Navigate the Alnylam $ALNY Stock Research Pages
- Introduction (valuation, milestones, pipeline, general commentary)
- RNAi Background (science, delivery technologies, patents, litigation)
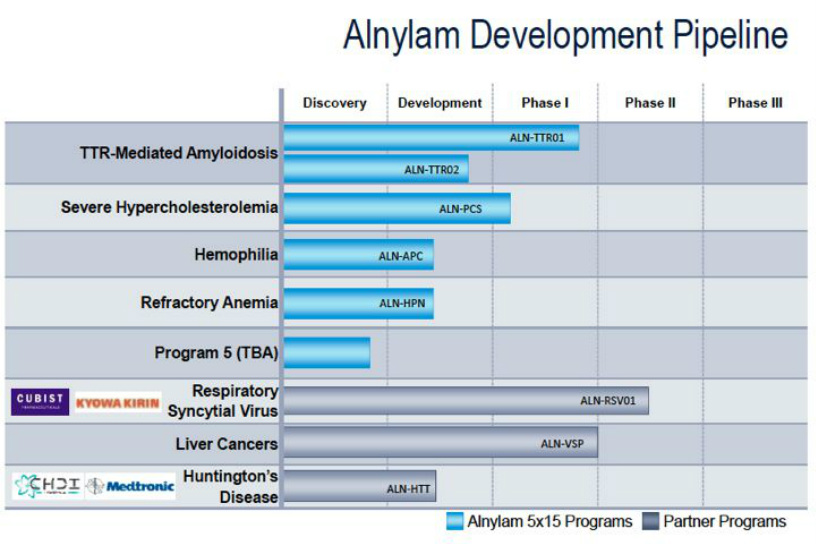
- ALN-TTR (lead 5x15 program for transthyretin amyloidosis)
- 5x15 Programs (details on 5x15 strategy and info on additional preclinical programs)
- Partnered Programs (partnered clinical and preclinical assets falling outside the 5x15 focus area)
- Alliances and Licensing (RNAi platform deals and other licensing agreements with pharma companies)
Alnylam Pharma (ALNY)Website link: www.alnylam.com
History of Updates (blue font indicates premium content for Chimera Research Group subscribers) 5/6/11 1q2011 conference call - click here for notes - click here for slides 5/25/11 Think Equity webcast - click here for notes 6/2011 VSP1 phase 1 data ASCO - click here for PR 8/1/11 2q11 CC - Click here for my notes. 8/24/11 BioPharm International podcast - click for notes 9/8/11 Stifel Nicolaus webcast - Click here for my notes. 9/13/11 Rodman and Renshaw - Click here for my notes. 9/19/11 UBS webcast - click here for my notes. 9/28/11 JMP webcast - click here for my notes. 10/21/11 Biocentury webcast - click here for my notes. 11/1/11 3q11 CC - click for slides - click here for my notes. 11/21/11 ALN-TTR data - CC notes - podcast notes 4/2012 Needham Healthcare Conference - click for notes. 5/2012 ASCO preview and partner updates - click for blog. 5/2012 ASCO presentation - click for blog. 7/2012 JMP webcast - click for notes. 7/2012 ALN-TTR02 data - click for quick commentary. Upcoming events:
|
"We are a biopharmaceutical company developing novel therapeutics based on RNA interference, or RNAi"
|
Links
- RNAi Therapeutics Blog - the best source for news and analysis on the RNAi space, including the ongoing litigation between ALNY and Tekmira $TKMR
- Click here to access all of my blog posts related to ALNY.
- 1/2010: Xconomy interview with Merck re RNAi field.
- 5/2011: Slide deck from Think Equity presentation.
- 6/29/11: ALNY filed an extensive 8k regarding the above litigation - I won't pretend to have consumed and understood this issue at this time. Comments welcome - send me an email.
- 8/2011: click here for BioPharm International podcast with ALNY.
- 8/2011: Click here to download 2q2011 earnings conference call slide deck.
- 11/2011: Click here to download 3q2011 earnings conference call slide deck.
- Click here for a website link that posts all new scientific advanced related to RNAi delivery.
Note re ALNY - most of their slide decks, posters etc, are protected PDF format so I cannot extract images to upload to the Alnylam research pages here at BiotechDueDiligence - so check the individual drug program sections for links to download the complete PDF files.
Management
- John Maraganore, Ph.D. - CEO
- Cynthia Clayton - Senior Director, Investor Relations and Corporate Communications
- Akshay Vaishnam, Md, PhD - Senior VP, Clinical Research
- Michael Mason - VP, Finance and Treasurer
- Barry Greene - President and COO
General Info and Outlook
- Unless otherwise specified, quoted passages are from the ALNY 2010 Annual Report (10k)
- Began operations in 2002
- $150m shelf filed July 2011
- 5/2/11: We are "transforming from platform company to product company"
- 1/31/2011: 172 employees, 142 in R&D. save $25m per yr based on 25% 9/2010 layoffs (caused by conclusion of NVS collaboration (see details below)
- 2011 Guidance: ye2011 cash balance >$275m, excluding any new significant partnerships. "Multiyear runway" (5/2/11)
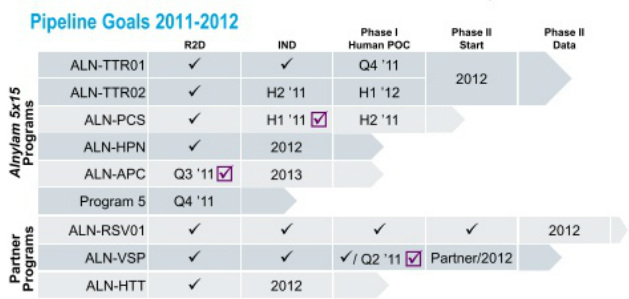
- 5/25/11 webcast: by this time in 2012 will have 5 programs in the clinic
- 5/25/11 webcast: expect to form additional partnerships in 2011
- “5x15 strategy” - focus on genetically defined diseases, goal is to have 5 programs in late stage trials by the end of 2015. Intend to commercialize these drugs alone in the US and perhaps other countries, seek ROW partners. The first 3 of these programs have been identified as ALN-TTR01, ALN-PCS, and ALN-HPN (see details on each program below). The fourth and fifth drug candidates will be selected and announced from current internal programs by the end of 2011 (reiterated 5/2/11). Each of these programs would share the following attributes:
2. The potential to have a significant impact in high unmet need patient populations
3. The ability to leverage our existing RNAi delivery platform
4. The opportunity to monitor an early biomarker in Phase I clinical trials for human proof of concept
5. The existence of clinically relevant endpoints for the filing of NDA with a focused patient database and possible accelerated paths for commercialization”
- “Our collaboration strategy is to form (1) non-exclusive platform and/or multi-target discovery alliances where our collaborators [examples: Roche, Novartis, Takeda] obtain access to our capabilities and intellectual property to develop their own RNAi therapeutic products; and (2) worldwide or specific geographic partnerships on select RNAi therapeutic programs [examples: Cubist, Medtronic].”
