blog post: Will AMR101 receive NCE status from the FDA? (Addendum) Blog post: 11/15/2011 Lazard webcast notes. Blog posts re Amarin' intellectual property protecting AMR101: Part 1 - currently issued patents to AMRN. 2/2012 Leerink Swann conference (click for notes) 3/2012 Citi Healthcare conference (click for notes) 5/2012 Recent updates from 1q-2012 earnings call - click for blog. 6/2012 Jefferies Healthcare conference (click for notes) Click here for archive of all blog posts related to AMRN. ValuationShares Outstanding: 135.78 million (as of 9/30/2011)
Fully Diluted Share Count: 158.5m (1.6m options and 21.1m warrants at average $1.48 as of 9/30/11) See current Valuation on Stocks page. See embedded selections from 11/2011 webcast slide deck |
|
Links
- Audio interview - discussion of Vascepa REDUCE IT Cardiovascular Outcomes trial (Consultant Live, 8/2012)
- "Amarin says its key drug is exceeding expectations" (The Day, New London, CT)
- Competing patent for EPA plus statin for triglyceride lowering (USPTO)
Company Introduction
from 3q-2011 10q:
"We are a clinical-stage biopharmaceutical company focused on developing improved treatments for cardiovascular disease. We are currently focusing our efforts on AMR101, a semi-synthetic omega-3 fatty acid, comprising not less than 96% ultra pure icosapent ethyl (ethyl-EPA). On October 16, 2009, we completed a private placement resulting in gross proceeds of $70.0 million. These proceeds were used primarily to fund
the MARINE and ANCHOR studies for AMR101. In connection with this private placement, a significant portion of our board of directors and executive management were changed, and our research and development activities, as well as certain executive functions, were consolidated in the United States. In connection with these changes, we re-focused our efforts on developing improved treatments for cardiovascular disease and ceased development of all product candidates outside of our cardiovascular disease focus.
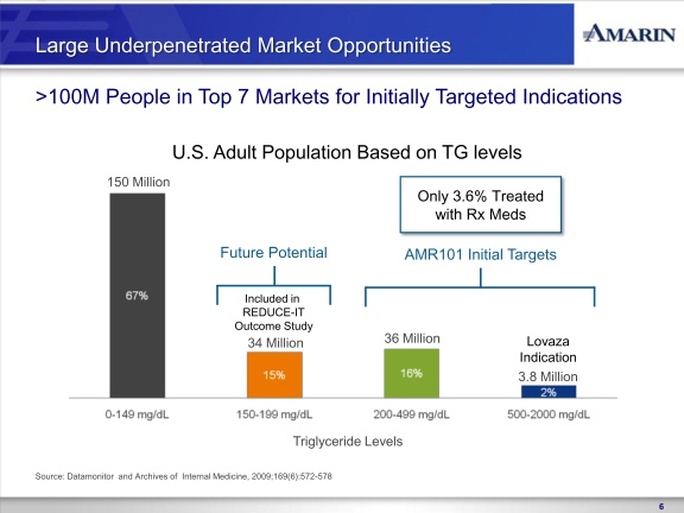
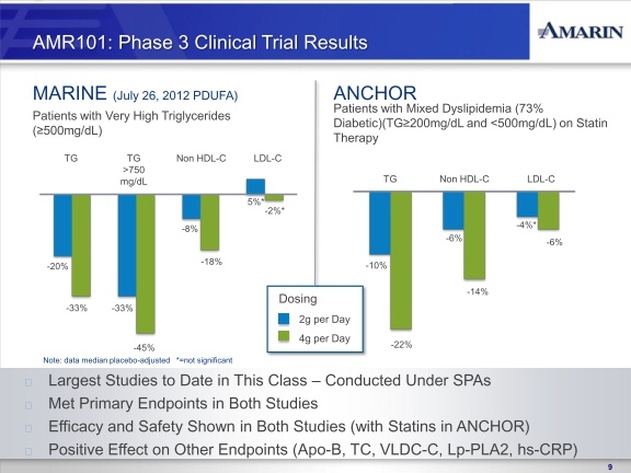
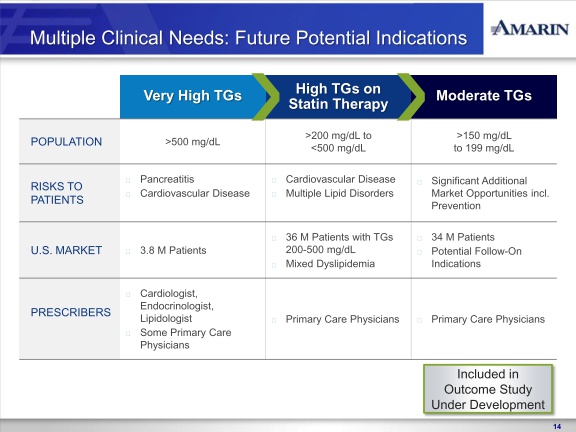
In November 2010, we reported positive top-line results from the MARINE trial, the first to complete of our two concurrently run Phase 3 clinical trials of AMR101. In the MARINE trial, AMR101 was investigated as a treatment for patients with very high triglycerides ( >500 mg/dL). The MARINE trial was a multi-center, placebo-controlled, randomized, double-blind, 12-week pivotal study to evaluate the efficacy and safety of 2 grams and 4 grams of AMR101 