Momenta Pharmaceuticals (MNTA)Website link: www.momentapharma.com
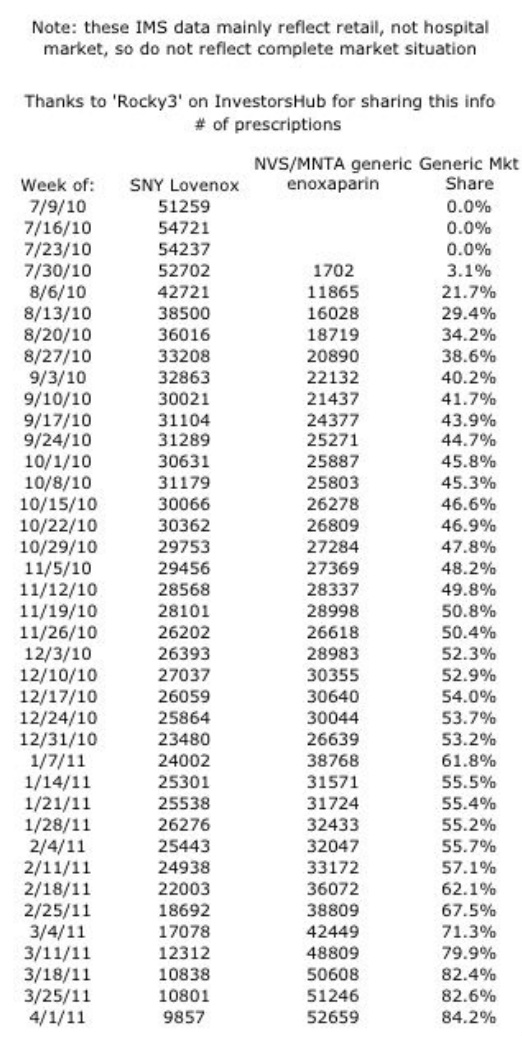
List of MNTA updates: Blue font denotes premium content for Chimera Research Group subscribers. Deutsche Bank webcast 5/2/11 - click here for my notes 1q2011 earnings 5/5/11: click here for PR --- click here for my notes BofA Merrill Lynch webcast 5/10/11: click here for my notes Jefferies webcast 6/6/11: click here for my notes Goldman Sachs webcast 6/8/11: click here for my notes TEVA GS webcast 6/7/11: click for my notes 2q2011 earnings 8/4/11: click here for my notes. Canaccord Genuity webcast 8/10/11: click here for my notes. Wedbush webcast 8/17/11: click here for my notes. Rodman and Renshaw webcast 9/13/11: click here for my notes. Morgan Stanley webcast 9/14/11: click here for my notes. UBS webcast 9/21/11: click here for my notes. BioCentury webcast 10/21/11: click here for my notes. NVS 3q2011 earnings webcast: click here for my notes. WPI 3q2011 earnings CC: click here for my notes. 5/2012 ASCO preview and partner, competitor updates - click for blog. 5/2012 ASCO abstract - click for blog. I will post notes on MNTA from time to time, but by far the most complete source for information is the Biotech Values and Momenta discussion boards at InvestorsHub, run by Dew Diligence and many other great posters (see link below) See table below for a key attribute of MNTA- they are raking in the cash from generic Lovenox sales with a 45% royalty that has steadily gained share in a multi-billion dollar market against SNY's product Upcoming Events
Links
|
|
Introduction
- Corporate Mission: "Momenta is a biotechnology company specializing in the characterization and process engineering of complex molecules. These complex molecules include proteins, polypeptides, and cell surface polysaccharides, like heparan-sulfate proteoglycans, or HSPGs. This results in a diversified product pipeline of complex generic, follow-on biologic, and novel drugs. These product opportunities are derived from our proprietary, innovative technology platform which we leverage to study the structure (thorough characterization of chemical components), structure-process (understand, design and control of manufacturing process), and structure-activity (understand and relate structure to biological and clinical activity) of complex molecule drugs." (2/2011 BIO CEO snapshot)
- Prorietary Technology: "Our integrated technology platform utilizes three different types of analytical tools to study the structure of complex molecules. First, we have accumulated a comprehensive library of enzymes that we use to break down the components of a complex molecule into smaller, more measurable units. Second, we apply proprietary improvements to established analytical techniques to gather and analyze information regarding the molecular components, structure and arrangement of the chemical building blocks that comprise these complex molecules. Third, we apply proprietary mathematical modeling to describe the composition of each complex molecule product candidate. It is the combination of these tools that enables us to thoroughly characterize complex HSPG, polypeptide and protein products." (2/2011 BIO CEO snapshot)
Management
- Craig Wheeler - CHief Executive Officer (CEO)
- Rick Shea - Chief Financial Officer (CFO)
- Ganesh Venkataraman - Chief Scientific Officer (CSO)
- Jim Roach - Chief Medical Officer (CMO)
- Bruce Leicher - General Counsel
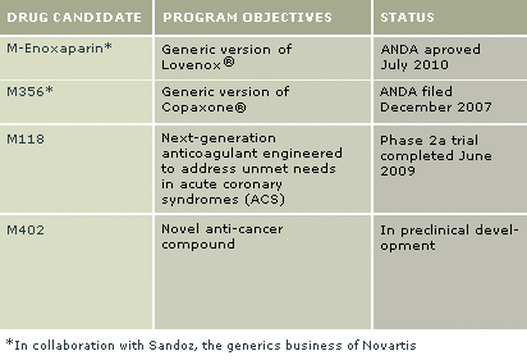
m-Enoxaparin: FDA approved and marketed generic Lovenox
M356: generic Copaxone for Multiple Sclerosis
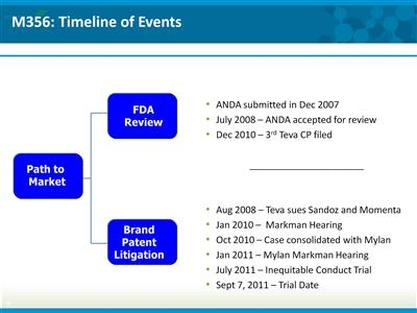
- ANDA is under review by FDA
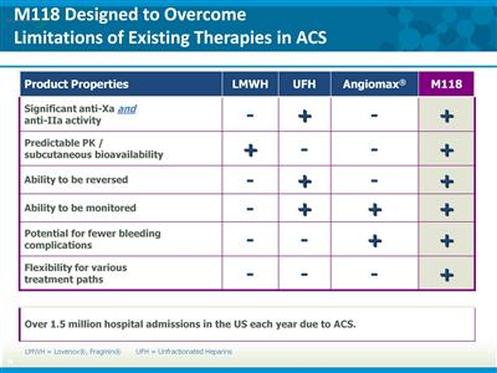
M118
