Be sure to check out my latest reports about BPAX:
BioSante Pharma BPAX Stock Research Index |
Libigel- introduction
- 1% testosterone transdermal gel for hypoactive sexual desire disorder (HSDD)- delivers 300 ug daily dose- increases testosterone to the normal range for estrogen-treated post-menopausal women. Click here for a PDF file with a nice overview of Libigel and its path to FDA approval
- Licensed from Antares Pharma $AIS (click here for research page) for the following countries: U.S., Canada, New Zealand, South Africa, Israel, Mexico, China (including Hong Kong) and Indonesia. Owe regulatory and development milestones payments, 4.5% royalties, and 25% of sublicensing income to AIS. AIS also owns manufacturing rights and has the right to use BPAX's data in other territories (ie, the EU). Click here to read the entire redacted license agreement
- 2010 10K (first time I have seen this precise language): "We have three SPAs in place concerning LibiGel. The first SPA [received January 2008] agreement covers the pivotal Phase III safety and efficacy trials of LibiGel in the treatment of FSD for “surgically” menopausal women. The second SPA covers our LibiGel program in the treatment of FSD in “naturally” menopausal women (received July 2008, to my knowledge no clinical trials have been run yet for this indication). The third SPA agreement covers the LibiGel stability, or shelf life, studies for the intended commercialization of LibiGel product."
- 5/9/11 webcast: ovaries produce 50% of woman's testosterone (hence efficacy trials are in surgically menopausal women)
- 5/9/11 webcast: Two papers published (JAMA and NEJM) showed 43% rate of FSD/low desire in female population (higher rate than males)
- 4/15/11 webcast: targeting menopausal population because these women have ~50% decreased testosterone levels vs pre-menopausal
- BPAX fully expects to receive priority review because there are no approved products for this indication in US
- Libigel is v low dose (300 micrograms per day) so no acne or hair side effects [ie, safety concerns are representative of post vioxx fda change- not specific to this product].
- 30% of women that complain about low desire/sevual dysfunction to their OB/GYN are currently treated with off label products (5/9/11 webcast)
- 11/2010, 2/2011, 5/2011: based on 2009 IMS data and primary research, now say >4m off-label prescriptions per year are filled in the US. Previously stated 2m off label rx/yr currently plus 1m more at formulating pharmacy- but testosterone doses that are too low or too high lose effect.
- 4/15/11 webcast: think female sexual dysfunction market is bigger than the $2b male ED because 43% women have sexual dys vs 31% for male, according to studies.
- 3/15/11 and 5/24/11 webcasts: Don't believe that current off-label testosterone rx for women are being reimbursed, cost about $50-80 per month- BPAX believes that Libigel co-pay would be less than that ($30-50/month), should get same insurance coverage as male ED drugs. Libigel will cost $8-10 per day, less than the $11-12 per day of male testosterone products. Note that a couple years ago BPAX did not think Libigel would get reimbursement and expect out-of-pocket marketing and price of <$5 per day
- 11/3/2010 webcast- US has 14000 target, high-prescribing gynecologists
Libigel - efficacy trials
- 2 randomized, double-blind, placebo controlled (1:1) phase 3 trials, each with up to 500 pts, 300 ug per day, 60 day lead-in then 6 months on treatment, 30-40 sites in US and Canada
- Patient population: Age 30-65 years, surgically menopausal women (have undergone hysterectomy and bilateral salpingo-oophorectomy prior to natural menopause), on stable estrogen dose, in >1 yr stable monogamous relationship and sexually active
- Click here for BLOOM-1 first trial ClinicalTrials.gov listing. Click here for BLOOM-2 second trial ClinicalTrials.gov listing.
- Elevated SBHG (>160 nmol/L) levels is a key exclusion crieria (usually due to estrogen therapy). Also is an upper limit on BMI/obesity allowed (5/24/11 webcast)
- Efficacy SPA w/ FDA covers: study design, efficacy endpoints, sample size, study conduct, statistical analysis plan.