in 229 patients with fasting triglyceride levels ³ 500 mg/dL. Patients with this level of triglycerides are characterized as having very high triglyceride levels, as outlined in the National Cholesterol Education Program (NCEP) Expert Panel (Adult Treatment Panel III, 2002), or the NCEP Guidelines. The primary endpoint in the trial was the percentage change in triglyceride level from baseline compared to placebo after 12 weeks of treatment. Reported top-line results of this study included announcement that AMR101 met the primary endpoint at both the 4 gram and 2 gram doses. In addition to achieving the primary endpoint of the trial, no statistically significant increase in low-density lipoprotein cholesterol, or LDL-C, was observed in this trial at either dose. Additionally, we
observed in the trial a safety profile for AMR101 similar to placebo.
Patients enrolled in the MARINE trial were given the option to be treated with AMR101 for a period of up to 40-weeks after their last dose in the pivotal trial. The results from this 40-week open label non-placebo controlled extension period are not part of the MARINE trial primary endpoints.
In April 2011, we reported positive top-line results from the ANCHOR trial, the second of our two Phase 3 clinical trials of AMR101. In the ANCHOR trial, AMR101 was investigated as a treatment for patients with high triglycerides ( >200 and <500 mg/dL) who are also receiving statin therapy. The ANCHOR trial was a multi-center, placebo-controlled, randomized, double-blind, 12-week pivotal study to evaluate the efficacy and safety of 2 grams and 4 grams of AMR101 in 702 patients with high triglycerides who were on optimized statin therapy. Patients in this trial are characterized as having high triglyceride levels, as outlined in the NCEP Guidelines, with mixed dyslipidemia (two or more lipid disorders). The primary endpoint in the trial was the percentage change in triglyceride level from baseline compared to placebo after 12 weeks of treatment. No prescription omega-3 based drug is currently approved in the U.S. for treating high triglyceride levels in statin-treated patients who
have mixed dyslipidemia. Reported top-line results of this study included an announcement that AMR101 met the study’s primary endpoint at both the 4 gram and 2 gram doses. In addition, AMR101 met each of the secondary endpoints in the trial, including at both doses the key secondary endpoint of LDL-C non-inferiority to statin therapy alone. Additionally, we observed in the trial a safety profile for AMR101 similar to placebo.
