Optimer Pharma OPTR Stock Research6/16/11 CBST competing CDAD drug p2 data - click here for blog post with data comparison and analysis.
8/16/11 CBST webcast at Wedbush -click for notes. 9/7/11 Baird webcast - OPTR notes - CBST notes. 9/19/11 UBS webcast - CBST notes. 9/27/11 JMP webcast - OPTR notes. 3q2011 earnings - CBST notes. |
Important Links |
Links
General Info and Outlook
- Please note- quoted passages are from OPTR 2010 10k annual report unless otherwise stated
- 2010 10k: plan to selectively in-license hospital speciality products for development and/or commercialization. Seek partners outside of core markets
- 2010 10k: Plan to seek partner for other markets such as Japan and China for Dificid. Develop US salesforce internally or thru collaborations. Target hospital-based and longterm care physicians including gastroenterologists, infectious disease specialists, and internists
- "The anti-infective market is one of the largest therapeutic categories worldwide. According to IMS Health, the combined market for prescription antibacterial drugs in 2009 was reported to be $39.7 billion worldwide. The largest antibacterial sales per region were: North America ($11.1 billion), Europe ($10.5 billion), Asia Pacific ($12.7 billion), and others ($5.4 billion). The market for anti-infective products is generally divided into two categories, nosocomial infections and community-acquired infections, which represent approximately 30% and 70% of the anti-infectives market, respectively. According to the U.S. Centers for Disease Control and Prevention, or CDC, approximately two million nosocomial infections occur annually in the United States and these infections can increase the average length of hospital stays by seven to nine days. Approximately four million nosocomial infections occur annually in Europe, three million in North America, two million in South America and two million in East Asia (excluding China). Nosocomial infections are costly to address, with an estimated annual aggregate healthcare cost in the United States and the United Kingdom of approximately $4.5 billion and $1.9 billion, respectively. In addition, in the United States, nosocomial infections cause approximately 100,000 deaths annually, making them one of the five leading causes of death in the United States. We believe that bacterial infections, especially infections caused by difficult-to-treat, drug resistant bacteria, cause or contribute to a majority of these deaths."
- As of 2/28/2011, 87 employees (23 w/ doctorates, 16 discovery, 42 clinical/RA, 9 commercialization/corporate development, 20 support/admin)
- ~$30-34m annual R&D expense 2008-2010, net loss $36-47m
- 3/2011 sold 6.9m shares at $11.25 for gross proceeds of $77.6m. Click here for press release.
- 2010 10k: cash on hand (including Astellas and 3/2011 financing) sufficient for next 18 months
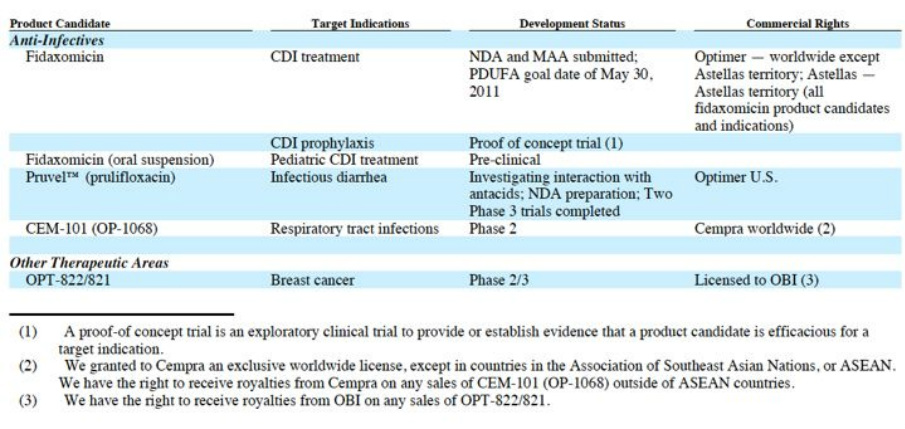
Dificid (fidaxomicin)
- Narrow spectrum macrocycle (18 member ring) antibiotic with minimal systemic absorption. Indicated for Clostridium difficile -infection, or CDI, the most common nosocomial, or hospital acquired, diarrhea
- "Pre-clinical data indicates that fidaxomicin is bactericidal and acts by inhibiting RNA polymerase, a bacterial enzyme. Fidaxomicin has also been shown to inhibit the growth of other potentially harmful bacteria such as Staphylococci, common bacteria that reside on the skin and in the GI tract, and Enterococci, common bacteria that reside in the GI tract."...these are all gram-positive bacteria
- Pursuing label expansion efforts including 1) developing an oral suspension formulation for pediatric and ICU/elderly pts who cannot swallow pills. 12/2010 granted Orphan Drug designation for pediatric population. Status: preclinical...and 2) for the prevention of CDI (prophylaxis). Status: "evaluating the possibility of conducting a proof-of-concept trial in high risk patients."
