Spectrum Pharma SPPI Stock Research: Belinostat - TopoTarget page
Belinostat
- HDAC inhibitor licensed 1/2010 from TopoTarget ($30m upfront, $320m plus 1m shares as milestones plus double-digit royalties).
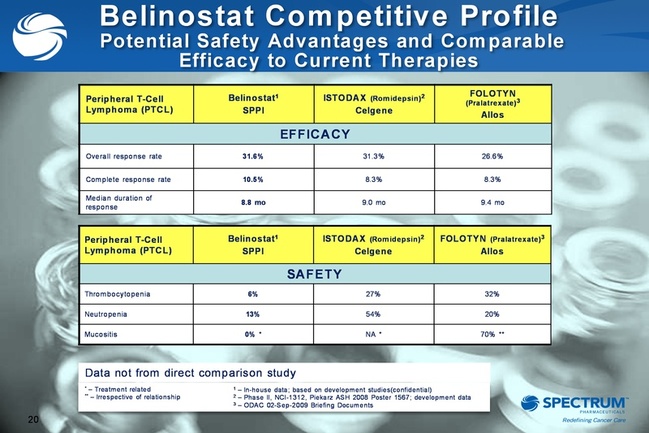
- competing drug for relapsed/refractory PTCL has 30% leukocytis vs none for belinostat (and no bone marrow toxicity. Can be administered as continuous IV infusion, single IV dose, or oral tablet).
- Has received FDA Fast Track status for peripheral T cell lymphoma (PTCL): 7500 pts/yr w/ poor prognosis.
- Pivotal PTCL monotherapy trial (SPA, requires 100 evaluable pts)
- 8/2010: no enrollment #s, topo won't discuss, 1q2011 10q says over 100 sites opened and plan to complete enrollment in mid to late 2h2011 [originally said would be enrolled by ye2009!].
- 7/2011 corporate slide deck now says "complete pt enrollment and file NDA in 2012"...this is a significant slip in timeline from when deal was announced and NDA was to be filed in 2011
- 1q2011 10q: will begin rolling NDA filing with preclinical and CMC sections by ye-2011
- Cancer of unknown promary (CUP): p2 randomized w/ carboplatin and paclitaxel, (run by TT, 12/2010: enrollment complete at 88 pts, release PFS data 3q11 and OS data 1q12, seek fda/emea advice for p3, est 0.8-1b mkt) this trial paid by topotarget, primary endpt PFS).
- NSCLC: 12/2010 started p1/2 trial in combo w/ Tarceva (35 pts) this and future trials cost split 70/30.
- Several other mono and combo trials underway by NCI (3/2010 p1/2 in thymic started, 30-48 pts expected; 1/2010 ovarian combo initiated; 1/2009 p1 liver started, now in p2 section; MDS, MM).
- Click here for list of all Belinostat trials listed in the ClinicalTrials.gov database.
- COM patents expire 2021 or later.
- 9/2010: 700+ pts, much unpublished data, planning to present some