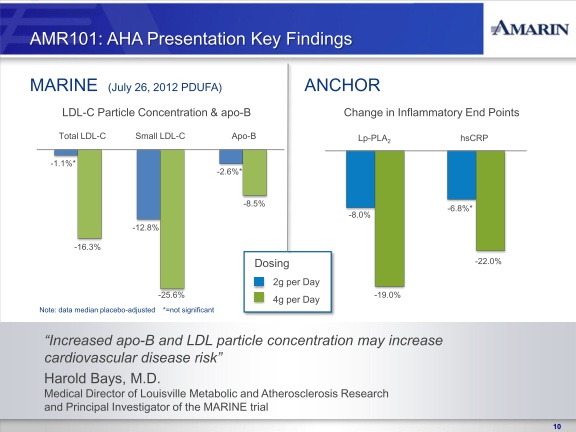
In addition to achieving the primary endpoints of the MARINE and ANCHOR trials, AMR101, particularly at the 4 gram dose, demonstrated significant reductions in various secondary and exploratory efficacy endpoints for other lipid and inflammate biomarkers which we believe are important as they potentially represent additional predictors of cardiovascular risk. These biomarkers include total cholesterol; non-HDLcholesterol; VLDL-cholesterol; Apo B (Apolipoprotein B), a sensitive index of residual cardiovascular risk which is generally considered to be a better predictor than LDL-C; Lp-PLA2 (Lipoprotein-phospholipase A2), an enzyme found in blood and atherosclerotic plaque and high levels of which have been implicated in the development and progression of atherosclerosis; and high sensitivity C-reactive protein (hsCRP), an important marker of vascular inflammation.
The MARINE and ANCHOR trials were conducted under separate Special Protocol Assessment, or SPA, with the FDA. An SPA is an evaluation by the FDA of a protocol with the goal of reaching an agreement that the Phase III trial protocol design, clinical endpoints, and statistical analyses are acceptable to support regulatory approval. The FDA agreed that, based on the information we submitted to the agency, the design and planned analysis of the MARINE and ANCHOR trials adequately address the objectives necessary to support a regulatory submission. An SPA is generally binding upon the FDA unless a substantial scientific issue essential to determining safety or efficacy is identified after the testing begins. Although we are not aware of any such issue, there is no assurance that the FDA will ultimately consider either of our SPAs to be binding. Moreover, any change to a study protocol can invalidate an SPA. If the FDA does not consider either of the SPAs to be binding, the agency could assert that additional studies or data are required to support a regulatory submission.
On September 26, 2011 we submitted a New Drug Application (NDA) to the FDA seeking approval to market and sell AMR101 in the United States for the indication studied in the MARINE trial (for the treatment of patients with very high triglycerides). Also included in our NDA submission were the safety and efficacy results of the ANCHOR trial. The FDA has 60 days after receipt of the NDA to preliminarily review the NDA and determine if the application is sufficiently complete to permit a substantive review and if it meets the threshold for filing.
In order to obtain a separate indication for AMR101 based on the ANCHOR trial results, our SPA with the FDA for the ANCHOR trial requires that we have a cardiovascular outcomes study substantially underway at the time of the NDA submission. In accordance with this SPA, the final results of an outcomes study are not required for FDA approval of indication studied in the ANCHOR trial. In accordance with our SPA for the MARINE trial an outcomes study is not required for the indication studied in such trial.
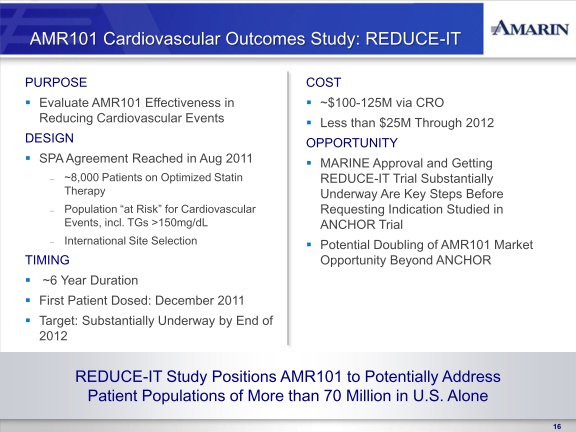
In August 2011, we reached agreement with the FDA on a SPA for the design of the cardiovascular outcomes study of AMR101, titled REDUCE-IT (Reduction of Cardiovascular Events with EPA – Intervention Trial). REDUCE-IT, is a multi-center, prospective, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the effectiveness of AMR101 in reducing first major cardiovascular events in an at-risk patient population. The control arm of the study will be patients on optimized statin therapy. The active arm of the study will be patients on optimized statin therapy plus AMR101. All subjects enrolled in the study will have elevated triglyceride levels (fasting TG levels of >135 mg/dL and < 500 mg/dL) and either coronary heart disease or risk factors for coronary heart disease. This study will be conducted internationally. Based on the results of REDUCE-IT, we may seek additional indications for AMR101 beyond the indication studied in the ANCHOR trial such as prevention of cardiac events, although there can be no assurance as to whether the results of the study will support to any such indication. In September 2011 we engaged a clinical research organization to begin initial trial and clinical site preparation. We will seek to have REDUCE-IT at least 50% enrolled by the end of 2012, and project the study to cost between $100 and $125 million over the anticipated six year study period, of which we anticipate not more than $25 million being paid before the end of 2012. We will require additional funds to complete REDUCE-IT.