- 3 issued and 10 pending US patents. 5 issued and 69 pending foreign patents. Issued patents cover specific polymorphic form (expire 2027) and manufacturing methods (expire 2023). Pending would expire 2023-2029. 3/2011 received US method of use patent that expires 3/2027. Click here for press release.
- Basic info on CDI and treatment:
Current therapeutic options for CDI include the off-label use of metronidazole and oral vancomycin, the latter being the only FDA approved treatment for CDI. However, approximately 20% to 30% of CDI patients who initially respond to these treatments experience a clinical recurrence following cessation of the CDI treatment.
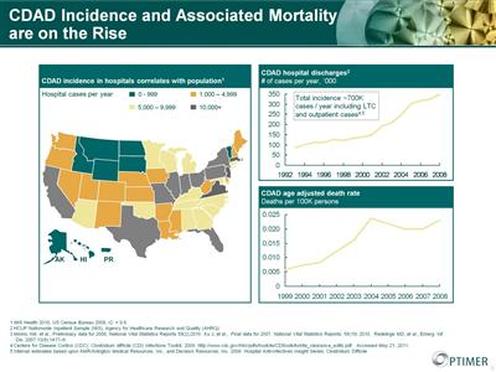
Primary risk factors for CDI include broad-spectrum antibiotic use (such as cephalosporins and fluoroquinolones), older age (over 65) and exposure to emerging hyper-virulent strains (BI/NAP1/027, 078, 001) of C. difficile. The increasing incidence of CDI, along with high rates of both treatment failures and recurrences with current therapies have resulted in greater awareness and concern about CDI among medical professionals and public health officials.
Outbreaks and illness related to C. difficile generally occur during or after therapy with broad-spectrum antibiotics. Broad-spectrum antibiotics can cause CDI by disrupting normally present gastrointestinal bacteria, or gut flora, thereby allowing C. difficile to proliferate. Studies have suggested that the use of proton pump inhibitors, or PPIs, a widely used group of heartburn drugs, may also be linked to C. difficile infections. CDI accounts for up to 33% of antibiotic-associated diarrhea incidences as well as many cases of antibiotic-associated colitis, or inflammation of the colon. C . difficile can be transmitted by direct or indirect contact with infected patients via spores that can live for months on dry surfaces.
We believe that the incidence of CDI may be higher than what is currently being reported because many hospitals are not required to and do not report incidents of CDI. For example, a survey conducted in May 2008 through August 2008 by the Association for Professionals in Infection Control and Epidemiology, or APIC, showed that 13 out of every 1,000 inpatients were either infected or colonized with C. difficile (94.4% infected) which is 6.5 to 20 times higher than previous incidence estimates.
Additionally, recent reports indicate that the incidence of community-acquired CDI cases may be increasing. For example, a study conducted in one major U.S. city and cited at the 2006 Interscience Conference on antimicrobial Agents and Chemotherapy, or ICAAC, reported that the percentage of CDI cases found to be community-acquired increased from 12% in 2003 to 22% in 2004 and to 29% in 2005. A recently published survey conducted by a Veterans Affairs hospital in North Carolina showed that 35% patients with CDI experienced onset of the disease in the community. In the community, exposure to CDI is believed to occur through household contacts of persons with diarrhea, or ingestion of contaminated food or soil. Some animals, including domesticated pets, may also carry C. difficile . According to a study cited in the New England Journal of Medicine, the increased rates of CDI and severity of the disease may be caused by a combination of factors, including the excessive use of antibiotics, a growing population over the age of 65 and the impact of new hyper-virulent strains of C. difficile . Recent CDI studies have demonstrated increasing frequency of new hyper-virulent strains, which can increase the risk of clinical failure, recurrence and mortality. The analysis of C. difficile isolates from our first Phase 3 trial presented at the 2009 ICAAC meeting demonstrated that even though the BI hyper-virulent group continues to be dominant in North America, 60% of the strains analyzed belonged to non-BI groups including G, J, K and Y groups. Hyper-virulent strains are also a growing concern in Europe.
Generally, CDI results in longer hospital stays and increases average patient cost which is often not reimbursed to the hospital. In more complicated cases of CDI, hospitalization may be prolonged by up to two weeks. A recent analysis suggests that patients with CDI have their hospital stay extended by at least 3 days compared with patients without the infection, with the incremental cost of approximately $13,700 per patient. The total annual costs associated with hospital cases of CDI in United States are estimated at $3.2 billion. According to the data presented at the 2006 ICAAC, CDI results in an estimated increase in average patient cost of over $6,000 per patient in the United Kingdom and the total projected annual cost for treating the disease in Europe is approximately $3.8 billion.
Metronidazole is generally used for patients in the United States and Europe experiencing their first episode or first recurrent episode of CDI. Metronidazole is a generic drug that is used off-label to treat CDI due to its low cost and historical efficacy. The typical treatment regimen for metronidazole is 250 mg every six hours, for a minimum of ten days. Metronidazole can be associated with numerous adverse side effects such as seizures, toxic reactions to alcohol, leukopenia, or reduction of white blood cells, neuropathy, a disease affecting one or more nerves, unpleasant taste and dry mouth.