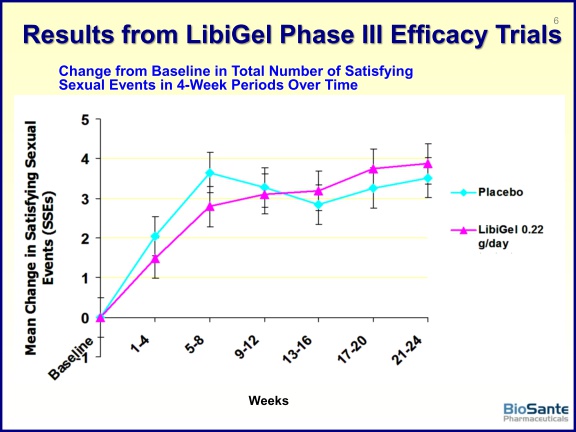
- Co-Primary endpoints are "The change in the 4-week total number of protocol defined satisfying sexual events [see next bullet] from the Baseline period to Weeks 21-24 as recorded by the subject in her diary [do this daily, using FDA-validated instrument], the Inventory of Sexual Events and Desire (ISED), a validated instrument for HSDD" and "The change from Baseline to Weeks 21-24 in the mean ISED desire score"...Secondary efficacy endpoint: "Change from Baseline in the mean Female Sexual Distress Scale-Revised (FSDS‑R) Question 13 score"
- p3 trials powered for increase of 1 satisfying sexual event (SSE= intercourse, giving or receiving oral sex, masturbation)- this change was considered to be clinically meaningful by the 2004 FDA advisory committee for Intrinsa
- expect much higher increase than that based on double-blind, placebo-controlled p2 data. Libigel produced a blood testosterone level in the normal range for premenopausal women.
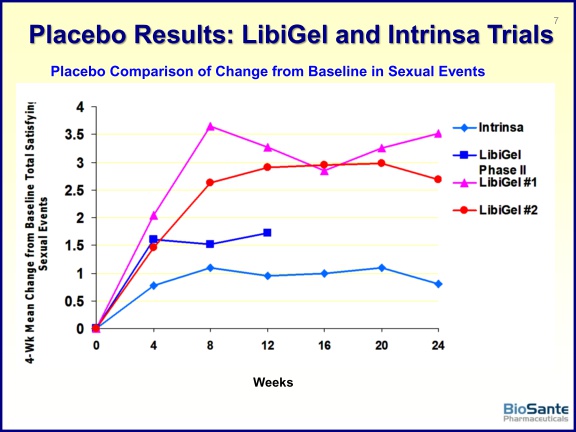
- Phase 2 data: saw 238% increase in number of satisfying sexual events from baseline vs placebo. Average of 2.5 SSE during baseline 4 wks, at month 3 this increased to 7.5 SSE on Libigel arm vs increase to 4.1 SSE on placebo. Note this trial had 3 month treatment vs 6 month in ongoing phase 3 trials. (click here for 2004 ISSWSH abstract - Click here for company press release)
- Give patients a bottle with 3 month supply, can weigh to monitor compliance.
- 2/2011: presented market research data based on survey of >100 OB/GYNs: >90% currently write off-label testosterone prescriptions for HSDD- are unsatisfied with this option but get an average of 37 unsolicited inquiries a month regarding sexual dysfunction. 94% viewed Libigel product favorably, contingent on safety data
- 2/24/2011 announced completion of enrollment in first efficacy trial, have over 4000 women enrolled between the three p3 studies, second efficacy trial will complete enrollment very soon
- 3/30/2011: announced completion of enrollment in 2nd efficacy trial- top-line data will be available fall 2011 - click here for press release
- 7/2010 started trial in new indication to measure effect of Libigel on congnition in menopausal women (120 pts, RDBPC, 6 month tx in australia).
- 10/2010: started continuation study to see if beenfit persists 4 wks after stopping libigel- 750 pts, est complete 9/2011.
Libigel - Safety trial
- Safety study: Covered by SPA described above, total of 2500-4000 pts for average exposure of 12 months (300 ug per day) prior to NDA submission and then continue to follow with placebo-controlled 48 month extension (5 years total exposure- count CV events and breast cancer incidence regardless of whether patients are still taking the drug). 4/15/11 webcast: also measure BP and cholesterol levels among others
- Click here for ClincalTrials.gov listing.
- Goal: "Demonstrate that the ratio of the rate of CV events on LibiGel 300 mcg/day compared to placebo has an upper bound of the 96% confidence interval ≤ 2"..primary endpoint is a composite outcome measure consisting of the total number of: CV death, nonfatal stroke, nonfatal myocardial infarction, hospitalized unstable angina, coronary revascularization, and DVTs.
- CEO comments (Rodman and Renshaw fireside chat) described as event rate noninferiority w/in 25-30% of placebo, 1/3 surgical, 2/3 naturally menopausal
- Patient criteria (higher risk than general target population): 50-85 yrs old, 2+ CV risk factors (based on RUTH study criteria)
- 2/14/11 webcast: 20% smokers, 20% diabetic, 65% high BP and cholesterol, 4-5% prior CV event history. 5/24/11 webcast: no limit on obesity (ie, BMI)
- Trial was initiated 1/2008, halted enrollment to save money 4/2009, resumed 1/2010.
