Navigate the OncoGenex OGXI research pages
Introduction (Financials, pipeline, milestones, catalysts, general info and links)
Custirsen aka OGX-011 (phase 3 asset for prostate and lung cancer partnered with TEVA, antisense targeting clusterin)
OGX-427 (phase 2 prostate and bladder cancer asset, unpartnered, antisense targeting HSP27)
Early pipeline products (all preclinical and focused on addressing mechanisms of cancer treatment resistance)
Custirsen aka OGX-011 (phase 3 asset for prostate and lung cancer partnered with TEVA, antisense targeting clusterin)
OGX-427 (phase 2 prostate and bladder cancer asset, unpartnered, antisense targeting HSP27)
Early pipeline products (all preclinical and focused on addressing mechanisms of cancer treatment resistance)
OGX-427
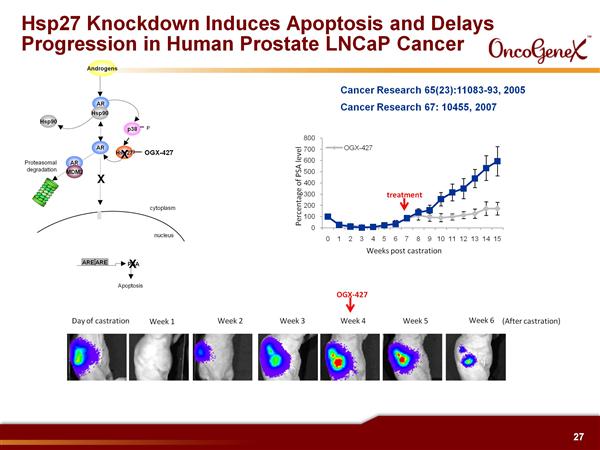
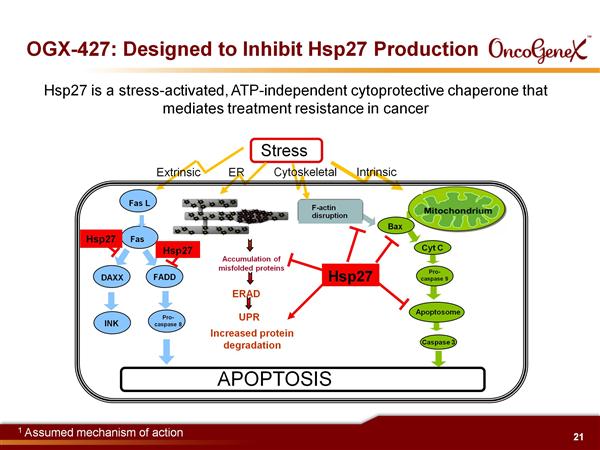
- Isis 2nd generation antisense against Heat Shock Protein 27 (HSP27), a cell survival protein plays a role in the acquisition of cancer treatment resistance to chemotherapy, radiation, and hormonal therapy.
- 1/2005 formed collaboration w/ Isis for certain antisense targets. 4/2005 HSP27 selected as target. License fee was promissary notes that eventually converted into 53k shares of stock, plus milestones (up to $5m between Isis and UBC) and royalties. Isis gets the drug back if OGXI abandons development. Isis is not manufacturing this product. Paid $0.75m milestone in 2010 for initiation of p2. Do not anticipate any milestones in 2011.
- Licensed IP related to HSP27 from U. British Columbia, owe small milestones (C$0.1m paid on p2 initiation) and royalties. Patent expires all jurisdictions 2023, depending on pending applications.