Oral vancomycin is used in the United States and also in Europe and Japan for the treatment of CDI. As a result of its broad antibacterial activity, intravenously administered vancomycin is frequently used for certain other life-threatening infections caused by multidrug resistant bacteria such as vancomycin-resistant Enterococci, or VRE. In an effort to slow the continuing emergence of vancomycin-resistant bacteria, the medical community discourages the use of the drug for the treatment of CDI except for patients who are not responding to metronidazole, for patients with severe, life-threatening colitis or for patients with risk factors that are predictive of negative treatment outcomes.
Despite these recommendations, based on our survey of 131 U.S. hospital physicians, hospital physicians would intend to prescribe oral vancomycin approximately 37% of the time, compared to our estimate of an approximately 27% expected prescription rate per the treatment guidelines. In addition, our internal market research suggests that hospitals in the U.S. are increasing oral use of generic reconstituted intravenous vancomycin relative to the use of branded oral vancomycin capsules, or Vancocin. Oral vancomycin’s recommended treatment protocol is 125 mg or 250 mg doses every six hours, for approximately ten days"
- Limitations of current treatments:
- Limited efficacy: 19% and 28% of pts do not respond to oral vancomycin and metronidazole, respecitively - at risk to develop more serious CDI
- High Recurrence Rate: Of those who initially respond, 25-27% of pts have a recurrence after metro/vanc therapy stopped (hosptial cost estimated at $10-35k). Dificid p3 trial showed improved recurrence/relapse rates.
- Bacterial Resistance: Vanc-resistant Cdif is commonly reported, and metro-resistance Cdif has also been reported
- Adverse Side Effects of metronidazole (systemic therapy) include "seizures, toxic reactions to alcohol, leukopenia, neuropathy, unpleasant taste and dry mouth." Dificid has minimal systemic exposure and a favorable safety profile
- Metro and Vanc are broaden spectrum antibiotics and therefore deplete the normal gut flora. These commensal bacteria actully suppress Cdiff, therefore these drugs can actually facilitate CDI. Dificid minimally disrupts normal gut flora
- Dosing is required every 6 hours for at least 7 days- inconvenient and leads to lower pt compliance. Dificid is dosed twice daily and resolves diarrhea faster
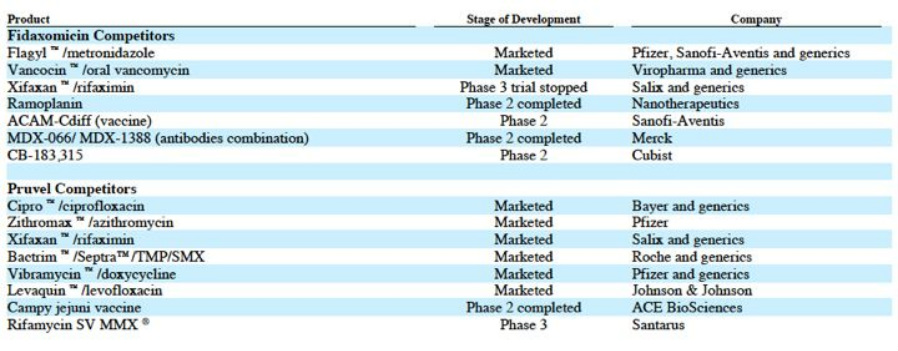
- See Chart below for additional competitor information
- 2/2007: regained worldwide rights from Par Pharma. Paid $5m milestone 6/2010 (success of second p3). Will owe 5% royalty in North American and Israel, 1.5% rest of world. Also owe 6.25% of licensing revenues outside North America and israel (so includes Astellas deal below). These payments are required for 7 years following commercial launch on country by country basis
- 2/2011 licensed to Astellas for Europe (and certain other countries in Middle East, Africa, and CIS). $69.2m upfront fee (Euro 50m), upt to Euro 115m regulatory/commercialization milestones. Royalties high teens to low twenties % on net sales. Astellas pays all future development and commercialization costs, including pending EMEA filing. OPTR also will supple drug to Astellas on cost-plus basis. Click here for press release.
- Note: antibiotic use and CDI incidence are quite low in Asia- this is presumably why Astellas did not license the drug for that territory (hat tip to @Inb4daPop)
- 4/2011 collaboration for co-promotion in US with Cubist $CBST (Click here for PR - Click here for more CBST info). OPTR will hie ~100 sales reps to target 1100 hospitals covering 70% of US CDI cases. With CBST will reach 2000 hospitals and 90% of CDI cases. Agreement is for 2 yrs. CBST receives $15m fee per year starting on first sale. Also eligible for $5m and $12.5m in milestones during 1st and 2nd yr, respectively, plus a portion of gross sales above certain thresholds.
- 5/2010 supply agreement with Biocon for API
- 7/2005 phase 2a trial completed: Open label, dose ranging in 48 pts at 5 sites.
