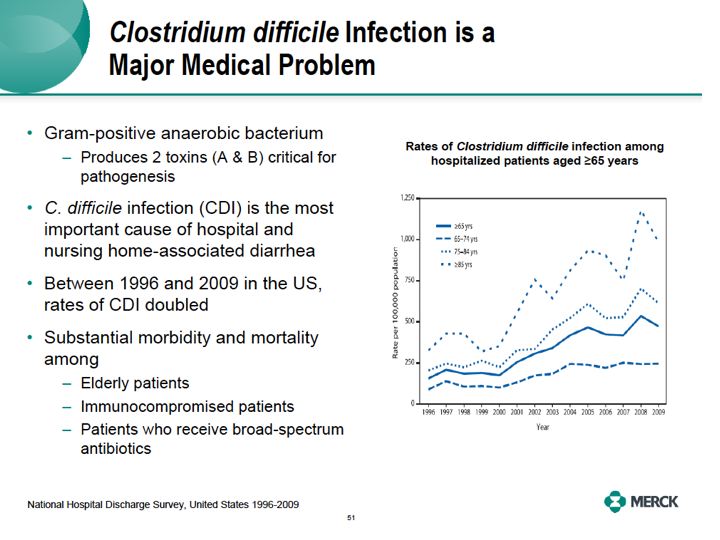
Let me start by highlighting our antimicrobial group of therapeutics, focusing on our first-in-class MK3415A monoclonal antibodies to Clostridium difficile. Clostridium difficile is a gram-positive rod anaerobic bacteria. It produces two toxins, nicely named toxin A and toxin B, critical for the pathogenicity of the bacteria in the human colon. C. difficile infection or CDI has now become the most important cause of hospital and nursing home associated diarrhea in the United States.There are now estimated to be over 0.5 million new cases of CDI in the US each year. The rates of disease have doubled in the past 10 years, especially in the elderly.
At the same time, a hyper virulent strain of this bug is now in hospitals in the US and Europe, leading to further increases in mortality and morbidity.The infection is more likely to cause substantial morbidity and mortality in some of our sickest patients, our elderly immunocompromised patients or patients who received large amounts of broad spectrum antibiotics.
What also makes clostridium difficile so difficult to treat for physicians and patients is that it frequently recurs despite therapy. Following an initial bout of CDI, a full 20% of people will recur. With further rounds of recurrence the likelihood of permanent clearance drops precipitously so that a recurrence after three bouts of CDI is highly likely. No medicines are available to prevent
this recurrence. So we do have a true unmet medical need.
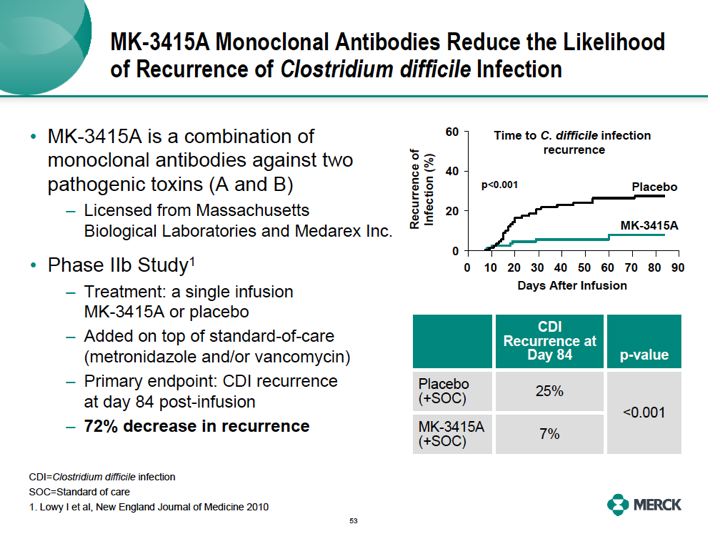
We think we have an answer for this though. MK3415A monoclonal antibody. It's a product that we licensed from MassBio and Medarex. MK3415A is a combination of two monoclonal antibodies, one against toxin A and one against toxin B, used to treat patients with a single infusion. In the Phase IIB trial, published in the New England Journal of Medicine, treatment with MK3415A when added to the standard of care with metronidazole and or Vancomycin reduced the likelihood of the recurrence of CDI by 72% through 12 weeks following the end of the one infusion. So, administration of MK3415A to patients at high risk of recurrence may greatly improve their chances of preventing ongoing disease and ongoing diarrhea.
The Phase III program for this drug has already commenced, representing the first time a biologic has been used in the treatment of this type of illness. Two adaptive design pivotal trials will enroll 2,800 patients who were hospitalized with C diff and will determine the antibody's safety and efficacy in preventing recurrence of C difficile infection.There will be four treatment arms,
the monoclonal against toxin A alone, toxin B alone, A plus B versus placebo. All with adjunctive standard of care. Therapy with metronidazole, vancomycin or fidaxomicin, recently approved for this disease will be allowed in the trials and the primary end point is prevention of disease recurrence. Let me remind you that there is no drug approved for prevention of CDI
recurrence. We expect filings in the US and Europe in 2014.


 RSS Feed
RSS Feed
