Tekmira TKMR Stock Research Page
www.tekmirapharm.com
Recent Updates:
8/11/11 2q11 CC - click here for notes.
9/7/11 Stifel Nicolaus webcast - click here for notes.
Click here for archive of all blog posts related to Tekmira.
Recent Updates:
8/11/11 2q11 CC - click here for notes.
9/7/11 Stifel Nicolaus webcast - click here for notes.
Click here for archive of all blog posts related to Tekmira.
General Info
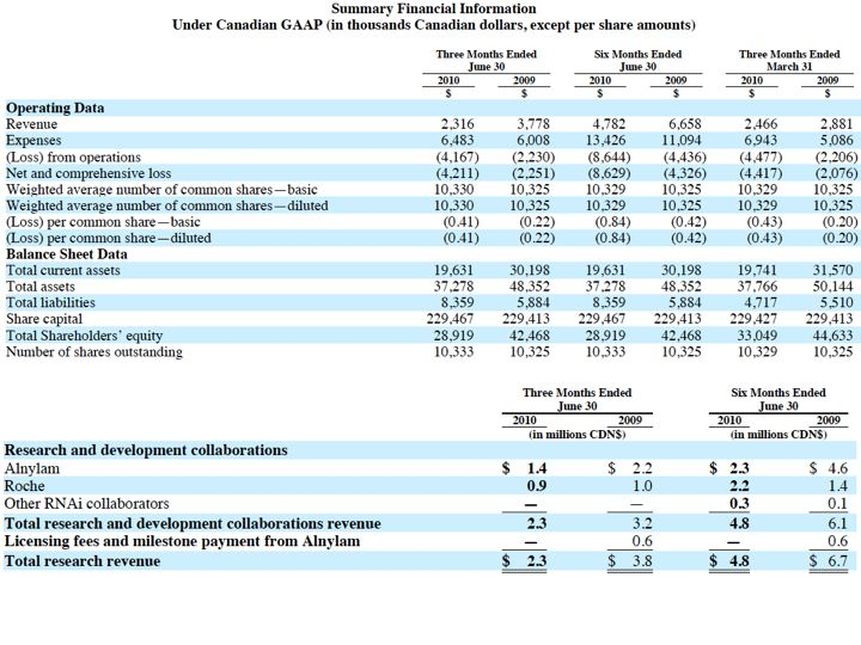
- Formed via the merger on Inex and the former Protiva in April 2007. At this time both Alnylam and Roche invested C$5m at C$12 per share to buy 0.42m shares each.
- Trades on Toronto Canadian Stock Exchange with symbol TKM and on NASDAQ with symbol TKMR
- Report results in Canadian dollars, expenses in US$ and C$, most revenue in US$
- "Our business strategy is to develop our own internal RNA interference (RNAi) therapeutic product candidates and to support our pharmaceutical partners as they advance RNAi product candidates using our lipid nanoparticle delivery technology."
- 11/2010 1:5 reverse stock split
- 11/2010 filed $50m, 25 month shelf
- 6/2011 sold 1.8m shares at C$2.85 plus 0.5m 5 years warrants at C$3.35 - net proceeds C$4.5m
- 2q2011 results: cash on hand plus collaborator and government funding sufficient into 2h2012.
RNAi Delivery Technology
- LNP: liquid nanoparticle
- SNALP: stable nucleic acid lipid particles
- "Our LNP delivery technology allows siRNA to be encapsulated in a particle made of lipids or fats that can be administered intravenously and travel through the blood stream to target organs or sites of disease. The nanoparticles are designed to stay in the circulation for periods of time to allow the particle to efficiently accumulate at sites of disease such as the liver or cancerous tumors. As illustrated in the diagram above, once the nanoparticles have accumulated at the target or tissue site, the cells take up the particle by a process called endocytosis in which the cell’s membrane surrounds the particle. This envelope or endosome pinches off from the cell’s membrane and migrates to the inside of the cell. The lipid nanoparticles undergo an interaction with the endosomal membrane and in the process the siRNA are released inside the cell’s cytoplasm. The released siRNA molecules disperse throughout the cell and engage the RISC complex in the cytoplasm, mediating RNAi."
LNP Manufacturing
- "We are developing scale-up and manufacturing technology, in-process controls, release testing and final product specifications for our products and our partners, products with the aim of ensuring quality, potency and suitable shelf-life, stability and ease of use"
- 11/2010 - recent upgrades at cost of $1.0m. will allow internal manufacturing of clinical trial material by year-end 2010
- 11/2010: Not currently to scale for phase 3 or commercial production
RNAi Competition
- Alnylam
- Big Pharma: Roche, Novartis, Merck, Takeda
- Small biotechs: Quark Pharma, Silence Therapeutics, Calando Pharma, Marina Biotech, RXi Pharma, Opko Health
- Isis Pharma (antisense technology)
Litigation with Alnylam and AlCana
- 3/16/2011: TKMR filed suit against ALNY - click for document
- 4/6/2011: ALNY filed answer and counterclaim - click for document
- 6/3/2011: TKMR filed answer and counterclaim, added AlCana as defendent
- TKMR has stated that based on the venue, the trial could be completed in approximately one year
- 2q2011: signed fixed monthly fee (undisclosed) agreement with Orrick, Herrington and Sutcliffe LLP attorneys, plus they would get additional fee if victorious.
