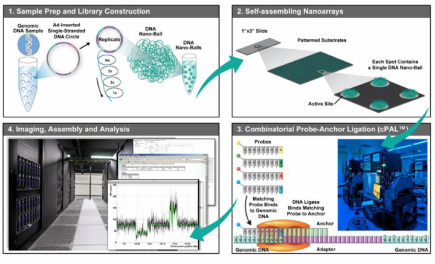
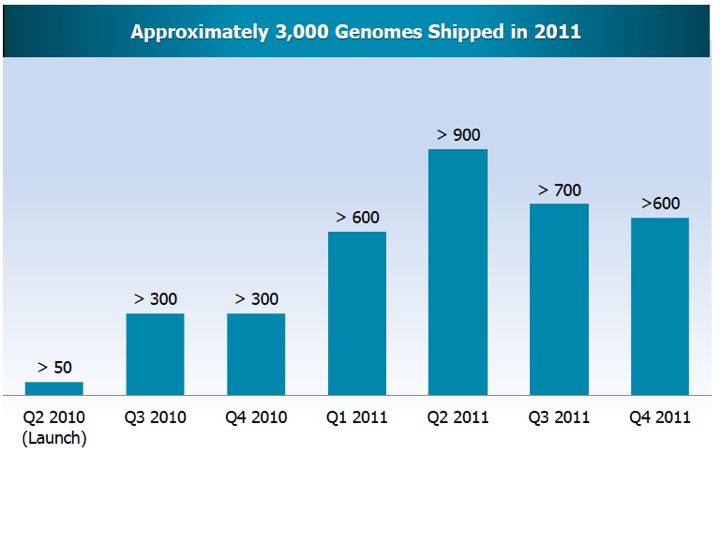
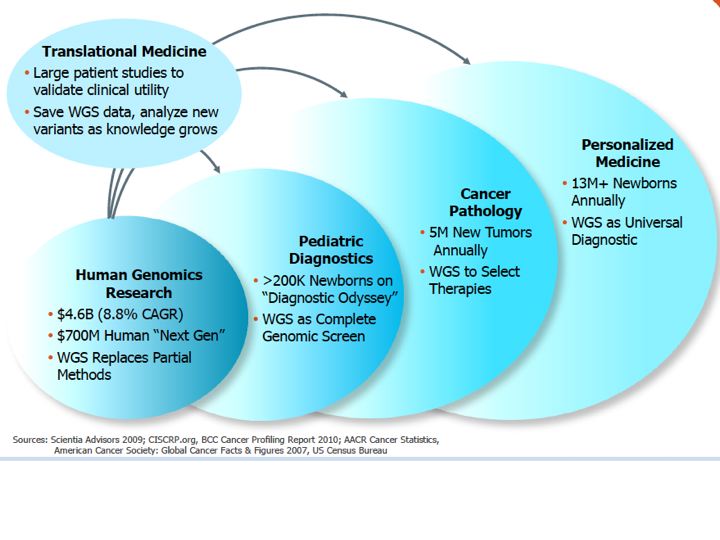
Complete Genomics (GNOM)Website link: www.completegenomics.com
December 2012 Updates: details on BGI-GNOM merger and attempts by ILMN to thwart the deal. Recent Updates 8/2012 Relevant notes from ILMN CC - blog. 11/2012 Notes from ILMN 3q-2012 conference call. |
|
|
Corporate Resources
|
Next Generation Sequencing (NGS) Resources
- Pacific Biosciences $PACB 2011 Annual Report form 10-K (click to download)
- PACB 1q-2012 Quarterly Report for 10-Q (click to download)
- PACB Slide Deck March 2012 (click to download via Google Docs)
- Illumina $ILMN 2011 Annual Report form 10-K (click to download)
- Illumina - offers $9500 price for CLIA-certified whole genome sequencing - direct competitor to GNOM
- Oxford Nanopore valuation doubles to about $450m following May 2012 surprise financing round initiated by existing investors (Link)
- April 2012 GenomeWeb article about a Nature Biotechnology paper comparing various competitors - "no single platform comes out on top" (Link)
- May 2012 GenomeWeb article on why new biotech Warp Drive Bio chose not to create their own sequencing facility and instead outsources for example to GNOM and Chinese competitor BGI (Link)
- December 2012 PartnersHealthCare launches $9000 personal whole genome sequencing service (ILMN technology).