- Phase 2 trial complete, awaiting partner to develop further (very expensive trials and competitive indication)
M402
- Preclinical oncology novel drug candidate
- IND and phase 1 trial have been guided to occur in 2011 (but has apparently slipped from prior mid-year IND guidance)
- 6/2011: PLOS One paper published demonstrating efficacy in mouse models of melanoma and breast cancer (Click here for article link)
M923
- Undisclosed product candidate that appeared on 2/2011 BIO CEO company summary as being in "Research" phase
- This could potentially be either a follow-on biologic (FOB) candidate or another novel product discovered using MNTA's technology
FDA pathways for submission and Product approval
505(j) aka Abbreviated New Drug Application (ANDA)
- Seeking generic approval of a product that is comparable to the branded, approval product that is already on the market (including formulation, dose, route of administration, and purity)
- Can be approved with designation to be automatically substitutable for the innovator product- ie, even though the doctor writes prescription for the innovator product, the pharmacy can give the generic drug instead
- Generally need to submit 3 month stability data, but no formal safety or efficacy data required
- "First to File" status can result in the generic company obtaining 6 months of exclusivity upon approval
- Changes to dose form, strength, route of administration, or formulation
- Generally need 12 month stability data for new formulation
- A bridging study is performed to compare the systemic levels of the new product compared to approved product
- Sponsor may conduct some sort of safety or efficacy trials, but can also sometimes access the safety and efficacy data of the reference product
- Unlike ANDA pathway, these approval are not usually deemed substitutable
- This pathway is faster and less expensive than NDA pathway
- Approved product can be granted 3 years of Hatch-Waxman exclusivity if the sponsor conducted some clinical trials
- Unlike NDAs, approval can be held up due to patent protection of the already approved product
- New molecular entity
- Requires full clinical safety and efficacy studies
Notes: TEVA 1/6/2011 Webcast
- (not all directly related to MNTA but included all of my notes anyway, as the status of Teva and MNTA Copaxone and Lovenox programs are tightly linked and have large implications from each company)
- Mylan claims construction hearing to be held 1/19/11, if decision handed down 1q2011, could file summary judgement 2q2011, judge's ruling by ye2011, then trial
- Re 0.5 mL formulation of Copaxone that received CRL- will request a meeting with FDA and then determine whether to go ahead with the required clinical trial that would take a couple of years
- Started to shift attention to GALA trial of 3x weekly copaxone...but said pursuing low volume and 3x/week dosing plans not mutually exclusive and could potentially be combined
- Re 2015 forecast of $9b in branded sales, Teva has Copaxone making up $2b of this, another $1.8-2b requires future acquisitions- want to keep current therapeutic focus areas: mentioned respiratory and women's health
- Mentioned another generic biologic to be filed in late 2011 or early 2012 (not referring to pending Lovenox ANDA)
- By 2015, Teva forecasts it will have $800m in generic biologic sales, up from <$100m right now (currently GCSF's, etc)- all via organic growth
- They talk about these as actively marketed/promoted products, rather than the fully substitutable generic biologics that MNTA strives to get approved
- Lovenox- mgmt has been told that they would get a response from FDA by the end of January 2011- could be approval, request for more info on immunogenicity, or something else