- 10/2011: update provided by TKMR.
TKM-PLK
- RNAi for cancer targeting polo like kinase 1 (PLK1 - involved in tumor cell proliferation)
- Preclinical: selectively kills cancer vs normal cells and blocked gene expression with single dose. Caused tumor regression and increased survival with continued dosing.
- 12/2010: phase 1 study started at 3 US medical centers in up to 52 patients with advanced solid tumors. Secondary endpoints include tumor response and drug PD in tumor biopsy samples
- 6/2011: Cooperative Research and Development Agreement (CRADA) with NCI for second phase 1 study. 9/2011 trial approved - will involve direct infusion into hepatic portal artery of patients with primary liver cancer or liver metastases. TKMR must fund $125k per quarter
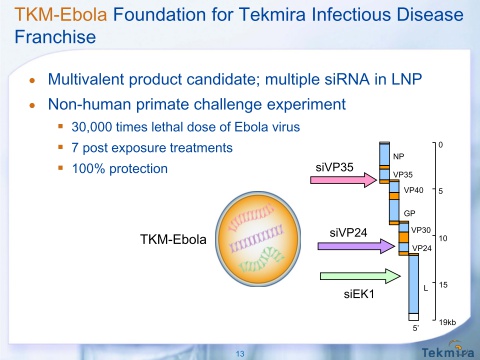
TKM-Ebola
- RNAi targeting 3 portions of Ebola virus genome important in 3 parts of life cycle. Human mortality from outbreaks of Zaire species of Ebola virus (ZEBOV) is ~90% and there are no current treatments.
- 5/2010 published data in The Lancet in non-human primates- received lethal dose of virus and then 7 daily RNAi treatments, 100% survival and free of virus within 14 days.
- 7/2010 contract signed with US government under Transformational Medical Technologies program. First phase of up to $34.7m over 3 years, covering preclinical, IND, and phase 1 trial in healthy volunteers. If program is extended thru FDA approval, TKMR could receive up to $140m. Company bills for direct and third party costs, plus a profit margin.
- Click here for article about government sponsored biodefence programs.
- 10/2010 received $2.4m NIH grant along with U. Texas Medical Branch covering Ebola and Marburg virus research
- 2q11 results: IND expected 2h2011
- "TKM-Ebola will be developed under specific regulatory guidelines to advance therapeutics that cannot meet the requirements for traditional approval because human efficacy studies are not feasible. We believe this could significantly accelerate the approval of TKM-Ebola." ie, after a phase 1 trial in healthy volunteers, pivotal efficacy trials will be conducted in animals
TKM-ApoB
- RNAi against ApoB for high cholesterol
- Same target as Kynamro (mipomersen) from Isis Pharma.
- 7/2009 started and 1/2010 completed phase 1 trial in 23 patients (6 placebo, 17 single ascending doses). "Of the two subjects treated at the highest dose level, one subject experienced an adverse event comprised of flu-like symptoms, cytokine release and transient hypotension consistent with stimulation of the immune system caused by the ApoB siRNA payload. The other subject treated at the highest dose level experienced no side effects. Based on the potential for the immune stimulation to interfere with further dose escalation, we decided to conclude the trial"
- "Based on a review of subsequent non-clinical data for TKM-ApoB, we have decided to delay the initiation of our next TKM-ApoB clinical trial. We had originally planned to initiate a Phase 1-2 clinical trial for TKM-ApoB by the end of 2010. In non-clinical studies, the performance characteristics of the specific lipid nanoparticle formulation used in the current TKM-ApoB product candidate have not met our expectations for the intended application"
- 9/2011: still evaluating new lipid formulations to move this program forward
WEE1/CSN5 program
- Collaboration with NCI identified WEE1 and CSN5 as targets for cancer
- Working on preclinical development of combo RNAi product before IND-enabling toxicology studies
- 6/2011 secured non-exclusive license to the targets from ALNY
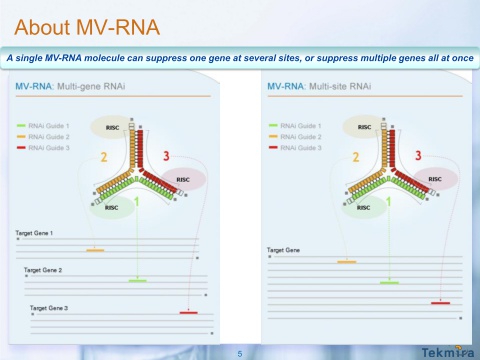
MV-RNA Technology
- 8/2011 licensed rights to multivalent RNA (MV-RNA) platform from Halo-Bio RNA Therapeutics.