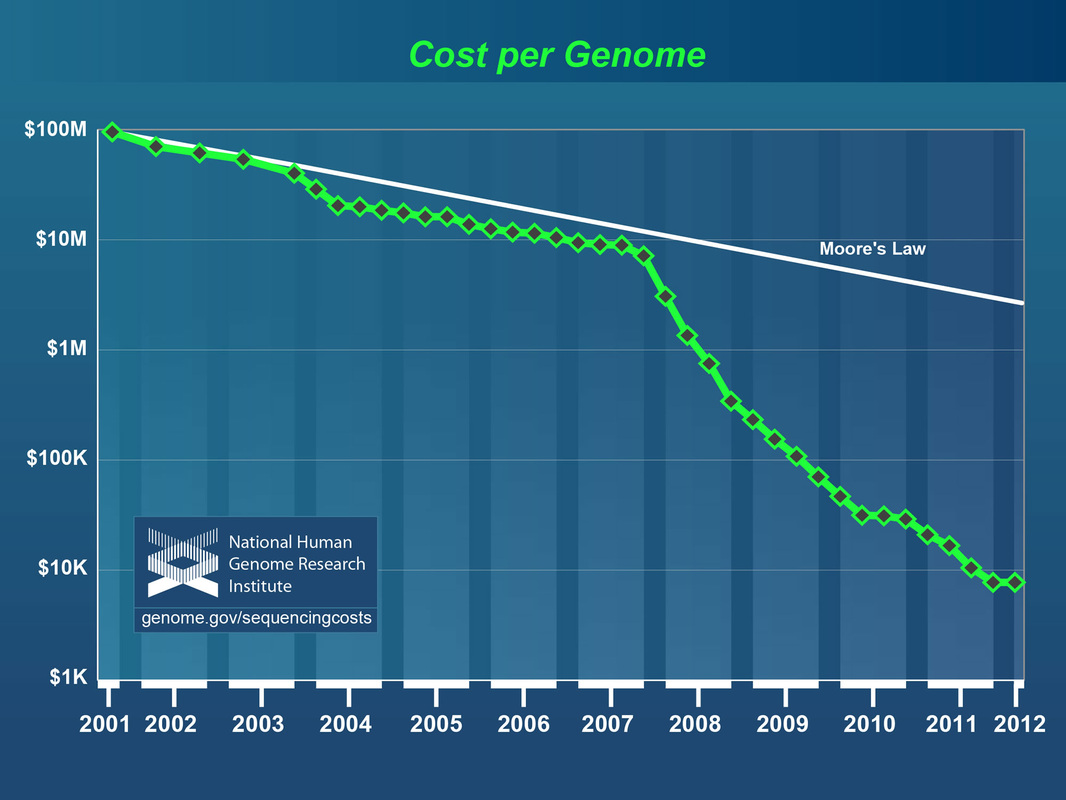
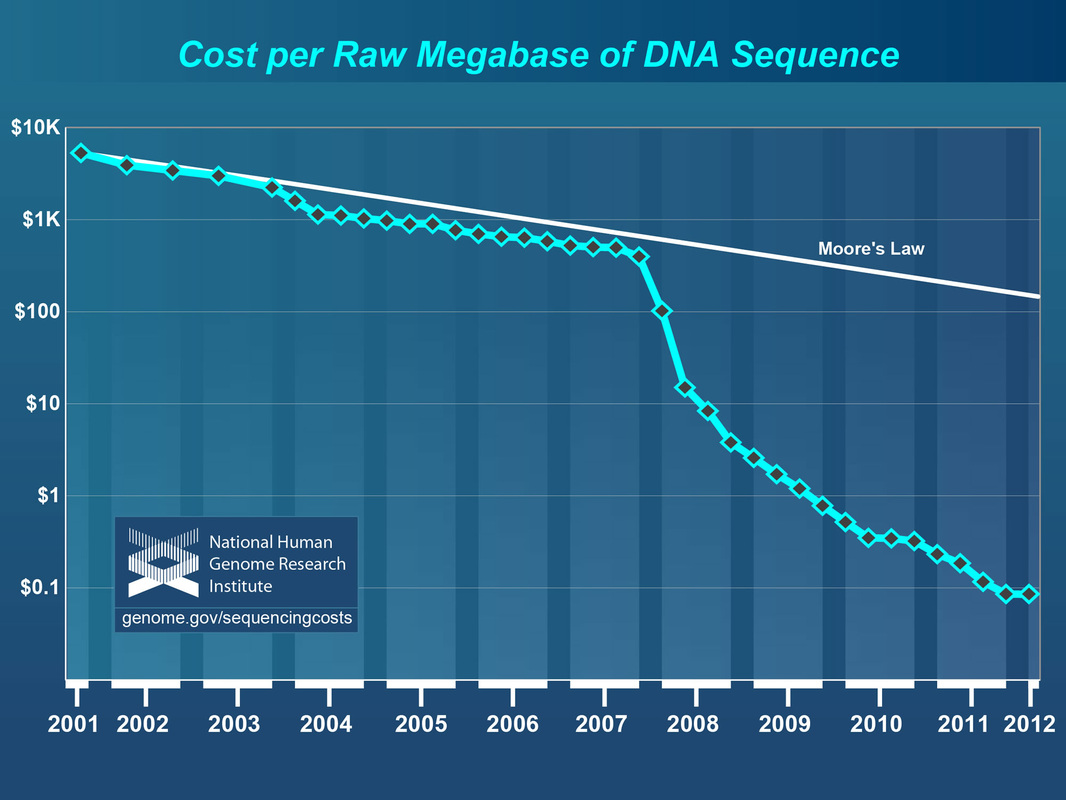
- Information on DNA sequencing costs - see figures below (Link)
First Quarter 2012 Earnings Call Notes - courtesy of Daniel Low (@ddlow - a must follow re GNOM)
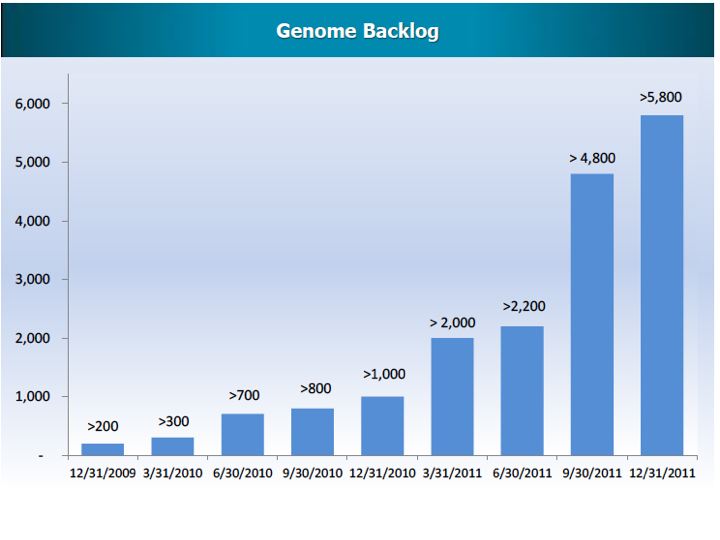
- Backlog of 5700 genomes with potential revenue of $27M (Currently have 2000 of the backlog with samples in hand, other samples will be shipped to Complete Genomics at customers discretion)
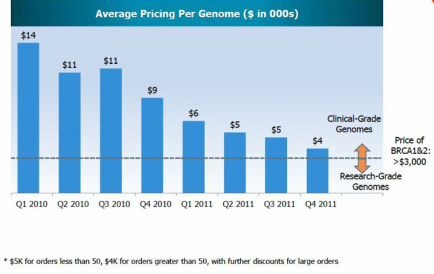
- Expect to ship 2000 sequenced genomes to customers for Q2 (assume $4000. per genome for revenue = $8M)
- Current Max capacity of 1000 genomes per month for Q1 and Q2.
- Currently reporting 150 customers (25 more as compared to last qtr.)
- Current turn-around time is at >80 days (Expect turn-around times to decrease 60 days for Q3 and 30-45 days for Q4)
- CLIA certification application expected to be filed in Q3 with expectation to be CLIA certified by end of Q3.
(expect a 2 week delay from filing date for State to receive, then 6-8 week for State turn around)
- LFR (Long Fragmented Read) technology expected to be commercially available Early 2013. (was announced at TCGC conference June 2012 that there would be a new paper involving the LFR technology coming out shortly in Nature)
Upcoming Milestones and Catalysts - courtesy of Daniel Low (he is long GNOM shares, do your own DD)
- End of May/June Expect delivery of 6 new machines (will be capable of 6 genomes per day per machine) [now on hold per restructuring announced 6/5/2012 via 8-k]
- End of Jun/July Expect CLIA application filing [Jill Hagenkord announced at TCGC that they will file in July. Up till now this was reported as a Q3 filing not more specific]
- End of Aug/Sep Expect to be CLIA certified (This could run into Oct)
- End of Aug/Sep Expect Mayo details and new contracts after CLIA certification approval
- End of Oct/Nov Expect current backlog to be worked out based on samples on hand
- End of Nov/Dec Expect upgrades to machines to process up to 10 genomes per day [This will demand based and with the new sequencers being delayed all upgrades now on hold until further notice.]