Myrexis MYRX: Cancer metabolism inhibitor and oral anti-interferon programs
MPC-9528 Cancer Metabolism inhibitor
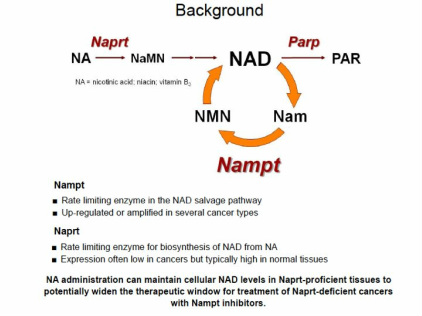
- Oral or IV, Nicotinamide phosphoribosyltransferase (Nampt) inhibitor with picomolar potency, blocks recycling of and greatly reduces Nicotinamide Adenine Dinucleotide (NAD) levels
- COM patent has been filed
- 4/5/11 webcast: hope to file IND later in 2011 (has slipped from 9/2010 and 11/2010 guidance for IND 1h2011)
- 6/6/11 webcast: first mention of MPC-8640 prodrug (unless maybe it was on an AACR poster)
- Because NAD depletion leads to inhibition of glycolysis, energy deprivation and cell death,while NAD in normal tissues is less affected, MPC-9528 is part of new field/class of cancer metabolism inhibitors
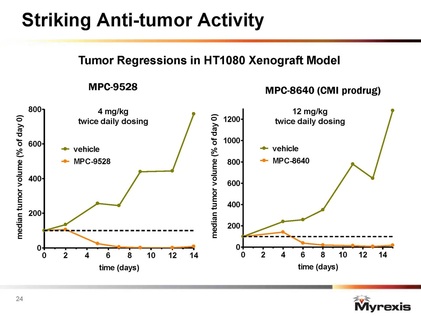
- Kills every tumor cell line tested to date in vitro, synergy w/ PARP inhibitors and other undisclosed agents, dramatic regressions in multiple preclinical tumor models
- 6/6/11 webcast: significant synergy w/ DNA damaging agents like 5-FU, temozolomide, and PARP inhibtiors- points towards CRC as first indication (new comment)
- 6/2011 ASCO presented preclinical data: click here for blog post with abstract and notes.
- Niacin (aka nicotinic acid aka Vitamin B3) completely blocks the reduction of NAD by MPC-9528 and its preclinical effects
- "Nicotinic acid is converted to NAD through an alternative pathway that is dependent upon the enzyme Nicotinic acid phosphoribosyltransferase (Naprt1) which does not involve Nampt. Our studies have found that approximately 40% of tumor cell lines are deficient in Naprt1 and in these cells, nicotinic acid had little to no effect on MPC-9528 tumoricidal activity. Furthermore, in animal model studies we found that a combination of nicotinic acid with MPC-9528 increased the tolerated dose of MPC-9528 while still causing tumoricidal effects on tumors deficient in Naprt1. This demonstrates the potential to increase the therapeutic index of MPC-9528 by combining it with nicotinic acid to treat patients with tumors that are deficient in Naprt1. A diagnostic method designed to measure Naprt1 expression could be used to identify those patients with Naprt1 deficient tumors that are most likely to benefit from this combination therapy." (1q2011 10q)
- 2/9/11 and 5/10/11 webcasts: companion diagnostic approach (see details above) will be used to decrease the size and cost of clinical trials
- 5/10/11 webcast: Very close to opening IND...may be better off keeping compound thru IND and maybe p1 before partnering. (Previously in Fall 2010 had stated that they were in active partnering discussions). At least one big pharma is hiring head of Cancer Metabolism. Now trying to establish what is best way to develop CM inhibitors.
- 6/6/11 webcast slides: pre-clinical IND studies ongoing, next step is IND filing.
- Click here to download 2011 AACR poster re co-administration w/ nicotinic acid
- Click here to download 2011 AACR poster on improved side effects w/ NA and sex-specific PK
- Click here to download 2011 ASCO poster re MPC-9526 activity in xenograft models.
MPI-4885520 Oral anti-interferon
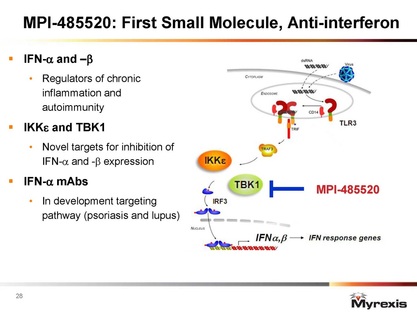
- Small molecule drug that inhibits interferon response downstream of IKK epsilon and TBK1 heterodimer.