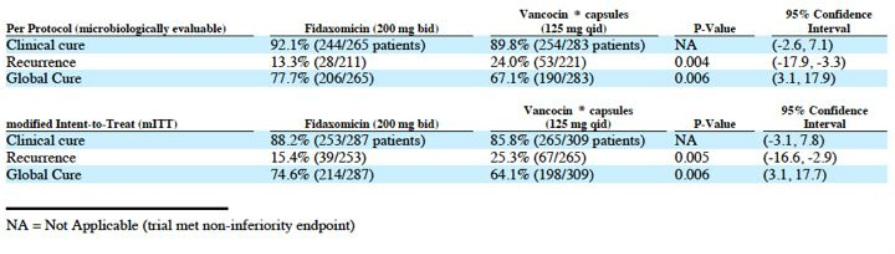
- 1st phase 2b/3 trial results reported 11/2008 (200mg twice daily vs vanc at recommended dose of 500 mg/day. 629 adults at >100 sites in North America). Primary endpoint was "clinical cure defined as patients requiring no further CDI therapy two days after completion of study medication, as determined by the treating physicians." Secondary endpoint was recurrence within 4 weeks. Global cure was exporatory endpoint in this trial. Transitioned to phase 3 in 3/2007 after 93 pts and interim blinded safety analysis. Results published in NEJM 2/2011 - see below for chart summarizing key data
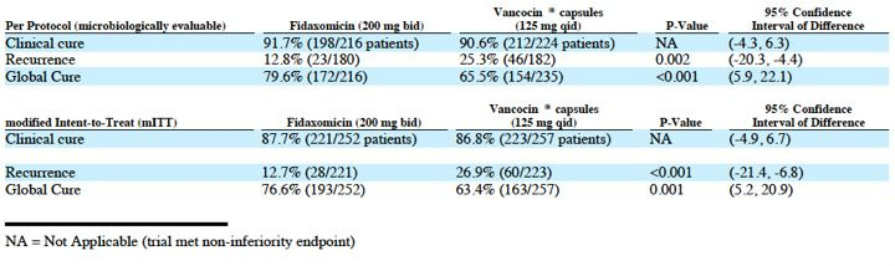
- 2nd phase 3 trial results reported 2/2010 (200 mg twice daily vs vancomycin 125 mg every 6 hrs, 535 adults at >100 sites). Primary endpoint was "clinical cure defined as patients requiring no further CDI therapy two days after completion of study medication, as determined by the treating physicians" Secondary endpoints were recurrence during subsequent 4 weeks and global cure (no recurrence in subsequent 4 week period) - see below for chart summarizing key data
- 9/2010 (ICAAC) reported additional data from phase 3 trials. In pts receiving other antibiotics in addition to Vanc or Dificid (28%), Dificid achieved significantly higher cure rate (90% vs 79%), lower recurrence rate (17% vs 29%), and higher global cure rate (73% vs 59%). In pts w/o concomitant antibiotics, Dificid-treated pts had much lower recurrence rate (12% vs 23% for vanc)
- Filed for EU approval 7/2010, accepted 8/2010
- 9/2010 started rolling FDA NDA (received fast track status for CDI), completed 11/2010, accepted 1/2011, PDUFA 5/30/2011 and approved 5/27/2011 - click here for press release -- click here for product label info -- Click here for NY Times article.
- Click here for a nice writeup post-approval, including summarizing data and comments from doctors. They were generlaly very excited about data, but won't automatically use the drug in first line. "It’s promising that the recurrence rate is lower with fidaxomicin than with vancomycin, but at this time that doesn’t translate into knowing that there is efficacy in treating recurrences” - Dr Christin Surawicz (University of Washington)...."But the fact is that fidaxomicin is as good as vancomycin in treating C. difficile, while doing a better job at sparing the components of the normal gut flora—which in turn appears to be translated into a lower recurrence rate. So it may be that physicians will use fidaxomicin in patients with recurrent disease, although specific trials of this indication are needed" - Dr Thomas Louie (University of Calgary)...."It’s expensive, so for patients with mild disease, I might not consider it as a first-line therapy” - Dr Kathleen Mullane (Tufts)
- FDA Anti-Infectives Drugs Advisory committee 4/5/2011. Received unanimous recommendation for approval. Click here for press release.
- Risk factor: "Generic antibiotic therapies typically are sold at lower prices than branded antibiotics and are generally preferred by managed care providers of health services. For example, because metronidazole and generic vancomycin “slurry” are available at such a low price, we believe it may be difficult to sell fidaxomicin as a first-line therapy for the treatment of CDI other than in certain limited circumstances, such as in patients at high risk of recurrence. If we or our collaborators are unable to demonstrate to physicians and patients that, based on experience, clinical data, side-effect profiles and other factors, our products are preferable to these generic antibiotic therapies, we may never generate meaningful product revenues. In addition, many antibiotics experience bacterial resistance over time because of their continued use. There can be no guarantee that bacteria would not develop resistance to fidaxomicin"
Competitor: Vancocin from Viropharma $VPHM
- A key issue in the US (VPHM does not have rights in other regions) CDI market is the potential for the oral vancomycin product (Vancocin from $VPHM) to go generic in the near future. See below for my notes on this issue from the $VPHM 2010 10k annual report.