- Study design: "At 2,500 women, the DMC statistician will perform a sample size re-estimation of unblinded data (2% burn of Type I error). If the chance for success is > 90%, the study will stop enrolling and the study continued one year prior to the primary"...see below for results of this interim analysis in October 2010- this threshold was not met, therefore enrollment continues and samples size re-estimation will occur after each new CV event
- "After a minimum average of one year of exposure to study drug, initial data analysis will occur and an NDA will be submitted to obtain marketing approval"...note that this average exposure has been achieved, but the enrollment has to continue in order to gain confidence that the statistical endpoint will be achieved
- 11/3/2010 webcast- expected CV event rate was 2% and expected breast cancer rate was 0.35% (2/14/11 webcast- this predication came from the July 2002 NIH Women's Health Initiative aka "WHI" data)...4/15/11 webcast expected breast cancer rate stated as 0.3-0.31%
- 11/3/2010 webcast updates: first woman recently reached 1000 days on study; this trial will represent the largest ever testosterone safety database; 5th unblinded safety review will occur 1q2011
- Safety Data Safety Monitoring Committee Updates-
---8/2009: passed blinded safety look (>1250 pts, 825 ptyrs, >900 over 1 yr, >100 over 2 yrs, 7 CV events, no deaths).
---10/2009: first UNblinded safety look (1055 pts, 883 pt years, 1 MI, 3 BC, no deaths).
---2/2010: second UNblinded safety look (>1200 pt years, 6 CV events, 4 BC).
---6/2010: third UNblinded safety look (1500 pt years, 7 CV events, 4 BC). 9/10: >2000 enrolled (>1000 over 1 yr).
---10/2010: safety presentation (2300 pts, avg 10.5 months of drug, 14 events 0.65%, 7 BC 0.32%- these rates are 25-30% of what was expected).
---10/18/10: hit 2500 enrollment, triggered 4th unblinded safety evaluation and first unblinded CV statistical evaluation, will continue to enroll and evaluate again after each event (2300 pt yrs, avg 10.7m on drug, 850 >1yr, 400>2 yr, 14 CV events, 7 BC).
---2/12/2011: 5th unblinded safety review announced: 2869 pts, >3000 women-years on drug, 17 CV events (0.57% rate), 8 breast cancers (0.27% rate)-- cannot yet terminated enrollment, continue study as per protocol, average enrollment now >12.5 months, >1000 on trial over 1 yr, >550 on trial > 2 years. - click here for press release - click here for my blog post about this event.
---6/2011: 6th UNblinded safety look (3307 pts, >3450 women-yrs, 22 CV events: 0.58% rate, 9 breast cancers (0.24% rate). Average exposure now 12.8 months, >1500 on trial over 1 yr, >700 over two years. By the time DSMB met and enrollment halted (see below), 250 additional women had been enrolled bring total to >3550 women and >3800 pt-years exposure.
- Recent enrollment status updates:
---3/15/2011 webcast: enrolled ~3000 women and have ~3200 pt-years of exposure. 3/30/2011 press release: over 3000 pts enrolled.
---4/15/11 webcast: >3000 women, >3300 women-years, >1000 on drug at least one year, >600 for over 2 yrs, handful for >3 yrs; avg exposure 13-14 months now
---5/24/11 webcast: 3500 enrolled, >3600 women-years, >1400 on drug over 1 yr, 700-800 over 2 yrs, 40 >3 yrs
---6/6//11 Announced completion of enrollment with a total of >3550 pts (Click here for PR). "The FDA-agreed sample size analysis indicates that enrollment should stop, based on meeting a minimum 90 percent predictive probability of success of the safety study to show the safety of LibiGel at the primary data analysis... one or both of the following criteria one year after enrollment is completed: the ratio of the rate of CV events on LibiGel compared to placebo has an upper bound of the 97.2% confidence interval ≤ 2, or the 97.2% confidence interval for the risk difference is ≤ 1% (and less than twice the observed CV events in the LibiGel group vs. placebo group)."
- Last pt was enrolled early June 2011 (would have reached maximum 4000 pts in July or August 2011), so study analysis will be conducted 12 months later, in 6/2012 (though sfety evaluation continues for 5 year for each woman). At that point BPAX can work on processing and analyzing data for a late 2012 NDA submission.
- Will BPAX get any efficacy data from this trial? The July 2011 Leerink Swann analyst report says yes - they claim that BPAX can use data from naturally menopausal women in this trial to show efficacy in this group (the efficacy trials described above include only surgically menopausal women). However, this is counter to my notes, including from 5/9/11 webcast in which BPAX said they would get "minimal efficacy data" from the safety study.