Navigate the Isis Pharma ISIS Research Pages
- Introduction (valuation, financials, outlook, company catalysts)
- Antisense background (science, technology, intellectual property, manufacturing)
- KYNAMRO - mipomersen (lead program in regulatory review for familial hypercholesterolemia)
- Mipomersen Liver Safety (special feature pages with extensive notes on long-term safety and efficacy of the drug)
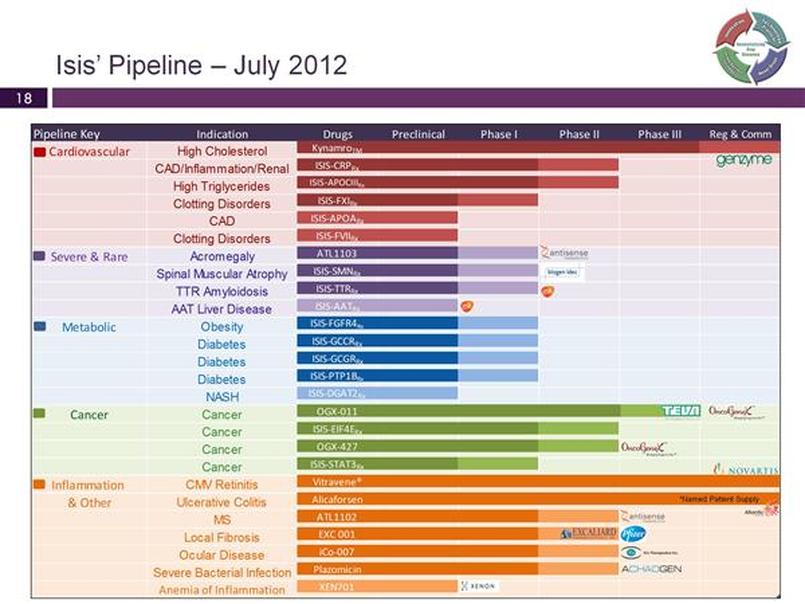
- Cardiovascuar Disease pipeline (addtional CV drugs besides mipomersen)
- Metabolic Disease pipeline (several drugs for type II diabetes, as well as obesity)
- Cancer pipeline (including partnered with Eli Lilly $LLY and OncoGenex $OGXI)
- Severe, Rare, and Neurodegenerative Disease pipeline (incude GSK collaboration)
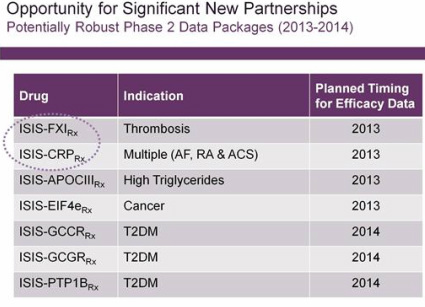
- Licensing deals and Satellite/spin-off companies (key business development activities)
- Discontinued Programs (not everything works out when you have a large and broad biotech portfolio)
Isis Pharma (ISIS)Website link: www.isispharm.com
Recent Updates: Blue font indicates premium content for Chimera Research Group subscribers Archive of webcast notes for 2010 through April 2011. Roth Conference webcast 3/15/11 (click here for notes) 1q2011 earnings 5/5/11 (link to notes and press release) 6/9/11 Jefferies webcast (click here for notes) 6/16/11 Annual meeting (no webcast notes - see Past Events page for slides) 8/4/11 2q2011 earnings (click here for notes) 9/8/11 Stifel Nicoluas (click here for notes) 9/12/11 CC on Longterm Mipomersen Safety and Efficacy (click here for notes) 9/19/11 UBS (click here for notes) 2/2012 Leerink Swann conference (click for notes) 3/2012 Citi Healthcare conference (click for notes) 4/2012 Roth Capital Growth conference (click for notes) 4/2012 Needham Healthcare conference (click for notes) 5/2012 Competitor Aegerion Pharma $AEGR 1q12 earnings (click for notes) 5/2012 ASCO preview and partner updates (click for blog) 7/2012 JMP webcast (click for notes) Upcoming events:
|
The leader in antisense technology, drug discovery and development
|
Links and Resources
- Click here for the Isis archive of old pipeline figures, slides etc that I have removed from the main ISIS page (useful to track company progress against management projections)
- Click here to view all of my blog posts related to Isis
- Click here for July 2011 Nature Biotechnology piece penned by ISIS CEO Stan Crooke.
- Click here to download September 2011 UBS conference slide deck.
- Click here to download January 2012 Research and Development Day slide deck (highly recommend).
General Info and Outlook
- "Our business strategy is to do what we do best—to discover unique antisense drugs and develop these drugs to key clinical value inflection points. In this way, our organization remains small and focused. We discover and conduct early development of new drugs, outlicense our drugs to partners and build a broad base of license fees, milestone payments and royalty income. We maximize the value of the drugs we discover by putting them in the hands of quality partners with late-stage development and commercialization expertise." (2010 10k)
- Each drug: "Why does target represent an attractive approach to treat patients with the diseases we are thinking about? Why do we think the target is particularly attractive for Antisense? What do we think the commercial potential of the drug is? What do we need to see as the drug moves forward in development to believe that we have a winner? And finally, how does each drug fit into our portfolio in a fashion that contributes to the overall value of Isis?
- 11/2010: Gen 2.5: 10x potency vs Gen 2.0, allows oral dosing-first for cancer and rare diseases, IND of a gen 2.5 candidate perhaps 18 months.
- As of 2/14/2011, have 270 employees plus 50 for manufacturing and 50 at Regulus
- 4/2010: discovery resources 25% each to Cardiovascular (CV), Metabolic, Cancer, Other
- See below for description of candidates and their status below, grouped by disease area, and followed by projects being run by "satellite" companies which license ISIS technology. Isis does not pay for these drugs, but receives milestones/royalties and in some cases equity stakes in the company and manufacturing income.