In accordance with our SPA for the ANCHOR trial, we currently intend to file a supplemental NDA (sNDA) seeking approval of the indicationstudied in the ANCHOR trial once we have a cardiovascular outcomes study substantially underway. The sNDA cannot be filed until after boththe initially submitted NDA for the indication studied in the MARINE trial is approved and the cardiovascular outcomes study is substantiallyunderway. However, as part of our interaction with the FDA regarding our NDA for the indication studied in the MARINE trial, we may explorewhether there is an opportunity for the indication studied in the ANCHOR trial to be considered in conjunction with the FDA’s review of theindication studied in the MARINE trial. However, there can be no assurance that the FDA will approve such an approach.
We have filed and are prosecuting numerous patent applications in the U.S. and internationally. In the U.S. we have filed patent applicationswhich seek to expand to eleven the number of patent families protecting the proprietary position of AMR101. For certain of these patentfamilies, we have filed multiple patent applications. Collectively the patent applications include numerous independent claims and dependentclaims. Several of our patent applications contain claims based upon unexpected and positive findings from the MARINE and ANCHOR trials.If granted, we believe that many of these resulting patents would expire in 2030. However, no assurance can be given that any of our patentapplications will be granted or, if they grant, that they will prevent competitors from competing with AMR101. Securing additional patentprotection for a product is a complex process involving many legal and factual questions. The patent applications we have filed in the U.S. andinternationally are at varying stages of examination, the timing of which is outside our control. Many of our patent applications are unlikely to beexamined by the U.S. Patent Office or other international patent offices during 2011. In addition, we may elect to submit, or patent office’s mayrequire, additional evidence to support certain of the claims we are pursuing. Providing such additional evidence could result in us incurringadditional costs. We cannot be certain what commercial value any granted patent in our patent portfolio will provide to us.
In order to commercialize AMR101, we must either develop a sales, marketing and distribution infrastructure or collaborate with third partiesthat have such experience. With the assistance of financial advisors, we have held discussions about collaboration and other strategicopportunities with larger pharmaceutical companies in the past, and may continue to hold such discussions in the future. These strategicopportunities may include licensing or similar transactions, joint ventures, partnerships, strategic alliances, business associations, or a sale of thecompany. However, no assurance can be given that we will enter into any such strategic transaction.
Until such time as we complete any such strategic transaction, if ever, we are continuing to develop plans to launch, market and sell AMR101 onour own. This includes making preparations for securing a sufficient commercial supply of AMR101 and expanding sales and marketingcapabilities. In order to secure a sufficient commercial supply of AMR101, we have completed agreements for the supply of materials forAMR101 to provide for raw materials that are incremental to our existing Japan-based API supplier. Each agreement contemplates a phasedcapacity expansion plan aimed at creating sufficient capacity to meet anticipated demand for API material for AMR101 following FDAapproval. These API suppliers are self-funding these expansion plans with contributions from Amarin. We are also considering adding a fourthAPI supplier. These agreements include requirements for the qualification of the suppliers’ material and facilities with the FDA prior to thecommercial sale of any such material. We will make no purchase commitments until such time as these qualification events have been completed.
Opportunities to market and sell AMR101 outside of the United States are also under evaluation.