- 6/6/11 webcast: don't know of anyone else with medicinal chemistry program against this target. Highly selective - no effect on TNFa pathway.
- Have 2 published patents
- 2/9/11, 4/5/11 and 5/10/11 webcasts: IKK-epsilon is oncogene in breast cancer, also key regulator of innate immunity (key regulatory in toll-like receptor TLR cascade, prevents downstream activation of interferons IFN-a&b)- program morphed from cancer to into looking at for autoimmune diseases. Lead compound has picomolar activity, orally bioavailable and is highly selective, in process of identifying best lead for IND (is a goal for next 12 months). don't know of anyone else with this target. have COM patents on leads. Plan to partner and develop leads for other indications.
- 5/10/11 webcast: This program is outside scope of company. Regard this as a program, not just a single compound. Initial preclinical RA study prompted by potential partners...even though their first interest is in lupus, takes 1 yr to generate preclinical lupus model data (6/6/11 webcast might have sounded like they are working on this in-house now). Collagen induced RA has been shown to be predictive for human activity. In partnering discussions for the program, series of IND candidates starting with lupus, then IBD, psoriasis. don't wish to take into IND alone because outside focus.
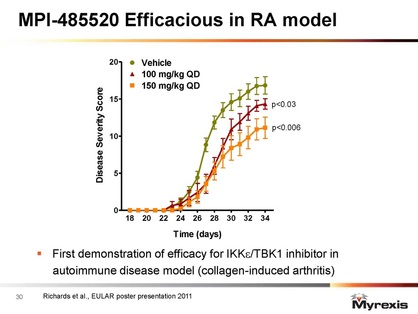
- 5/2011: positive preclinical data presented at EULAR meeting. "In the well characterized collagen-induced mouse model of arthritis, mice treated with MPI-0485520 [once daily oral dose 100 or 150 mg/kg] showed a dose-dependent and statistically significant reduction in the severity of clinical symptoms and paw and joint histopathology, as well as lower weight loss compared to control mice." Click here for PR.
- 4/5/11 webcast: also did once daily dosing (half life is an hour) treatment in model of established disease- achieved statistically-significant results w/ lower dose- done by potential partner and not yet allowed show data
- 6/6/11 webcast slides: preclinical IND studies ongoing, next step is IND enabling studies (so this program is behind the cancer metabolism program)
- 2/9/11 webcast: This program is focus of active partnering discussions (have terms sheets), MYRX will not take this program into IND/phase 1 studies without partner
Other Preclinical Programs
- At last update, company said none of these would enter clinic without partner (but multiple are "IND ready").
- Unsure of their status following reduction in force and stated focus on above 4 programs
- MPI-479605 (TTK Inhibitor, data 11/09 aacr boston)
- MPI-461359 (orally avail HIV maturation inhibitor)
- several cancer kinase targeted lead optimization projects.
MPC-4326 HIV maturation inhibitor
- 6/2010: suspended development for commercial reasons [portfolio review- FDA keeps raising bar for size and length of trials- costs 2x and more time than cancer for 1/2 the market] and will seek to outlicense
- HIV maturation inhibitor- first in class/unique MOA-could add to SOC, solid tablet formulation, fast track status
- completed p2 interaction studies w/ other HIV meds positive data 9/09, 24 week (% ptsw/ undetactable virus) two-arm (maybe multiple doses?)
- controlled p2b initiated 12/09- goal- noninferiority to truvada, recruit pts failing current therapies in each of 3 classes. 60% of strains sensitive [based on gag polymorphism-would seek premium pricing], enrich trial w/ companion viral genotyping test (10 day run in then only randomize responders, 2 doses vs soc triple therapy, 132 pts for 24 wks, complete 1q10).
- 740 pts (180 hiv) so far tx'd. stat-sig p2 viral load reduction top 3 of 4 doses [25, 50,100,200] vs placebo, 10 days liquid dosing, then measure on day 11.
- 2nd p2 in combo started w/ tablet then switched to liquid, id'd responders vs non. recent p2 w/ new 100 mg tablet formulation (acceptable bioavail and stability-drug interaction p1 pk data presented 2/10).
- MPC9055 backup program is ready to enter p2.
- Acq 1/09 from Panacos for 7m (no future obligations)- b/c was 18 months ahead in devel of myrx's own related compound-knew how to solve formulation problems (use birch bark as raw material and have patent til 2026 on synthesis from here to drug- reduce COGs from 50% to 5% of selling price).
- partnering discussion w/ usual suspects in HIV.
- Last patent expires 2015 (must refer to COM based on above notes)