"Now Europe first introduced a biosimilar pathway in 2005 for three molecules including human growth hormone, erythropoietin and the colony stimulating factors, granulocyte colony stimulating factors.These three were among the earliest biotech products and they're the simplest in size and structure. In the EU the approval packages using this new pathway required analytical, preclinical, clinical and immunogenicity studies and draft guidelines for the regulatory approval of the important monoclonal antibody biosimilars in Europe are currently in discussion and we're waiting for those guidelines to be formally approved and issued. In the United States, the biologics price competition and innovation act of 2010 created a legal pathway for approval of biosimilars, but it did not detail the regulatory requirements for the development and licensure steps. So we're also waiting for these guidelines to be issued, hopefully before the end of this year"
"Now, while waiting for this forma guidance, we continue to discuss our specific programs with the regulators. We've had numerous meetings to date, we've gotten some feedback from them, we extrapolate from those learning's to all of our development programs. So we desperately need the overview, both from the US and Europe to understand what the full requirements for this business are going to be"
We've committed over 200 people in R&D to the development of the Merck bioventures portfolio.We've established partnerships to obtain critical molecules, such as the deal already mentioned with Hanwha to obtain a biosimilar version of Enbrel. We've invested over $200 million in building our internal manufacturing infrastructure for biosimilar and novel programs.
Internally we have six facilities that we're making our biological products in. And then, recently, we concluded a partnership with a MedImmune -- with MedImmune for their manufacturing capacity in their brand new facility in Frederick Maryland. Finally, for clinical development we've partnered with Parexel in an exclusive biosimilars deal. They're working to provide us
with access to approximately 500 worldwide clinical sites to do our biosimilar clinical work."
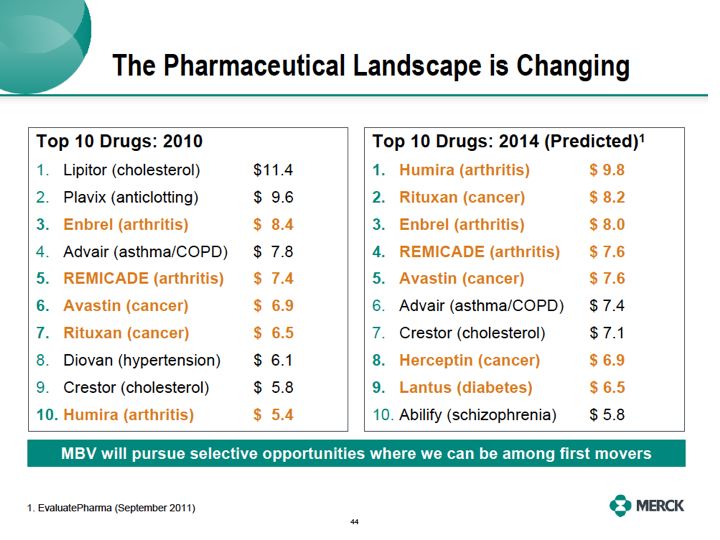
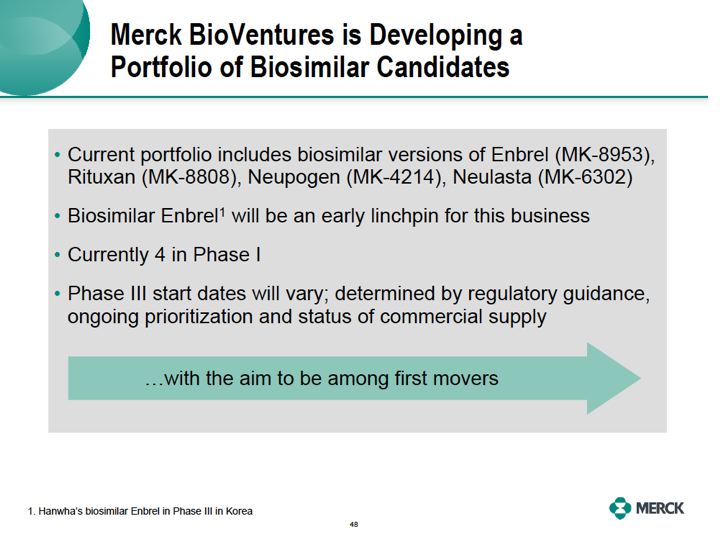
"Now, because Enbrel is one of the earliest freedom to operate dates for Merck we believe it's going to be the lynchpin in this new business. This portfolio also allows us to continually prioritize our investments behind the best opportunities. Using the strategies outlined earlier by Peter, we'll select from these molecules that have the highest potential relative to the development
costs, highest probability of technical success and those molecules for which we have the highest probability of being a first mover."
"Now, while we won't wait for the formal global guidance on biosimilars to be issued, before moving to late stage development, we do need formal clarity around a number of crucial issues. For example, the apparent requirement for using only locally sourced comparator material for our Phase III studies that are intended for global registration.That's a challenge. In this context
of regulatory ambiguity, we don't expect to have five molecules in Phase III by the end of 2012.
Instead, the timing of Phase III starts will be determined by the regulatory guidance and the ongoing prioritization of our portfolio.That said, I'll remind you that it's the freedom to operate dates that are most critical in the timing of the launch of our products."
"Finally, biosimilar -- the market opportunity for biosimilars is large, with important patent expiry starting in the middle of this decade. Merck BioVentures has made progress in developing a portfolio of biosimilar targets, securing partnerships to enhance the portfolio and capabilities while actively engaging with regulators as they develop the final pathways. All of this is with a
goal of pursuing the highest potential molecules and making them ready to enter the market when we have the freedom to operate."

 RSS Feed
RSS Feed
