BioSante Pharma BPAX Stock Research Index |
Cancer Vaccines (formerly known as GVAX platform)
- Oncology immunotherapy platform acquired in Cell Genesys $CEGE merger . Goal: stimulate the patient's immune system to fight against the cancer. Given via injection in an outpatient setting
- Licensed from Johns Hopkins and Whitehead Institute. 17 patent families including over 60 patents expiring between 2012 and 2026- would owe milestones and royalty payments
- 2010 10k: "Our cancer vaccines are comprised of tumor cells that are genetically modified to secrete an immune-stimulating cytokine known as granulocyte-macrophage colony-stimulating factor, or GM-CSF, and are then irradiated for safety. Since our cancer vaccines consist of whole tumor cells, the cancer patient’s immune system can be activated against multiple tumor cell components, or antigens, potentially resulting in greater clinical benefit than if the vaccine consisted of only a single tumor cell component. Additionally, the secretion of GM-CSF by the modified tumor cells can enhance greatly the immune response by recruiting and activating dendritic cells at the injection site, a critical step in the optimal response by the immune system to any immunotherapy product. The antitumor immune response which occurs throughout the body following administration of our cancer vaccine potentially can result in the destruction of tumor cells that persist or recur following surgery, radiation therapy or chemotherapy treatment."
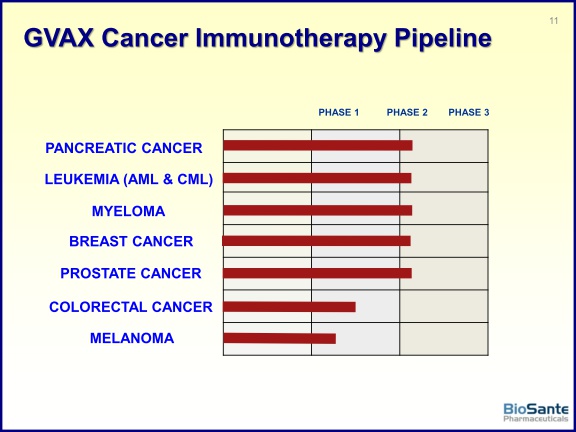
- The program has received orphan drug designations in four indications: pancreatic cancer, AML, CML, and most recently melanoma (click here for 1/25/11 press release)...designation for more indications may be pending. Orphan drugs get 7 years market exclusivity, waived user fees, 50% R&D tax credit and do not have to be discounted for government purchases
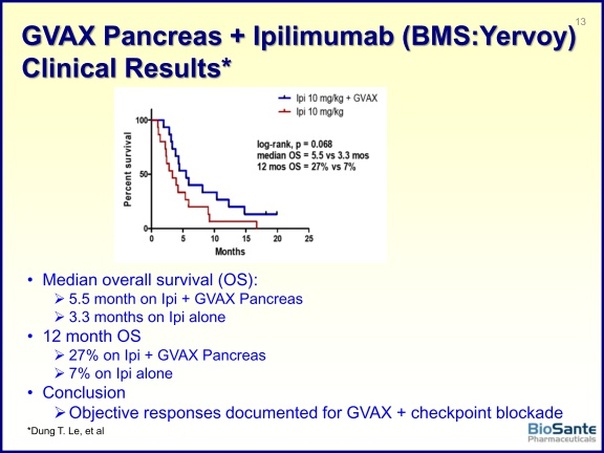
- 3/3/2011: announced publication of phase 2 data from pancreatic cancer vaccine trial in 60 pts at Johns Hopkins. Median disease free survival was 17.3 months, median overall survival was 24.8 months. This compares favorably to historical data of 15-20 months survival- but the press release was highly inappropriate in the manner in which it directly compared the data. Had originally guided this data by ye2010 and then 2/14/11 webcast said 1q2011. Click here for my blog post about this data release.
- 6/2011: presented breast cancer vaccine data at ASCO: 20 pts with metastatic, HER2+ breast cancer; achieved 50% clinical benefit rate at 6 months. Click here for more details. (note there was no company PR about this presentation)
- 3/15/11 webcast: pharma industry is currently more risk averse in partnering than ever, want to see more data. No timeline for a deal, could be a single indication or the whole portfolio (3/2010 had said think could monetize in next 12 months)
- 4/15/11webcast: 15 p1 or p2 cancer vaccine trials ongoing paid by JHU or NCI or foundations...other initiations this year. Can't say when will release more data. Goal is to monetize, hope to be able announce something very soon (this is a more optimistic statement compared to recent presentations)
- Ongoing indications include CML, AML, breast cancer, multiple myeloma, pancreatic cancer. Melanoma and prostate cancer trials will begin in 2011 (according to 5/9/11 webcast- prostate re-initiation timeline has slipped from 4q2010 and 1q2011...)
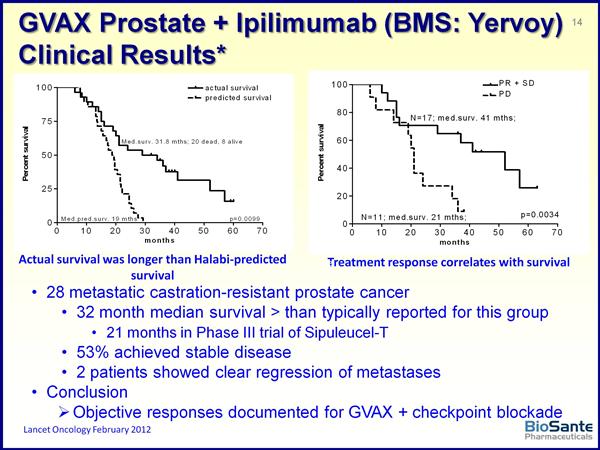
- 6/2011: announced that FDA has lifted clinical hold on GVAX prostate program (Click here for PR). Planning for new p2 trial is underway. BPAX paid for drug manufacturing, study will be funded by Johns Hopkins, Prostate Cancer Fndn, and OneinSix Fndn.
- 4/18/2011: announced license (note- no upfront payment, only undisclosed milestones/royalties) to Aduro BioTech to use pancreatic and prostate cancer vaccines in combination w/ Aduro's Listeria-based vaccine platform, specifically CRS-207 which expresses tumor-associated antigen mesothelin. Plan for 4q2011 pancreatic cancer sequential vaccination combo p2 trial in 90 pts (synergy has been demonstrated in mouse models) - click here for press release