- "MV-RNA technology comprises single macromolecules capable of mediating RNAi at multiple unique target sites. MV-RNA can target three sites on a single gene or up to three separate genes simultaneously. Tekmira has successfully demonstrated multi-gene knockdown using MV-RNA enabled by proprietary LNP formulations."
- "Under the license agreement, Tekmira has received exclusive worldwide rights to Halo-Bio's MV-RNA technology for the development of therapeutic products. Tekmira and Halo-Bio will continue an ongoing research collaboration to design and optimize MV-RNA molecules against gene targets of interest. Financial terms of the license agreement were not disclosed."
Bristol-Myers Squibb collaboration
- 5/2010 expanded research collaboration to silence targets of interest. BMS does preclinical work and shares data with TKMR. C$3.0m upfront, BMS receives right of first negotiation for RNAi products against these targets. TKMR will supply a set number of LNP patches over 4 year period (11/2010 - no batches produced so no revenue received yet).
- 5/2011 expanded to include new LNP formulations for delivery to tumors and tissues outside the liver and to support ongoing target validation work
Alnylam agreements
- LNP technology: 1/2007 and amended 5/2008: "exclusive license to certain lipid nanoparticle intellectual property for the discovery, development, and commercialization of RNAi therapeutic products" Received C$9.4m ($8.0m) upfront and eligible for up to $16m milestones and single digit royalties per product ($1.325m and low single digit royalty would flow thru to Univ. British Columbia).
- 8/2007 cross-license. ALNY paid C$3m and received right to opt-in to PLK1 program until the start of phase 2 if they pay for 50% of program costs.
- TKMR has right to select up to 7 targets (4 nonexclusive, 3 exclusive), 2 optioned to date (ApoB, PLK1). Would owe single digit royalties on all targets, and up to $8.5m milestones per nonexclusive target.
- 4/2009: Initiation of ALN-VSP phase 1 trial triggers C$0.6m milestone
- 7/2010: initiation of ALN-TTR01 phase 1 trial triggers $0.5m milestone
- 1/2009 manufacturing agreement: TKMR is exlusive manufacturer of any product using TKMR technology through phase 2 clinical trials. Contractual minimum payments of C$11.2m in 2009-2011.
- 11/2010: ALNY no longer provides research funding to TKMR, so revenue shown in table above is from manufacturing agreement only. C$6.1m revenue in 2008, C$8.8m in 2009.
- ALNY agreed to not acquire more than 10% of TKMR equity before 1/12/2012
Roche agreement
- 5/2008 research agreement, extended 2/2009 and has been completed.
- 5/2009 product development agreement for support thru IND filings. Pays up to C$8.8m per product thru IND filing. Also eligible for up to C$16m milestones and single digit royalties per product (for TKMR IP accessed via ALNY). TKMR will manufacture for preclinical studies and make one batch for phase 1 trial.
- 11/2010: "Currently, there is one systemic RNAi product in development under the agreement. Roche recently provided us with guidance that the IND filing of the product candidate will be delayed and will not be filed before the end of 2010. Under the agreement, Roche may select a second product for development"
- 11/2010: Roche informed TKMR that they are discontinuing research and development in the field of RNAi. Except for finishing some product stability studies, they will not development the product further.
Merck agreement
- Up to C$15m milestones for first product incorporating Tekmira/Protiva IP (C$17m for each additional), plus single digit royalties
- TKMR granted license to some MRK patents as well.
Takeda agreement
- 12/2008 research agreement, extended 1q2010 to provide additional LNP batches
- Also eligible for up to C$16m milestones and single digit royalties per product (for TKMR IP accessed via ALNY).
Pfizer agreement
- "We have a research collaboration agreement with Pfizer whereby Pfizer is evaluating our LNP technology to deliver certain siRNA molecules provided by Pfizer"
Talon Therapeutics agreement
- Formerly Hana Biosciences
- 5/2006 license agreement, they pay all expenses and owe milestones (up to $19m following amendments below) and single digit royalties. Received C$1.7m and 1.12m Hana shares (worth $12m at the time)
- 5/2009 and 10/2010 amended to alter milestone structures and provide a $6m payment used to retire obligation to creditors
- Marqibo (Optisomal Vincristine) - 12/2009 phase 2 complete in relapsed ALL, 7/2011 NDA submitted for accelerated approval. Plan phase 3 in NHL and elderly ALL
- Alocrest (formerly INX-0125, Optisomal Vinorelbine). Phase 1 completed in 2007. Seeking partner for further development.
- Brakiva (formerly INX-0076, Optisomal Topotecan). Phase 1 initiated 11/2008.
Aradigm agreement
- 12/2004 licensing deal for IP, entitled to $4.75m milestones per disease indication (up to 2), plus royalties