- Other “partner-based programs” are also advancing as described below, as well as undisclosed early stage internal programs and a research alliance with Takeda (see below)
- R&D expenses $106.4m in 2010, $108.7m in 2009, and $96.9m in 2008...2010 10k: expect losses over next several years at least"
- 5/2/11: R&D expense $26m in 1q2011, will decrease slightly the rest of the year
- Manufacturing: "Under our agreements with Tekmira, we are obligated to utilize Tekmira for the manufacture of all LNP-formulated product candidates covered by Tekmira's intellectual property beginning during pre-clinical development and continuing through Phase II clinical trials. During 2009, we and Tekmira entered into a manufacturing and supply agreement under which we are committed to pay Tekmira a minimum of CAD$11.2 million (representing U.S.$9.2 million at the time of execution) through December 2011 for manufacturing services. Tekmira is currently manufacturing the clinical drug supply for our Phase I clinical trials of ALN-VSP and ALN-TTR01"
Competition in RNAi Space
- "Chemically synthesized siRNA drugs": Merck (they previous had an ALNY license, but acquired Sirna 12/2006 and terminated ALNY 9/2007), Novartis, Takeda, Kyowa Hakko Kirin, Marina Biotech, Calando, Quark, Silence Therapeutics, RXi Pharma, Tekmira, Sylentis, Dicerna Pharma, ZaBeCor. Many of these companies have licensed ALNY IP.
- Gene therapy in which "exogenously-introduced genes produce siRNA-like molecules within cells": Benitec
- MicroRNA therapeutics: Rosetta Genomics, Santaris Pharma, miRagen Therapeutics, Mirna Therapeutics, Asuragen
- Antisense therapeutics: "Antisense technology uses short, single-stranded, DNA-like molecules to block mRNAs encoding specific proteins. An antisense oligonucleotide, or ASO, contains a sequence of bases complementary to a sequence within its target mRNA, enabling it to attach to the mRNA by base-pairing. The attachment of the ASO may lead to breakdown of the mRNA, or may physically block the mRNA from associating with the protein synthesis machinery of the cell. In either case, production of the protein encoded by the mRNA may be reduced. Typically, the backbone of an ASO, the linkages that hold its constituent bases together, will carry a number of chemical modifications that do not exist in naturally occurring DNA. These modifications are intended to improve the stability and pharmaceutical properties of the ASO. While we believe that RNAi drugs may potentially have significant advantages over ASOs, including greater potency and specificity, others are developing ASO drugs that are currently at a more advanced stage of development than RNAi drugs. For example, Isis has developed an ASO drug, Vitravene®, which is currently on the market, and has several ASO product candidates in clinical trials, including mipomersen, which is a lipid-lowering drug being developed by Isis in collaboration with Genzyme Corporation, or Genzyme. In addition, a number of other companies have product candidates in various stages of pre-clinical and clinical development. Included in these companies are Santaris, Genta Incorporated and AVI BioPharma, Inc. Because of their later stage of development, ASOs, rather than siRNAs, may become the preferred technology for drugs that target mRNAs in order to turn off the activity of specific genes."
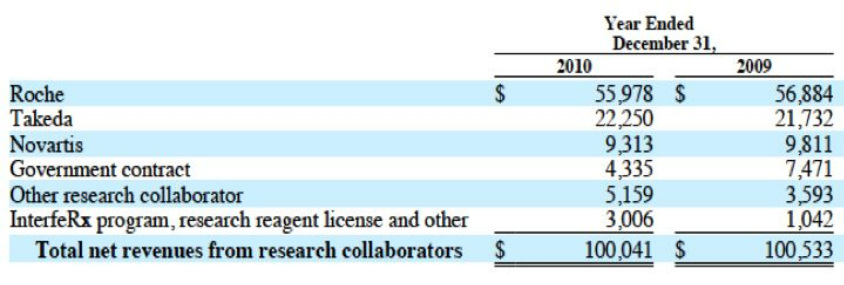
Note: NVS and government revenue will cease after 2010, Roche and Takeda will remain steady
"Other includes Regulus accounting flowthru, Cubist deal
1q2011 revenue: $14.0m from Roche, $5.8m from Takeda, $1.1m other (via 3/31/11 10q)
"Other includes Regulus accounting flowthru, Cubist deal
1q2011 revenue: $14.0m from Roche, $5.8m from Takeda, $1.1m other (via 3/31/11 10q)