Unless we enter into a strategic collaboration in connection with the launch, marketing and sale of AMR101 to provide us with additionalcapital, we will need to raise additional capital on our own to support these efforts. We will also need additional capital to complete ourREDUCE-IT cardiovascular outcomes trial. Additional financing may not be available when we need it or may not be available on terms that areacceptable to us. If adequate funds are not available to us on a timely basis, or at all, we may be required to delay the establishment of sales andmarketing capabilities or terminate or delay our planned cardiovascular outcomes study. If we seek to raise additional funds, we may do sothrough the sale of additional equity, debt or convertible securities. Debt financing, if available, would result in increased fixed paymentobligations and may involve agreements that include covenants limiting or restricting our ability to take specific actions such as incurring debt,making capital expenditures or declaring dividends. The terms of any financings may be dilutive to, or otherwise adversely affect, holders of ouroutstanding securities. If we raise additional funds through collaboration, strategic alliance and licensing arrangements with third parties, we mayhave to relinquish valuable rights to our technologies, future revenue streams or product candidates, or grant licenses on terms that are notfavorable to us."
"We are a clinical-stage biopharmaceutical company focused on developing improved treatments for cardiovascular disease. We are currently focusing our efforts on AMR101, a semi-synthetic omega-3 fatty acid, comprising not less than 96% ultra pure icosapent ethyl (ethyl-EPA). On October 16, 2009, we completed a private placement resulting in gross proceeds of $70.0 million. These proceeds were used primarily to fund
the MARINE and ANCHOR studies for AMR101. In connection with this private placement, a significant portion of our board of directors and executive management were changed, and our research and development activities, as well as certain executive functions, were consolidated in the United States. In connection with these changes, we re-focused our efforts on developing improved treatments for cardiovascular disease and ceased development of all product candidates outside of our cardiovascular disease focus.
In November 2010, we reported positive top-line results from the MARINE trial, the first to complete of our two concurrently run Phase 3 clinical trials of AMR101. In the MARINE trial, AMR101 was investigated as a treatment for patients with very high triglycerides ( >500 mg/dL). The MARINE trial was a multi-center, placebo-controlled, randomized, double-blind, 12-week pivotal study to evaluate the efficacy and safety of 2 grams and 4 grams of AMR101 in 229 patients with fasting triglyceride levels ³ 500 mg/dL. Patients with this level of triglycerides are characterized as having very high triglyceride levels, as outlined in the National Cholesterol Education Program (NCEP) Expert Panel (Adult Treatment Panel III, 2002), or the NCEP Guidelines. The primary endpoint in the trial was the percentage change in triglyceride level from baseline compared to placebo after 12 weeks of treatment. Reported top-line results of this study included announcement that AMR101 met the primary endpoint at both the 4 gram and 2 gram doses. In addition to achieving the primary endpoint of the trial, no statistically significant increase in low-density lipoprotein cholesterol, or LDL-C, was observed in this trial at either dose. Additionally, we
observed in the trial a safety profile for AMR101 similar to placebo.
Patients enrolled in the MARINE trial were given the option to be treated with AMR101 for a period of up to 40-weeks after their last dose in the pivotal trial. The results from this 40-week open label non-placebo controlled extension period are not part of the MARINE trial primary endpoints.
In April 2011, we reported positive top-line results from the ANCHOR trial, the second of our two Phase 3 clinical trials of AMR101. In the ANCHOR trial, AMR101 was investigated as a treatment for patients with high triglycerides ( >200 and <500 mg/dL) who are also receiving statin therapy. The ANCHOR trial was a multi-center, placebo-controlled, randomized, double-blind, 12-week pivotal study to evaluate the efficacy and safety of 2 grams and 4 grams of AMR101 in 702 patients with high triglycerides who were on optimized statin therapy. Patients in this trial are characterized as having high triglyceride levels, as outlined in the NCEP Guidelines, with mixed dyslipidemia (two or more lipid disorders). The primary endpoint in the trial was the percentage change in triglyceride level from baseline compared to placebo after 12 weeks of treatment. No prescription omega-3 based drug is currently approved in the U.S. for treating high triglyceride levels in statin-treated patients who
have mixed dyslipidemia. Reported top-line results of this study included an announcement that AMR101 met the study’s primary endpoint at both the 4 gram and 2 gram doses. In addition, AMR101 met each of the secondary endpoints in the trial, including at both doses the key secondary endpoint of LDL-C non-inferiority to statin therapy alone. Additionally, we observed in the trial a safety profile for AMR101 similar to placebo.
