Spectrum Pharma SPPI Stock Research: Zevalin page
Zevalin
- Note that Zevalin always used to be the top of the pipeline chart featured program that received the bulk of the company's attention. However, anemic sales growth and the windfall sales of Fusilev thanks to the generic shortage has changed that, and now the company prominently features Fusilev. It remains to be seen if the sales will persist once generic competition is restored.
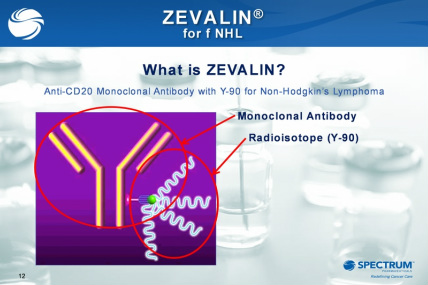
- single dose treatment, target cancer with anti-CD20 antibody plus itrium 90 "payload" -kills targeted and neighboring cells; now own 100% of US rights (first acquired 12/2008)
- Variety of licensed patents expiring 2009 to 2015
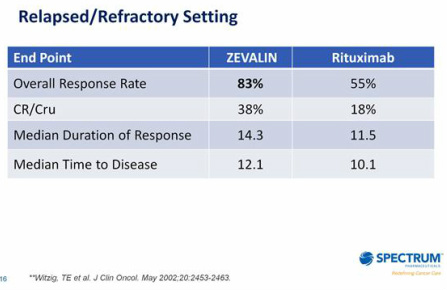
- competes w/ Rituxan, Bexxar, and Treanda - which must be given every 6-8 wks.
- Product is also independently sold by Bayer in Europe and Fuji in Japan
- 9/2009 received FDA approved for first-line setting: Now approved for all lines of NHL therapy (from <1000 to 46000 pts now- goal is 100s of M sales) - previously was only used in salvage setting and this perception has proven difficult to break
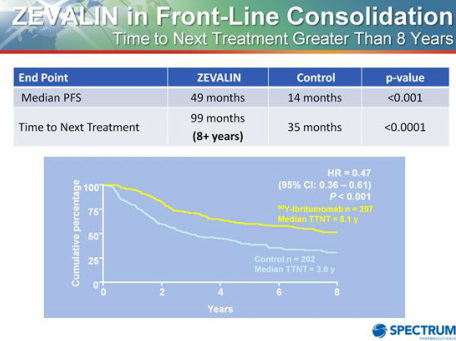
- 6/2011: Several data presentations were made at the International Conference on Malignant Lymphoma - including showing that median OS from FIT trial now exceeds 92 months - Click here for PR with more details.
- 1/2010 hospital reimbursement took effect [reimb rate going up 1/1/2011 to ASP+5%], hope community-based clinics setting change approval comes in 2010 too (11/2010: work on state by state, no barriers right now).
- Bioscan currently is required to assess distribution (so course of therapy takes 6-9 days vs just a 10 min infusion. not req in EU, is in US, FDA wants to gather and compare data 1/11 submitted sNDA to remove (reqd in 3/40 approved nations).
- Concern- hospital orders hot dose and then pt WBC counts too low to admin - who pays?
- 9/2010: down to 40x sales force- won't inc w/o big sales or demand increase.
- 11/2010: exploring possibility of registrational trial in new indication.
- Click here for a link of all trials involving Zevalin listed on ClinicalTrials.gov database.
- 7/2011 Corporate slide deck: near-term events is initiation of Diffuse Large B-cell Lymphoma trial
- 12/2010: positive p2 first line NHL results (60 pts, 1 yr 53% CR, 31% PR- similar to chemo alone or chemo then zev)