Spectrum Pharma SPPI Stock Research - Fusilev page
Fusilev
- Note that Zevalin always used to be the top of the pipeline chart featured program that received the bulk of the company's attention. However, anemic sales growth and the windfall sales of Fusilev thanks to the generic shortage has changed that, and now the company prominently features Fusilev. It remains to be seen if the sales will persist once generic competition is restored.
- Fusilev: injectable for rescue after high-dose methotrexate (MTX) in osteosarcoma patients (received FDA approval for this small indication in 3/2008). Ready-to-use (TRU) dosage form sNDA approved 4/2011.
- Formulation patent expires 2019
- 3/2006: acquired asset from Targent - will owe a $5m milestone in 2011 because net sales for the year will exceed $40m.
- Ex-US sales (completely independent of SPPI) are about $100m in Japan by Takeda and >$80m in Europe by Pfizer $PFE and Sanofi-Aventis $SNY.
- 11/2008 shortage of generic leuocovorin (used w/ 5FU in CRC), fda asked sppi to supply, sold 22M in 4 months.
- 10/2009 recd CR for mCRC- did not demo non-inferiority to leucovorin, met w/ FDA 1/10: addl data but no new studies reqd (preclinical and healthy volunteer safety data needed), met multiple times since, response filed 10/29/2010 and accepted w/ 4/29/2011 PDUFA.
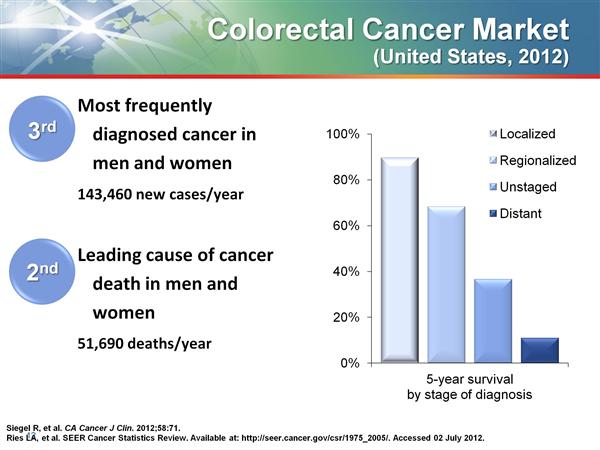
- 4/2011: Fusilev was approved for use in combo w/ 5-FU for palliative treatment in advanced metastatic colorectal cancer (a much larger market, which already made up the bulk of Fusilev sales on an off-label basis)
- As of April 2011, reports surfaced that Teva would recommence shipping generic leucovorin. This remains to be seen, and will greatly affect the level of sales of Fusilev (more so than receiving approval for CRC). SPPI released a PR in June 2011 trumpeting that they have plenty of supply to handle demand - time will tell us whether they needed that capacity or not.
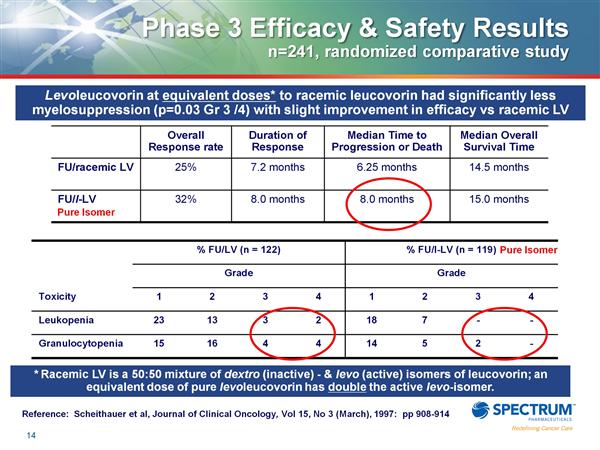
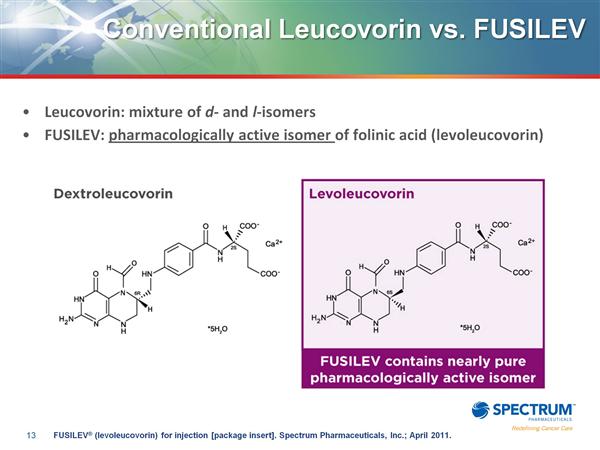
- Only data supplied originally was old paper showing pure isomer (Fusilev) is safer and as effective as mixture (generic).
- Company predicts would be 100M sales if approved for CRC (95% of sales).
- currently $180m ex-us sales (on market 10 yrs).
- 2nd shortage began 6/10, ongoing 11/10.
- Fusilev was manufactured by Cangene (formerly Chesapeake Biol Labs)-shutting down Dec-March and refused to delay this to produce SPPI's increased orders.