In addition to achieving the primary endpoints of the MARINE and ANCHOR trials, AMR101, particularly at the 4 gram dose, demonstrated significant reductions in various secondary and exploratory efficacy endpoints for other lipid and inflammate biomarkers which we believe are important as they potentially represent additional predictors of cardiovascular risk. These biomarkers include total cholesterol; non-HDLcholesterol; VLDL-cholesterol; Apo B (Apolipoprotein B), a sensitive index of residual cardiovascular risk which is generally considered to be a better predictor than LDL-C; Lp-PLA2 (Lipoprotein-phospholipase A2), an enzyme found in blood and atherosclerotic plaque and high levels of which have been implicated in the development and progression of atherosclerosis; and high sensitivity C-reactive protein (hsCRP), an important marker of vascular inflammation.
The MARINE and ANCHOR trials were conducted under separate Special Protocol Assessment, or SPA, with the FDA. An SPA is an evaluation by the FDA of a protocol with the goal of reaching an agreement that the Phase III trial protocol design, clinical endpoints, and statistical analyses are acceptable to support regulatory approval. The FDA agreed that, based on the information we submitted to the agency, the design and planned analysis of the MARINE and ANCHOR trials adequately address the objectives necessary to support a regulatory submission. An SPA is generally binding upon the FDA unless a substantial scientific issue essential to determining safety or efficacy is identified after the testing begins. Although we are not aware of any such issue, there is no assurance that the FDA will ultimately consider either of our SPAs to be binding. Moreover, any change to a study protocol can invalidate an SPA. If the FDA does not consider either of the SPAs to be binding, the agency could assert that additional studies or data are required to support a regulatory submission.
On September 26, 2011 we submitted a New Drug Application (NDA) to the FDA seeking approval to market and sell AMR101 in the United States for the indication studied in the MARINE trial (for the treatment of patients with very high triglycerides). Also included in our NDA submission were the safety and efficacy results of the ANCHOR trial. The FDA has 60 days after receipt of the NDA to preliminarily review the NDA and determine if the application is sufficiently complete to permit a substantive review and if it meets the threshold for filing.
In order to obtain a separate indication for AMR101 based on the ANCHOR trial results, our SPA with the FDA for the ANCHOR trial requires that we have a cardiovascular outcomes study substantially underway at the time of the NDA submission. In accordance with this SPA, the final results of an outcomes study are not required for FDA approval of indication studied in the ANCHOR trial. In accordance with our SPA for the MARINE trial an outcomes study is not required for the indication studied in such trial.
In August 2011, we reached agreement with the FDA on a SPA for the design of the cardiovascular outcomes study of AMR101, titled REDUCE-IT (Reduction of Cardiovascular Events with EPA – Intervention Trial). REDUCE-IT, is a multi-center, prospective, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the effectiveness of AMR101 in reducing first major cardiovascular events in an at-risk patient population. The control arm of the study will be patients on optimized statin therapy. The active arm of the study will be patients on optimized statin therapy plus AMR101. All subjects enrolled in the study will have elevated triglyceride levels (fasting TG levels of >135 mg/dL and < 500 mg/dL) and either coronary heart disease or risk factors for coronary heart disease. This study will be conducted internationally. Based on the results of REDUCE-IT, we may seek additional indications for AMR101 beyond the indication studied in the ANCHOR trial such as prevention of cardiac events, although there can be no assurance as to whether the results of the study will support to any such indication. In September 2011 we engaged a clinical research organization to begin initial trial and clinical site preparation. We will seek to have REDUCE-IT at least 50% enrolled by the end of 2012, and project the study to cost between $100 and $125 million over the anticipated six year study period, of which we anticipate not more than $25 million being paid before the end of 2012. We will require additional funds to complete REDUCE-IT.
In accordance with our SPA for the ANCHOR trial, we currently intend to file a supplemental NDA (sNDA) seeking approval of the indicationstudied in the ANCHOR trial once we have a cardiovascular outcomes study substantially underway. The sNDA cannot be filed until after boththe initially submitted NDA for the indication studied in the MARINE trial is approved and the cardiovascular outcomes study is substantiallyunderway. However, as part of our interaction with the FDA regarding our NDA for the indication studied in the MARINE trial, we may explorewhether there is an opportunity for the indication studied in the ANCHOR trial to be considered in conjunction with the FDA’s review of theindication studied in the MARINE trial. However, there can be no assurance that the FDA will approve such an approach.