- Vancocin is FDA approved to "treat antibiotic-associated pseudomembranous colitis caused by Clostridium difficile infection (CDI), or C. difficile, and enterocolitis caused by Staphylococcus aureus [rare, contributes small % of sales], including methicillin-resistant strains”
- Net sales of Vancocin were $259.6m in 2010, $213.1m in 2009, and $231.6 in 2008
- "Vancocin net sales during the three months ended March 31, 2011 were $69 million, or a 24 percent increase over the respective period in 2010 due to net realized price and volume growth...Note that VPHM did not give 2011 Vancocin sales guidance (but did give specific guidance for their other key product Cinryze)
- # of Vancocin prescriptions decreased 5.8% in 2010 vs 2009 and decreased 9.3% in 2009 vs 2008
- Unit sales of Vancocin decreased 1.4% in 2010 vs 2009 and decreased 17% in 2009 vs 2008
- So you can see from the above that the unit's used per pt have increased (mirrors comments made by OPTR management), plus price of Vancocin has increased (I didn't bother to do the math to get exact price hike...)
- "CDI is an infection of the GI tract. The clinical manifestations, ranging from diarrhea to toxicmegacolon and sometimes death, are a result of toxins produced by the bacterium that cause inflammation in the colon. Hospitalized patients, those residing in long-term care centers, those greater than 65 years of age, and patients that have received broad-spectrum antibiotic therapy, are at greatest risk to acquire CDI. CDI is not a nationally reportable disease and as such it is difficult to estimate the actual incidence of disease with precision. Based on reports from the Centers for Disease Control and Prevention (CDC) and peer-reviewed publications, we estimate that at least 500,000 patients were affected by CDI in 2008. From 2004 to 2008, many clinicians reported treating increasing numbers of patients with severe CDI and increased mortality rates. Clinicians have also noted that some patients progressed from mild/moderate disease to severe disease or death more rapidly than had previously been observed. The overall incidence of CDI may have plateaued or even decreased since 2008, however reliable data on current incidence are limited. Although the causes for this change in CDI remain under active investigation, the CDC has postulated that a combination of changes in antibiotic use and infection control practices, along with the emergence of a hypervirulent strain of C. difficile, are likely contributors to the increased incidence seen during the 2004 to 2008 time period. As of late 2008, this strain (referred to as the toxinotype III, BI, or NAP1/027 strain) has been identified in at least 40 states in the U.S."
- "Vancocin is the only drug approved by the FDA for the treatment of antibiotic-associated pseudomembranous colitis caused by C. difficile. Historically metronidazole has been commonly used as first-line treatment for CDI, while Vancocin has been reserved for those patients who have failed metronidazole, have recurrent disease, or who are suffering from severe CDI. We believe that changes in the epidemiology of CDI, in particular the increasing frequency of severe disease, and data suggesting that failure or relapse occur more commonly in patients treated with metronidazole have led to an increase in the use of Vancocin."
- "For patients suffering from severe cases of Clostridium difficile infections or CDI, our product Vancocin® (vancomycin HCl, USP) remains the gold standard. Our disagreement with the proposed path forward to demonstrate bioequivalence of generic copies of Vancocin continues and the final outcome of this disagreement to this day still is not clear. Rest assured that we will continue to do everything we can to ensure that the best interests and safety of patients are being served. And, though Vancocin is the trusted drug to treat primary CDI, no product available today or in late stage development can prevent recurrence."
- "In November 2004, we acquired all rights in the U.S. and its territories to manufacture, market and sell Vancocin, the oral capsule formulation of vancomycin hydrochloride, as well as rights to certain related vancomycin products, from Lilly. Vancocin is a potent antibiotic approved by the FDA to treat antibiotic-associated pseudomembranous colitis caused by C. difficile and enterocolitis caused by S. aureus, including methicillinresistant strains. Lilly retained its rights to vancomycin outside of the U.S. and its territories. We paid Lilly an upfront cash payment of $116.0 million and we are obligated to pay additional purchase price consideration based on annual net sales of Vancocin through 2011. As of December 31, 2010, we have paid an aggregate of $44.1 million to Lilly in additional purchase price consideration, as our net sales of Vancocin surpassed the maximum obligation level of $65 million in 2005 through 2010. We are obligated to pay Lilly additional amounts based on 35% of annual net sales between $45 and $65 million of Vancocin in 2011. No additional payments are due to Lilly on net sales of Vancocin below or above the net sales levels. We account for additional purchase price consideration as contingent consideration and record an adjustment to the carrying amount of the related intangible assets and a cumulative adjustment to the intangible amortization upon achievement of the related sales milestones. In the event we develop any product line extensions, revive discontinued vancomycin product lines (injectable or oral solutions), make improvements of existing products, or expand the label to cover new indications, Lilly would receive a royalty on net sales on these additional products for a predetermined time period"
- “the last core patent protecting vancocin expired in 1996…In order to continue to obtain commercial benefits from Vancocin, we will rely on product manufacturing trade secrets, know-how and related non-patent intellectual property, and regulatory barriers to competitive products. We own three pending U.S. patent applications covering vancomycin related technology."
