Navigate the Synta Pharma SNTA Research Notes Pages
- Introduction (valuation, financials, outlooks, catalysts)
- Ganetespib - Background (HSP90, basic drug info, regulatory strategies and business development)
- Ganetespib - HSP90 inhibitor safety and toxicity (info on liver and ocular toxicities associated with this class of drugs)
- Ganetespib - NSCLC (lead programs in lung cancer including ALK+ group and combo w/ docetaxel)
- Ganetespib - Clinical Trials (complete list of trials in other indications and links to data presentations)
- Elesclomol & Pipeline (information about other drug candidates in the SNTA pipeline)
- HSP90 Inhibitors - learn more about targeting heat shock protein 90 and all inhibitors in development
Ganetespib Lead Program: Non-small Cell Lung Cancer (NSCLC)
I've separated my notes on this lead indication because it is so far ahead of the rest of the trials listed below and will be there key value driver for SNTA going forward. Note that this is the only ganetespib trial being funded by SNTA and will involve <$10m external costs in 2011
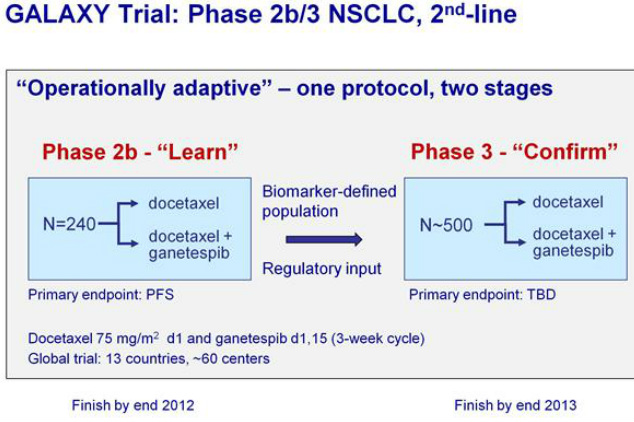
--- Phase 2b stage will involve 240 pts and a primary endpoint of progression-free survival (PFS)
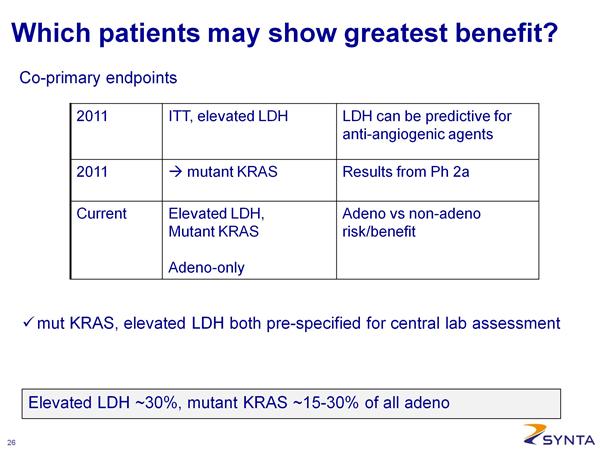
--- Phase 2b results will be used to choose a histological or biomarker-defined population for the phase 3 stage
--- Phase 3 portion will involve 400-600 patients and a primary endpoint of overall survival (OS)
--- Phase 2b results are expected in late 2011 or early 2012 (repeated 4/15/11 webcast)
--- The ongoing combination study have shown that the combination is well tolerated (complete phase 1 combination study results will be released later in 2011, see below for study details)
A) KRAS mutation (enrolled 15 pts)
B) EGFR mutation (enrolled 17 pts)