- 4/2011 received EU composition patent (expiration date not listed). Also protected in US, Australia, NZ, India, South Africa. Additional US patents pending
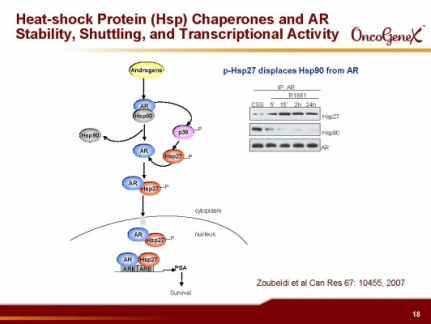
- Reduction of HSP27 facilitates apoptosis by: increasing Bax leading to increased Cytochrome C, increasing IK-B leading to decreased NF-KB, increasing protein aggregation leading to increased ER stress, decreasing IGF-1 and IL-6 signal transduction, increasing FasL mediated cell death, decreasing Androgen receptor activity. Delays tumor progression in preclinical models (10/2011 publication re pancreatic cancer)
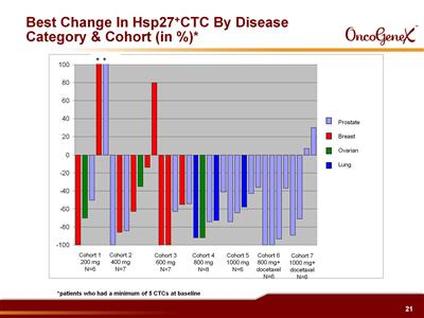
- 6/2010 ASCO final data presented from p1 in solid tumors (breast, prostate, ovarian, NSCLC). Monotherapy: 36 pts total (200-1000 mg doses, up to 10 cycles, median 2 cycles). 3/17 evaluable pts had tumor decrease >20%. CA-125 (click for info) decline in 2/4 evaluable ovarian cancer pts. PSA decline >30% in 3/15 PC pts, reductions in circulating tumor cells (CTCs). >30% serum HSP27 decrease in 25% of pts at 800 and 1000 mg doses. [this doesn't seem that great to me]. Combo w/ docetaxel: 12 pts total (800 and 1000 mg doses, median 6 cycles). 5/10 pts had >20% decrease in measurable disease, PSA reduction >30% in 5/9 pts, reductions in CTCs, persistent >30% serum HSP27 decreases in 35% of pts. MTD was not reached in this trial
- 8/2009 started 2nd, investigator-sponsored p1 in bladder cancer using instillation of drug via catheter prior to tumor excision- so can measure knockdown in excised tissues, 36 pts). Will measure PK/PD for this administration method, plus measure changes in HSP27 levels in bladder cancer cells.
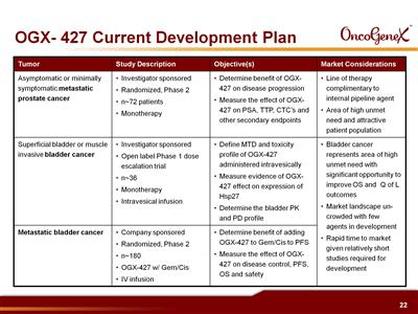
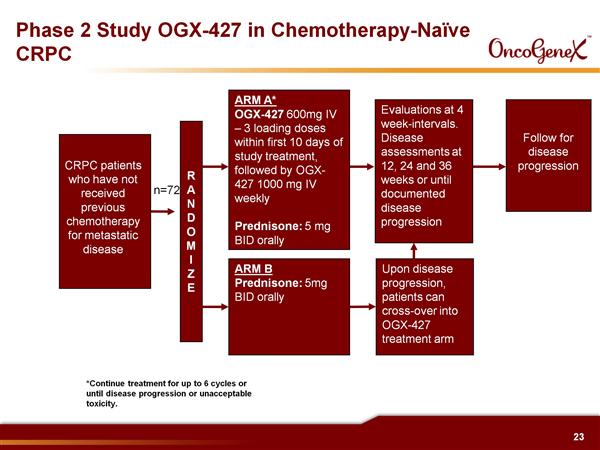
- 9/2010: Initiated randomized, controlled p2 single agent trial in prechemotherapy advanced HRPC (asymptomatic or mildly symptomatic, can have failed experimental MDVN and JNJ drugs, grant rec'd, monotherapy in up to 72 pts, primary endpoint is disease progression at 12 wks, will also measure changes in PSA, CTCs, time to progression).
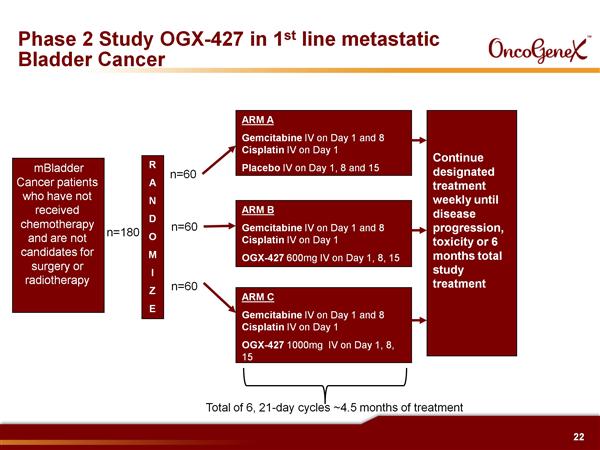
- Plan randomized p2 in 1st-line metastatic bladder cancer to initiate 2h2011 (180 pts, three arms, in combination with first-line chemotherapy, US, Canada, and Europe) - initiated 10/2011 - click for PR
- 8/2010: Partnering discussions have begun- potential in multiple indications would benefit from partner. 2010 10k: assessing alternatives, including partnering, to potentially further expand OGX-427 development.