- "We have become aware of information suggesting that other potential competitors are attempting to develop a competing generic product. For example, multiple generic manufacturers have publicly stated that they have filed to receive product approval and commence a marketing launch of a generic version of oral Vancocin. We are not able to predict the time period in which a generic drug may enter the market, as this timing will be affected by a number of factors, including:
--the nature of any clinical trials which are required, if any;
--the timing of filing an Abbreviated New Drug Application, or an ANDA, the amount of time required by the FDA to review the ANDA and whether a generic drug application is afforded an accelerated review by the FDA;
--the specific formulation of drug for which approval is being sought; and
--the time required to develop appropriate manufacturing procedures.
- "On March 17, 2006, we learned that the OGD changed its approach regarding the conditions that must be met in order for a generic drug applicant to request a waiver of in-vivo bioequivalence testing for vancomycin hydrochloride capsules. Specifically, we were informed that a generic applicant may be able to request such a waiver provided that dissolution testing demonstrates that the test product is rapidly dissolving at certain specified conditions. This deviated from our understanding of OGD’s historical practices which would require, for a poorly-absorbed, locally acting gastrointestinal drug (such as Vancocin) a demonstration of bioequivalence through clinical studies or a demonstration of bioequivalence using an appropriately validated in-vitro methodology."
- "On March 17, 2006, we filed a Petition for Stay of Action with the FDA regarding the requirements for waivers of in-vivo bioequivalence testing for Vancocin, and we have amended that petition several times through additional filings in support of our opposition to any approach that does not require rigorous scientific methods to demonstrate a rate and extent of drug release to the site of action consistent with good medicine and science."
- "In December 2008, the FDA changed OGD’s 2006 bioequivalence recommendation by issuing draft guidance for establishing bioequivalence to Vancocin which would permit generic products that have the same inactive ingredients in the same quantities as Vancocin, or Q1 and Q2 the same, and that meet certain other conditions, to demonstrate bioequivalence through comparative dissolution testing. Under this latest proposed method, any generic product that is not Q1 and Q2 the same as Vancocin would need to conduct an in vivo study with clinical endpoints to demonstrate bioequivalence with Vancocin."
- "The FDA convened a meeting of its Advisory Committee for Pharmaceutical Science and Clinical Pharmacology to discuss bioequivalence recommendations for oral vancomycin hydrochloride capsule drug products on August 4, 2009. The Advisory Committee was asked if the proposed guidelines are sufficient for establishing bioequivalence for generic vancomycin oral capsules. The Advisory Committee voted unanimously in favor of the component of the proposed OGD recommendation that requires bioequivalence to be demonstrated through comparable dissolution in media of pH 1.2, 4.5 and 6.8 for potential vancomycin HCl capsule generic products"
- "On September 10, 2010, we brought an action for declaratory relief (the “Complaint”) against the Food and Drug Administration, Margaret A. Hamburg, M.D., in her official capacity as Commissioner of Food and Drug Administration, the United States Department of Health and Human Services (“HHS”), and Kathleen Sebelius, in her official capacity as Secretary of HHS, (collectively “FDA”) in the United States District Court for the District of Columbia (the “Court”). Pursuant to the Complaint, we are seeking review under the Administrative Procedure Act (“APA”) of the FDA’s decision to change its regulations to abandon its longstanding rule that applicants for an Abbreviated New Drug Application (“ANDA”) seeking waiver of the requirement of 21 C.F.R. § 320.21 (2010) that bioequivalence must be demonstrated by in vivo evidence must satisfy one of the enumerated waiver criteria set forth in 21 C.F.R. § 320.22. We have requested that the Court determine that (i) the plain reading of FDA’s regulations requires an ANDA applicant seeking a waiver of the in vivo bioequivalence testing requirement to first meet one of the criteria set forth in 21 C.F.R. § 320.22 and (ii) FDA’s amendment of its regulations governing waiver of submission of in vivo bioequivalence evidence, without engaging in notice-and-comment rulemaking, violates 5 U.S.C. § 553 of the APA and is therefore invalid. Subsequently, the DOJ filed a motion to dismiss the Complaint and the parties have filed briefs supporting their positions regarding the motion to dismiss"
Potential Competitor: CB 183,315 from $CBST
- Cubist $CBST announced phase 2 data in June 2011 - click here for my blog post with analysis
- Find more info on Cubist and CB 183,315 on the CBST info page.