C) wild-type (WT, neither mutation- this represents 70% of all NSCLC cases, 25 pts as of 2/26/11 presentation)
--- The primary endpoint (at least 2 progression-free pts at 16 weeks) was met in the WT cohort (70% disease control rate, triggered enrollment of 9 additional patients)
--- Based on preliminary results, in 9/2010 SNTA expanded the trial to include additional WT pts and new cohorts
D) expansion of C) with up to 35 additional EGFR/KRAS wild-type patients, as well as up to 14 pts each with 3 undisclosed HSP90 client protein mutations or genotypes of interest (the goal being to identify a biomarker associated with gantespib response, see below)
E) combination with Docetaxel (pts who progressed but had some clinical benefit could roll over to this arm, n=5 pts as of 2/26/11 data presentation
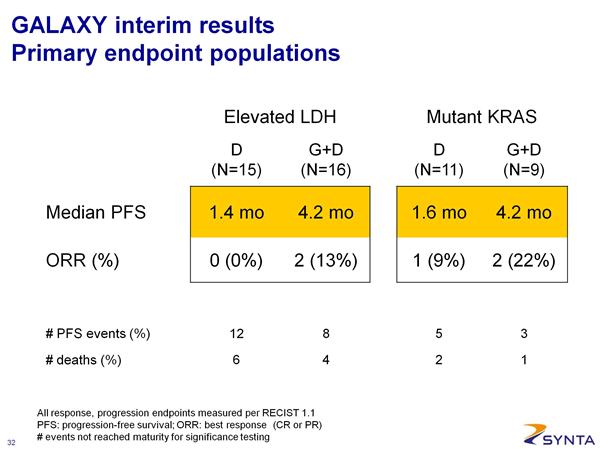
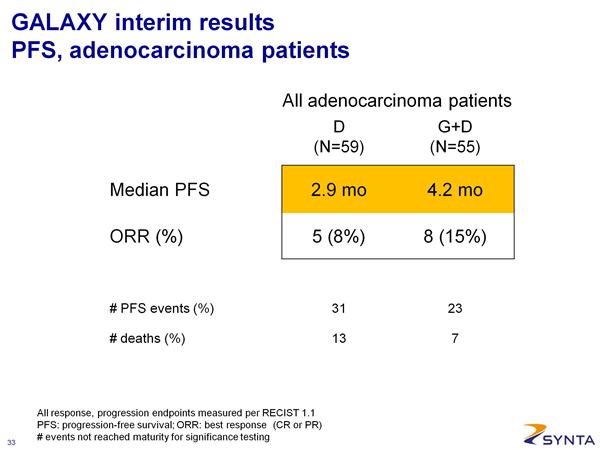
*** 2/26/2011: Interim data presented at a scientific conference. Here is a summary of the data reported (note that the PDF file of the presenation is also available on the SNTA website)
- Phase 2b/3 "GALAXY" NSCLC trial of docetaxel +/- gantespib --- (NCT01348126). SNTA-sponsored. First patient dosed in phase 2b portion 7/2011 (slightly behind forecast of 2q2011, Click here for PR).
--- Phase 2b stage will involve 240 pts and a primary endpoint of progression-free survival (PFS)
--- Phase 2b results will be used to choose a histological or biomarker-defined population for the phase 3 stage
--- Phase 3 portion will involve 400-600 patients and a primary endpoint of overall survival (OS)
--- Phase 2b results are expected in late 2011 or early 2012 (repeated 4/15/11 webcast)
- The above trial was announced 2/2011 (click for PR). 4/15/11 described as careful, thoughtful clinical program- not rush into 800 pts p3 trial as we have been encouraged to do.