- Teva feels they can achieve better return on equity (ROE) thru acquisitions vs share buyback
- Seeking to expand in Brazil, Japan, Russia, Mexico; plus branded products or medium sized companies, don't think acquisitions in US are over
Notes: Teva 2/8/2011 Webcast
- Teva claims that "any generic Copaxone is, at the nearest, years away"
- Disappointed that their generic Lovenox was not approved in 2010. "We hope to launch enoxaparin in 2011, but we really are not going to talk about it much at this time"
- Sounds like exonaparin sales were included in the guidance issued with 4q10 results
- Would not answer question regarding the issues contained in the Minor Deficiency Letter received January 2011 or the timeline for next steps. Now saying they don't plan to comment further until product launch (!)
Notes: Teva 2/9/2011 Investor Day Webcast
- Claim CRL for 0.5 mL low volume Copaxone gives them more confidence that trials would be required for any generic (this is absurd of course, as the regulatory requirements for a generic copaxone ANDA filing have nothing to do with requirement for a sNDA for a changed formulation)
- Currently having "internal debate" about next steps for low-volume Copaxone program, likely will seek meeting with FDA
- Based on January appearance in court, Teva said that Sandoz attorneys admitted that they are not ready to launch generic copaxone, that their product infringes the Teva marker patents, and that they still need to file amendment - so obviously still time before approval (The nature of these comments were flatly deined by MNTA CEO at their 4q2010 conference call)
- Teva does not consider itself a discovery company- do not have the "critical mass"--instead they seek to license phase 1 and 2 products- phase 3 products are too expensive
- Therapeutic areas of interest mentioned include: women's health, neuroology, wound healing, injectable corticosteroids
Notes: 2/10/2011 MNTA 4q2010 results conference call webcast
- 100's of thousands of pts have already received NVS Sandoz/MNTA generic Lovenox (enoxaparin), savings $100's of millions in healthcare costs
- Operating profit for the product is 68.5%, MNTA receives 45% of profits
- CEO acknowledged that TEVA comments have moved MNTA stock price recently...reiterated that "minor deficiency letters" can be quite significant, such as the immunogenicity response that MNTA had to file...can take several months or long...ANDA review generally completely stops until response is received
- CEO noted that TEVA claimed their enox approval was going to be one month after MNTA (ie, August 2010), then by year end 2010, then maybe by end of January 2011, now they have not yet filed a response to FDA and won't comment anymore
- ANDA review backlog currently averages 26 months. Generic Copaxone ANDA has been at FDA for 2.5 years (accepted July 2008), so not too bad given complexity of the product. MNTA strongly believes this product is approvable via 505j pathway. Copaxone is a synthetic product, so no supply chain issues to resolve like for heparin based generic Lovenox
- Teva received a CRL for the sNDA application for low volume (0.5 mL) copaxone- said that an efficacy trial was required because the formulation change could alter the clinical profile
- M356 (MNTA generic copaxone) is, in contrast, the same product as approved Copaxone, and has been accepted as ANDA, so the sNDA CRL has no impact on this product. FDA has not asked for safety or efficacy trials
- 30 month period after filing expired June 2010, so would not get 6 month first filer exclusivity unless granted an exception
- 30 month legal stay of approval has expired, so NVS/MNTA have option to launch at risk upon approval
- Expect Teva to do everything they can to delay the Copaxone patent trial, but think it likely would start 2h2011