- See table below for comparison of this drug to Dificid (fidaxomicin)
Potential competitor: VP20621 from $VPHM
- I was not familiar with this program until I began researching Dificid and Vancocin. This program has failure written all over in my opinion based on my preliminary research and "gut" feeling ;)
- “Despite the historically high rate of clinical success with Vancocin® (vancomycin HCl, USP), its Achilles’ heel has been the approximately 20 to 30 percent of incidents of recurrence. The goal of ViroPharma’s novel VP20621 (non-toxigenic C. difficile) program is to prevent such recurrence of disease. ViroPharma acquired VP20621 in February 2006 from Dr. Dale Gerding, of the Hines VA. The concept behind this novel treatment approach aims to prevent disease recurrence and involves the oral administration of non-toxin producing spores of C. difficile following initial treatment of acute CDI. The underlying concept is to first treat the disease with an effective product like Vancocin and eradicate the dangerous toxin-producing C. difficile which causes severe CDI. The treated patient could potentially then be dosed with oral VP20621 to re-colonize the GI tract and prevent the “bad”, or toxic bugs from re-infecting the colonuntil normal GI flora returns and the patient is no longer susceptible to disease. We presented data in late 2010 which was very encouraging and showed that we could achieve potentially protective colonization the GI tract of subjects at risk of infection with toxigenic C. difficile, and in December of 2010, we filed our U.S. IND. We intend to begin Phase 2 studies in the first half of this year.”
- "We are also developing VP20621 to prevent recurrence of CDI using a novel non-antibiotic approach. On August 6, 2009 we announced that dosing had begun in the Phase 1 clinical trial for VP20621 to determine the safety and tolerability of VP20621 dosed orally as a single and as repeat escalating doses in healthy young and older adults. We have successfully completed our Phase 1 clinical trial, filed an IND in December of 2010 and anticipate moving the compound into a Phase 2 clinical trial in the first half of 2011.”
- “In February 2006, we announced that we had entered into a licensing agreement with Dr. Dale Gerding, of the Hines VA, for the rights to develop a non-toxigenic strain of C. difficile (VP 20621) for the treatment and prevention of CDI. Under the license agreement, we are required to make royalty payments to Dr. Gerding based on a low single digit percentage of our net sales of the product. If certain milestones are achieved, we will be obligated to pay Dr. Gerding additional milestone payments if and when certain regulatory developments are achieved, in an aggregate amount equal to $850,000 in total, with no single milestone payment exceeding $250,000. The license agreement will remain in effect for ten years from the date any product is first commercialized, on a country-by-country basis, unless earlier terminated. The agreement contains a standard early termination provision which provides for early termination by either party in the event certain conditions have occurred, including, but not limited to, either party’s breach of the agreement, either party’s filing for bankruptcy or either party making an assignment for the benefit of its creditors."
- "We plan to initially focus our efforts on the opportunity to prevent recurrence of CDI, using oral administration of VP 20621 spores. According to published literature, approximately 20 to 30 percent of patients suffering from CDI will have at least one episode of relapse of disease. The goal of this VP20621 program is to prevent such recurrence of disease using a novel non-antibiotic approach. The underlying concept of this approach is to first treat the disease with an effective product like Vancocin and eradicate the dangerous toxin-producing C. difficile which causes severe CDI. The treated patient could potentially then be dosed with oral VP20621 to re-colonize the GI tract and prevent the pathogenic C. difficile bacteria from re-infecting the colon until normal GI flora returns and the patient is no longer susceptible to disease."
- In September 2010 we announced results of a completed Phase 1 study which was designed to determine the safety and tolerability of VP20621 dosed orally as single and repeat escalating doses in healthy young (18 to 45 years of age) and older (60 years of age and older) adults. After VP20621 was shown to be generally well tolerated following single and repeat doses in younger and older healthy subjects, we also performed repeat dosing in older adults following exposure to oral antibiotic. Subjects above 60 years of age were pre-dosed with oral vancomycin to disrupt their gastrointestinal flora and render them potentially susceptible to C. difficile colonization; these subjects were subsequently given either placebo or VP20621 doses of 10(4), 10(6), or 10(8) spores once daily for 14 days. The study demonstrated that VP20621 was generally well tolerated at all dose levels; there were no serious or severe adverse events, and no discontinuations from study drug due to adverse events. All 27 volunteers (100%) who were given VP20621 had positive non-toxigenic C. difficile stool cultures by day 6, suggesting that VP20621 rapidly colonizes the susceptible GI tract. No subject dosed with VP20621 tested positive for toxin-producing strains of C. difficile during the 28-day study period. By comparison, 5 of 9 subjects (56%) who received placebo (i.e. did not receive VP20621) tested positive for either toxin-negative or toxin-positive C. difficile during the study period.