--- The ongoing combination study have shown that the combination is well tolerated (complete phase 1 combination study results will be released later in 2011, see below for study details)
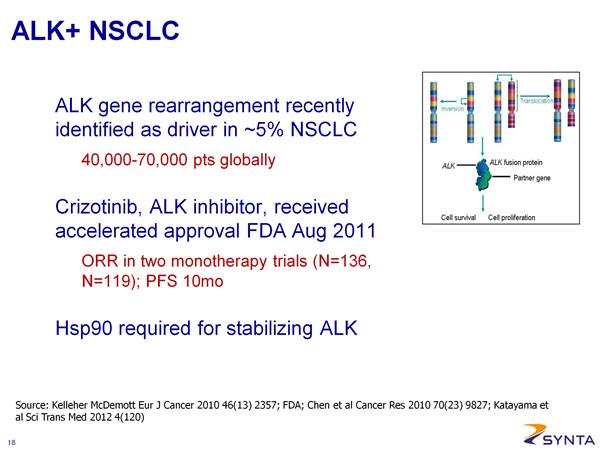
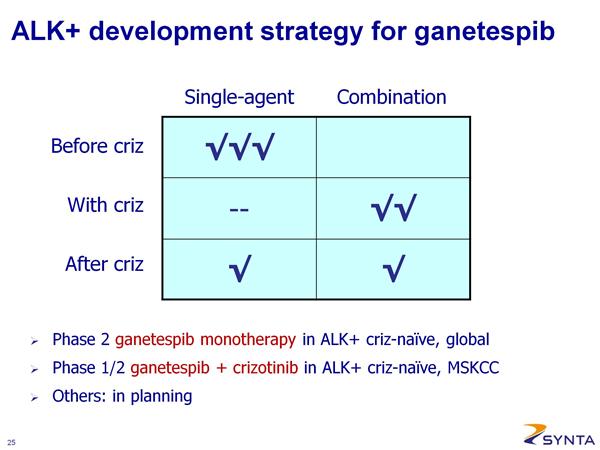
- Phase 2 (potentially pivotal) open-label in ALK+ NSCLC --- (NCT01562015). SNTA-sponsored. Targeting 100 patients. Receive weekly treatment 3/4 weeks. Recruiting as of May 2012.
- Phase 1/2 IST in combination with crizotinib (XALKORI) in ALK+ NSCLC (NCT01579994, 5/2012 active and recruiting, enrollment started April 2012). Run at Memorial Sloan Kettering Cancer Center. Dose escalate with ganetespib 100, 150, 200 mg/m2 (18 patient 3+3 phase 1 portion design). Phase 2 portion primary endpoint is PFS. ALK status determined by break-apart FISH assay.
- Phase 2 open-label in stage IIIb or IV non-small cell lung cancer (NSCLC) ---(NCT01031225). SNTA-sponsored, weekly dosing, first patient enrolled 1/2010. Listing says this trial is still enrolling as of 10/2011. Progression in this population is generally about 8 weeks (3/14/11 webcast)
A) KRAS mutation (enrolled 15 pts)
B) EGFR mutation (enrolled 17 pts)
C) wild-type (WT, neither mutation- this represents 70% of all NSCLC cases, 25 pts as of 2/26/11 presentation)
--- The primary endpoint (at least 2 progression-free pts at 16 weeks) was met in the WT cohort (70% disease control rate, triggered enrollment of 9 additional patients)
--- Based on preliminary results, in 9/2010 SNTA expanded the trial to include additional WT pts and new cohorts
D) expansion of C) with up to 35 additional EGFR/KRAS wild-type patients, as well as up to 14 pts each with 3 undisclosed HSP90 client protein mutations or genotypes of interest (the goal being to identify a biomarker associated with gantespib response, see below)
E) combination with Docetaxel (pts who progressed but had some clinical benefit could roll over to this arm, n=5 pts as of 2/26/11 data presentation
*** 2/26/2011: Interim data presented at a scientific conference. Here is a summary of the data reported (note that the PDF file of the presenation is also available on the SNTA website)
- 36 pts evaluated for safety, 33 pts evaluable for efficacy from cohorts C and D (see definitions above)
- These are pretreated patients (median 2 prior therapies, max was 10 prior therapies) and (3/14/11, 4/15/11 webcasts emphasized) are required to have progressive disease (ie, tumors growing with some velocity), which makes it harder to demonstrate clinical activity compared to in pts with stable disease
- Most common side effects were diarrhea, nausea, fatigue, and loss of appetite, most mild-moderate severity. Diarrhea is most serious, but if pre-treat with OTC medications, reduce the number of bowel movements from ~12 to 2-3 in first 24 hrs after infusion-- easily managed
- No serious liver or ocular toxicities observed
- 22 of 33 pts had disease stabilization (<20% increase), 10 of these had lesion shrinkage, including 3 confirmed partial response (PR) of durations 6, 6, and 14 months and each is ongoing still
- 76 pts evaluable (14 in cohort A, 13 in cohort B, 48 in cohorts C and D - see definitions about).
