Spectrum Pharmaceuticals SPPI Stock ResearchWebsite link: www.sppirx.com
Last updates: 4q2010 results 3/21/11: click here for PR - Click here for my CC notes Fusilev ready-to-use formulation approved by FDA: click here for press release Fusilev FDA approval for 2nd/3rd line colorectal cancer use: click here for PR 1q2011 results: click here for press release - Click here for my CC notes 6/7/11 Jefferies webcast - click here for my notes 9/8/11 Stifel Nicolaud webcast - click here for my notes. 9/12/11 Rodman and Renshaw webcast - click here for my notes. 9/28/11 Jefferies webcast - click here for my notes. 5/2012 ASCO abstracts - click for blog. 7/2012 JMP webcast - click for notes. Links: Click here for archive of all blog posts related to SPPI. Click here to download company summary from 2/2011 BIO CEO conference. |
|
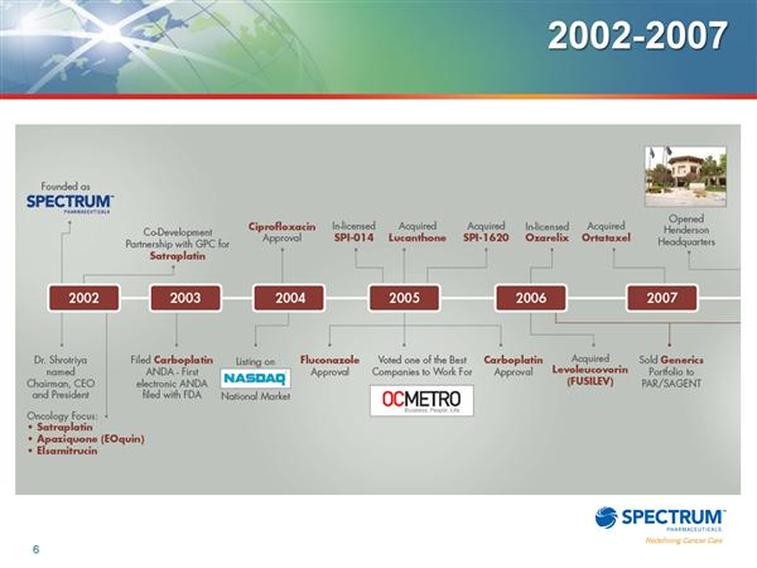
General Info
- 1q2011 10q: cash on hand sufficient through 12-18 months (ie, 3/2012 to 9/2012), but may raise money, especially in conjunction with an acquisition or in-licensing (see below)
- 1q2011 10q: expect net cash use for R&D to be $30-40m in 2011, not counting SG&A or in-licensing
- 1q2011 10q: expect SG&A to be at pace of ~$12.8m per qtr for 2011
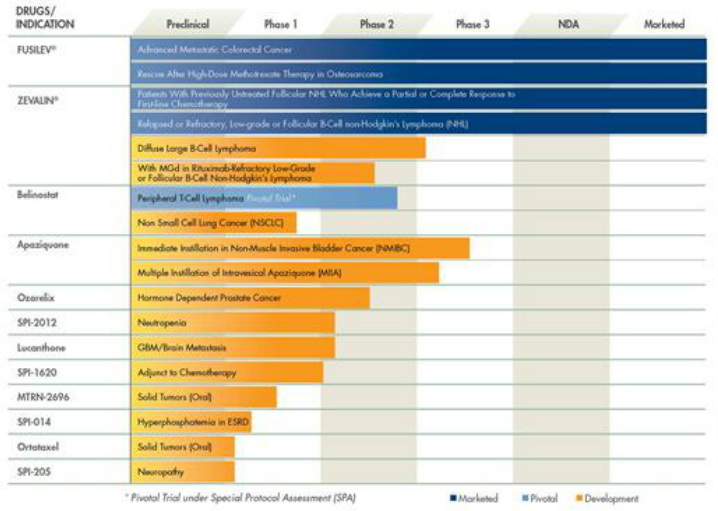
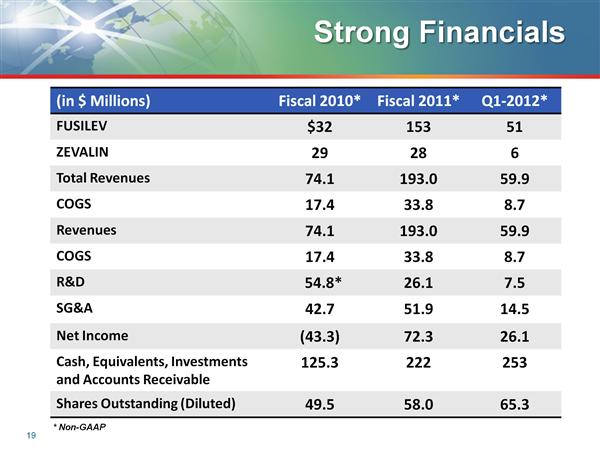
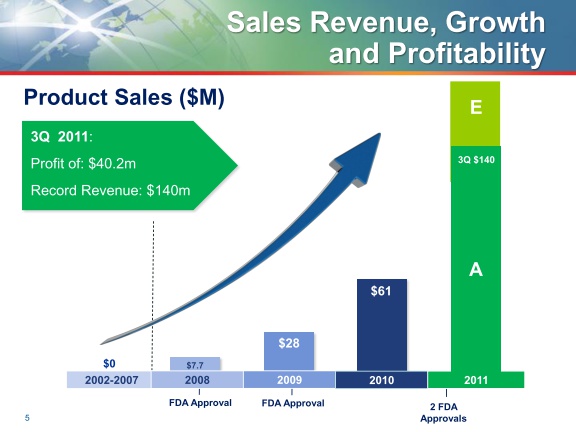
- 1q2011: $34.6m sales of Fusilev and $5.9m of Zevalin (down y/y and sequentially)
- No debt.
- June 2011: authorized up to $25m share repurchase program thru the end of 2012 (though I am on record of being skeptical of this being anything more than a PR stunt)