- "Based on these results, we anticipate moving the compound into a Phase 2 clinical trial for the prevention of recurrence of CDI in patients recently treated with an antibiotic for CDI. Separately, we are also evaluating the potential for VP20621 to be used as primary prevention for CDI in certain high-risk populations"
2010 10k notes and other pipeline assets
- "Pruvel is a prodrug in the fluoroquinolone class of antibiotics, a widely-used class of broad-spectrum antibiotics. We are developing Pruvel as a treatment for infectious diarrhea. In July 2008, we reported positive top-line data from the first Phase 3 trial conducted in Mexico and Peru and in February 2009, we reported positive top-line data from the second Phase 3 trial conducted in India, Guatemala and Mexico. The top-line analysis of data from these studies showed that Pruvel met the primary endpoint of time to last unformed stool, or TLUS, compared to placebo. On November 9, 2010, due to a higher than expected incidence of cutaneous rash during the course of a study of the possible interaction between Pruvel and antacids, we informed the FDA that we were voluntarily terminating the research study. All reported events of cutaneous rash from the drug interaction study were mild or moderate in severity and required little or no treatment and all resolved completely. We are currently conducting an investigation into the cause of the rash prior to initiating any new study with Pruvel. Rashes are a known and infrequent side effect of fluoroquinolone antibiotics, such as Pruvel, and rashes occurred at or below the rate generally expected for other fluoroquinolones in both of our previous Pruvel Phase 3 clinical trials. Pending the results of our investigation, we are not currently able to estimate when we will initiate any new study or the extent of the delay in our planned submission of an NDA for Pruvel. We currently hold rights to commercialize Pruvel in the United States, and it is sold by other parties in Japan, Italy and certain other European countries."
- 6/2004 in-licensed Pruvel from Nipppon Shinyakuc for the US. $1m upfront fee and $1m milestone due on NDA submission
- "For our other product candidate Pruvel, we have licensed one issued U.S. patent from Nippon Shinyaku which covers the compound prulifloxacin; however, this patent expired in February 2009. Another U.S. patent covering the novel polymorphic form of the key intermediate in the manufacturing process was issued in 2009 and will expire in 2023. The remainder of our patents and patent applications, and licensed patents and patent applications, relate to our other products and technology, and expire between 2015 and 2023."
- "We are developing additional product candidates using our proprietary technology, including our Optimer One-Pot Synthesis, or OPopS™ drug discovery platform. OPopS is a computer-aided technology that allows the development of potential drug candidates through carbohydrate mediated medicinal chemistry and enables the rapid synthesis of a wide variety of proprietary molecules. It includes GlycoOptimization, which enables the modification of a carbohydrate group on an existing drug to improve its properties, and De Novo Glycosylation, which introduces new carbohydrate groups on existing drugs to create new patentable compounds with improvement of pharmacokinetics."
- "We previously acquired exclusive rights to OPT-822/821, a combination of a novel carbohydrate-based cancer immunotherapy together with an adjuvant, which we licensed from Memorial Sloan-Kettering Cancer Center, or MSKCC [ in 2002]. In October 2009, we assigned to our subsidiary, Optimer Biotechnology, Inc., or OBI, certain of our patent rights and know-how related to OPT-822/821 and also assigned to OBI our rights and obligations under a related license agreement with MSKCC. In April 2010, OBI filed an investigational new drug application, or IND, in Taiwan for OPT-822/821, and in January 2011, OBI initiated a Phase 2/3 clinical trial for the treatment of metastatic breast cancer which it intends to conduct in Taiwan, South Korea, Hong-Kong and Singapore [expected up to 342 pts, primary endpt is PFS, secondary endpoint OS, cyclophosphamide+/-OPT-822/821 2:1 randomized vs placebo]. Other potential indications for OPT-822/821 are being evaluated. We have the right to receive up to $10 million from OBI in future milestone payments related to the development of OPT-822/821 as well as royalties on net sales of this product candidate. In February 2011, pursuant to an amendment to an October 2009 financing agreement, OBI sold newly-issued shares of its common stock for gross proceeds of approximately 462.0 million New Taiwan Dollars (approximately $15.5 million based on then- current exchange rates). We purchased 277.2 million New Taiwan Dollars (approximately $9.3 million based on then-current exchange rates) of the shares issued in the financing, such that we currently maintain a 60% equity interest in OBI."
- OBI also had rights to OPT-88, but further development was abandoned in 2/2011
- "Carbohydrate antigens are known to stimulate the immune response against cancer cells in the body. We have applied our OPopS technology to manufacture effectively complex carbohydrate cancer antigens, including Globo-H, a prominent antigen in breast cancer cells, and sialyl Lewis a, an antigen in breast and small lung cancer cells. OPT-822/821 is a novel cancer immunotherapy and is composed of Globo H linked to a protein carrier. MSKCC completed Phase 1 safety studies of OPT-822/821 in prostate cancer patients and breast cancer patients in 1999 and 2001, respectively. In these studies, OPT-822/821 appeared to be well tolerated and to stimulate responses to tumor antigens. Twenty-one of 27 metastatic breast cancer patients treated with OPT-822/821 in the study survived after four years, with 48% of patients surviving more than nine years following completion of the Phase 1 safety study."
- CEM-101 (aka OP-1068): macrolide antibiotic effective against resistant gram-positive bacterial strains. The drug part of class of 500 molecules licensed to Cempra Pharma in 3/2006. Received undisclosed equity stake as upfront, received $0.5m milestone 7/2010, eligible for up to $25m more milestones plus royalties. Phase 1 completed and plans for phase 2 trial in moderate to severe community-acquired bacteria pneumonia announced