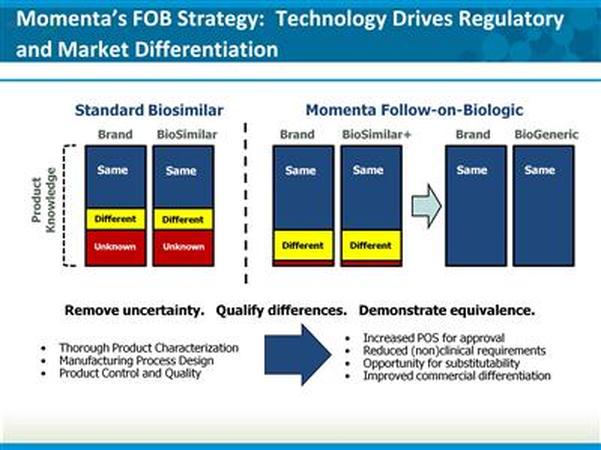
- regarding follow-on biologics (FOBs)- MNTA believes that the FDA process will allow them to differentiate their products, including by seeking interchangable designations
- MNTA is seeking a broad platform portfolio partnership for FOBs. Have had substantive discussions with many companies who have stated intentions to enter the FOB arena. Need alliance on strategy with the partner as well. Have the luxury to advance programs internally further due to Lovenox sales- but hope for deal in 2011
- M118 have yet to find a partner, but still committed to it. Perceived licensing arena for CV drugs is improved compared to when first tried to partner, but that doesn't mean they are predicting success. During first round of talks, companies were cutting their own portfolios, recent failures in the space have helped- along with the desire to have oral and injectable possibilities in the pipeline, and have initiated a second round of talks
- M402 cancer product, IND seen mid-year 2011, classic single and multi dose phase 1 in 2011
- $170m reported enox sales - subtract cost of goods sold (COGS) and contractually allowed SGA expense leaves $117m contractual profit- 68.5% operating margin in 4q, up from 60% 3q10. The current figure is about right going forward, will fluctuate somewhat
- Operating expenses net of collaboration revenue will be $15-18m quarterly.
- Have increased headcount from 170 to 195, mostly in FOB and novel programs- have a backlog of promising programs in each of these that hadn't been able to invest in previously
- Question and answer session:
- enox market share in range of 35-45%. Have not seen any radical pricing changes from SNY
- Would not answer whether MNTA got a minor deficiency letter from FDA after resubmitting following immunogenicty data request
- Some limitations of their ability to partner the two FOB programs that were previously included in NVS partnership. One was eliminated from portfolio due to commercial market reasons. 2nd, MNTA did work on it for Sandoz, who own a piece of it going forward, but don't expect significant revenue contribution from this program
- Flatly denied report from Teva that M356 needs to be reformulated (don't know what lawyer was trying to say). It was stated at hearing that MNTA intends to use a different method for measuring molecular weight (so as to not infringe Teva patents. But don't believe this change with delay the ANDA review
- No comment in response to question as to why not sell the entire company
- BLA type pathway for FOBs is workable but not MNTAs preference. Concedes "patent clearance process"
Notes: 3/7/2011 Cowen Webcast
- Copaxone review questions from FDA are going as anticipated
- Slower FDA ANDA backlog- is now 26 months vs 14 months when the ANDA was filed for Lovenox
- Have been working on FOB program for about 4 years
- Want platform partnership for FOB programs- hope within the next year, but no rush because of cashflow from lovenox
- 2nd round of partnering ongoing for M118 (a year ago they said they "couldn't afford" to fund next trial alone. They did not repeat this statement at this webcast)
- Existing biologics companies have had a disincentive to develop the platform technology that MNTA has- because they more they learn about their drugs the more regulatory problems they could have
- Lovenox shift from 45% profit share to royalty would change upon the launch of a competing generix enox, not upon approval
- No comment about the rumors of generic Copaxone trials being discussed with clinicians- but reiterated that filing was accepted as ANDA and would be no efficacy trials needed, any trials would be to confirm product identity only