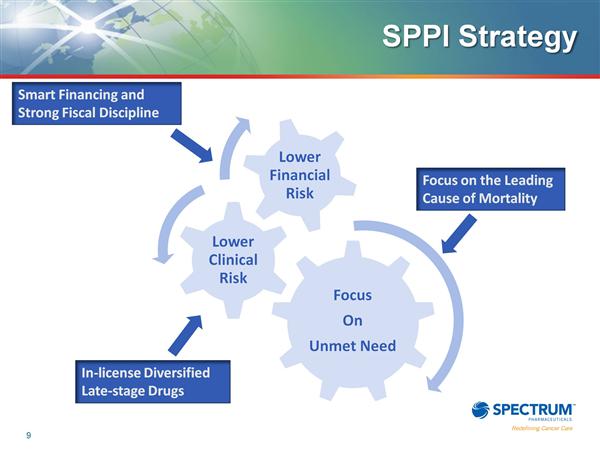
- 1q2011 10q: "It is our goal to identify new strategic opportunities that will create strong synergies with our currently marketed drugs and identify and pursue partnerships for out-licensing certain of our drugs in development. To this end, we will continue to explore strategic collaborations as these relate to drugs that are either in advanced clinical trials or are currently on the market. We believe that such opportunistic collaborations will provide synergies with respect to how we deploy our internal resources. In this regard, we intend to identify and secure drugs that have significant growth potential either through enhanced marketing and sales efforts or through pursuit of additional clinical development." [This goal is likely behind the recent approval to increase the # of authorized shares to issue. SPPI does not need to raise money for current programs, but desires to expand with more clinical or commercial assets, and if they do so they would most likely issue shares to pay for such acquisitions]
- Renewed lease 7/09 for 7 years.
- 9/2009 raised $50m at $7.55 including 0.5 warrants w/ only 3 month lifespan (recd $47.5m net).
- No drug discovery (says is bad business model to compete w/ big companies).
- Added to russell 2000 6/2009.
- Don't use clinical research organizations (CROs_ to run their trials- more expensive but almost no turnover among their 50 employees that do this function.
- Have established Canadian and Indian subsidiaries to facilitate running clinical trial sites in those countries