- Overall disease control rate at 8 weeks (complete response CR + partial response PR + stable disease SD = 54%). Objective Response Rate (ORR, consisting of CR+PR) was 5.3% (4/76)
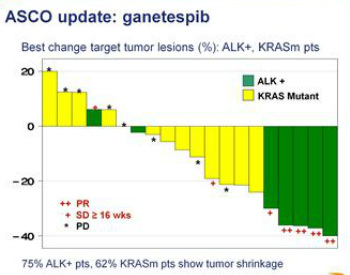
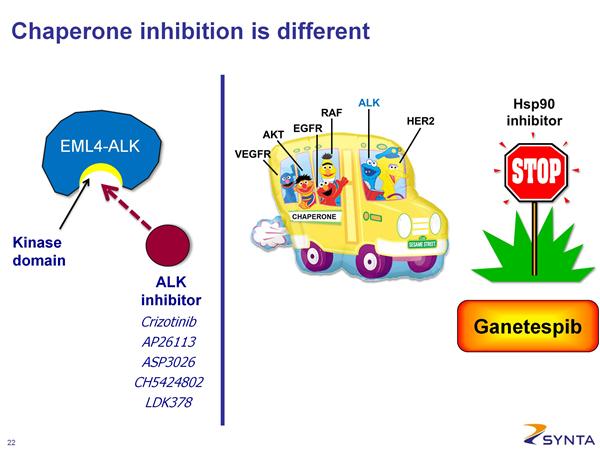
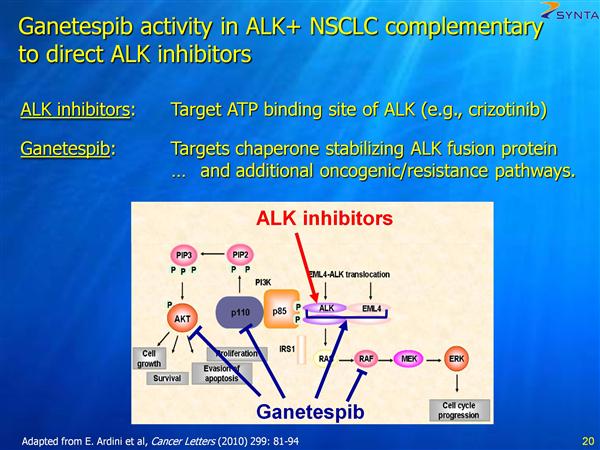
- 23/48 cohort C/D pts were tested for ALK rearrangement. 6/8 (75%) of patients with ALK rearrangement had tumor shrinkage, including 4 durable, objective responses (ORR = 50%). 1/8 pts had no change in tumor size, 1/8 pts had SD (<20% growth). 7/8 received ganetespib drug for at least 16 weeks. (Note that PFE drug Xalkori aka crizotinib for ALK population, and a certain future combo trial pairing, was approved 8/26/11 - click here for PR)
- Cohort A (EGFR mutation): 5/14 (36%) had tumor shrinkage, 7/14 (50%) had stable disease. No PR or CR.
- Cohort B (kRas mutation): 8/13 (62%) had tumor shrinkage. 5/13 (38%) had SD by RECIST criteria. No PR or CR.
- Side effects included grade 3 fatigue, dyspnea, or diarrhea in 9-12% of pts. 78% of pts experience diarrhea, 50% fatigue, 38% nausea, loss of appetite in 33%.