We have filed and are prosecuting numerous patent applications in the U.S. and internationally. In the U.S. we have filed patent applicationswhich seek to expand to eleven the number of patent families protecting the proprietary position of AMR101. For certain of these patentfamilies, we have filed multiple patent applications. Collectively the patent applications include numerous independent claims and dependentclaims. Several of our patent applications contain claims based upon unexpected and positive findings from the MARINE and ANCHOR trials.If granted, we believe that many of these resulting patents would expire in 2030. However, no assurance can be given that any of our patentapplications will be granted or, if they grant, that they will prevent competitors from competing with AMR101. Securing additional patentprotection for a product is a complex process involving many legal and factual questions. The patent applications we have filed in the U.S. andinternationally are at varying stages of examination, the timing of which is outside our control. Many of our patent applications are unlikely to beexamined by the U.S. Patent Office or other international patent offices during 2011. In addition, we may elect to submit, or patent office’s mayrequire, additional evidence to support certain of the claims we are pursuing. Providing such additional evidence could result in us incurringadditional costs. We cannot be certain what commercial value any granted patent in our patent portfolio will provide to us.
In order to commercialize AMR101, we must either develop a sales, marketing and distribution infrastructure or collaborate with third partiesthat have such experience. With the assistance of financial advisors, we have held discussions about collaboration and other strategicopportunities with larger pharmaceutical companies in the past, and may continue to hold such discussions in the future. These strategicopportunities may include licensing or similar transactions, joint ventures, partnerships, strategic alliances, business associations, or a sale of thecompany. However, no assurance can be given that we will enter into any such strategic transaction.
Until such time as we complete any such strategic transaction, if ever, we are continuing to develop plans to launch, market and sell AMR101 onour own. This includes making preparations for securing a sufficient commercial supply of AMR101 and expanding sales and marketingcapabilities. In order to secure a sufficient commercial supply of AMR101, we have completed agreements for the supply of materials forAMR101 to provide for raw materials that are incremental to our existing Japan-based API supplier. Each agreement contemplates a phasedcapacity expansion plan aimed at creating sufficient capacity to meet anticipated demand for API material for AMR101 following FDAapproval. These API suppliers are self-funding these expansion plans with contributions from Amarin. We are also considering adding a fourthAPI supplier. These agreements include requirements for the qualification of the suppliers’ material and facilities with the FDA prior to thecommercial sale of any such material. We will make no purchase commitments until such time as these qualification events have been completed.
Opportunities to market and sell AMR101 outside of the United States are also under evaluation.
Unless we enter into a strategic collaboration in connection with the launch, marketing and sale of AMR101 to provide us with additionalcapital, we will need to raise additional capital on our own to support these efforts. We will also need additional capital to complete ourREDUCE-IT cardiovascular outcomes trial. Additional financing may not be available when we need it or may not be available on terms that areacceptable to us. If adequate funds are not available to us on a timely basis, or at all, we may be required to delay the establishment of sales andmarketing capabilities or terminate or delay our planned cardiovascular outcomes study. If we seek to raise additional funds, we may do sothrough the sale of additional equity, debt or convertible securities. Debt financing, if available, would result in increased fixed paymentobligations and may involve agreements that include covenants limiting or restricting our ability to take specific actions such as incurring debt,making capital expenditures or declaring dividends. The terms of any financings may be dilutive to, or otherwise adversely affect, holders of ouroutstanding securities. If we raise additional funds through collaboration, strategic alliance and licensing arrangements with third parties, we mayhave to relinquish valuable rights to our technologies, future revenue streams or product candidates, or grant licenses on terms that are notfavorable to us."
Patent Notes
- Issued US patent #6,479,544: "Therapeutic combinations of fatty acids" - Link.
- US Patent Application #12/702,889 - Document link from ChasingtheAlpha
- US Patent Application #13/040,977 - Prevention of Cardiovascular disease
- US Patent Application #12/888,994 - Omega-3 Combination with statins
- US Patent Application #12/769,885 - "bioavailability long shelf life"
- US Patent Application #13/061,865 - COMPOSITION COMPRISING EICOSAPENTAENOIC ACID AND NICOTINIC ACID