Navigate the Synta Pharma SNTA Research Notes Pages
- Introduction (valuation, financials, outlooks, catalysts)
- Ganetespib - Background (HSP90, basic drug info, regulatory strategies and business development)
- Ganetespib - HSP90 inhibitor safety and toxicity (info on liver and ocular toxicities associated with this class of drugs)
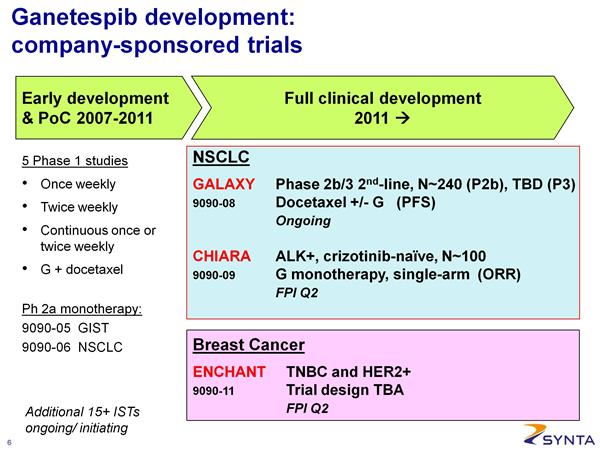
- Ganetespib - NSCLC (lead programs in lung cancer including ALK+ group and combo w/ docetaxel)
- Ganetespib - Clinical Trials (complete list of trials in other indications and links to data presentations)
- Elesclomol & Pipeline (information about other drug candidates in the SNTA pipeline)
- HSP90 Inhibitors - learn more about targeting heat shock protein 90 and all inhibitors in development
Ganetespib (f.k.a. STA-9090) Overview
- Injectable, synthetic heat shock protein 90 (HSP90, click here for basic info) inhibitor for cancer. Click here for more info on why HSP90 inhibition is rationale target for cancer therapy.
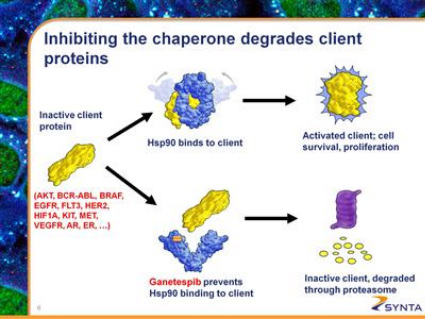
- 2010 10k: "Ganetespib potently inhibits Hsp90, a chaperone protein required for the proper folding and activation of other cellular proteins, particularly kinases. Many of these "client proteins" of Hsp90—such as AKT, BCR-ABL, BRAF, KIT, MET, EGFR, FLT3, HER2, PDGFRA, VEGFR—have been shown to be critical to cancer cell growth, proliferation, and survival and are the targets of clinically validated and approved cancer drugs, such as Gleevec, Avastin, Herceptin, Sutent, Nexavar, Tarceva, and Erbitux."
- COM patent offers worldwide protection to mid 2020's, formulation and biomarker patents extend several additional years. 2010 10k: "With respect to our Hsp90 inhibitor program, we have 6 issued U.S. and foreign patents, and 104 pending U.S. and foreign counterpart patent applications. Any U.S. or foreign patent that issues covering ganetespib will expire no earlier than 2025. Our Hsp90 inhibitor patent portfolio covers ganetespib and structurally related analogs, pharmaceutical compositions, and methods for treating cancer. Additionally, we have multiple U.S. and corresponding foreign patent applications directed to other Hsp90 inhibitors."
- Structure has not been disclosed-described in literature citation below as a "resorcinol derivative." Relevant US patents may be: 7608635, 7662813,7825148
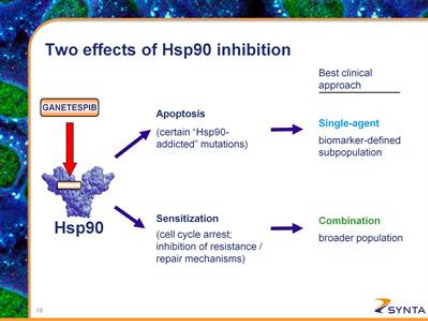
- Dual mechanism of action as single agent and in combination with other agents- see figure below
- Most advanced HSP90 inhibitor in clinical development (3/14/11: asserted recently by investigators at TAT meeting in Europe), 11 phase 2 trials ongoing
- 10-100x more potent (median 47x among >100 tested cancer cell lines) and structurally unrelated to first generation ansamycin family inhibitors such as 17-AAG (development stopped due to weak potency and liver toxicity-linked to a reactive moiety on 17aag) and IPI-504
- This class of Hsp90 inhibitors preferentially accumulate 15-20x more in tumor vs. normal cells and includes:
- Ganetespib: p1 disease control rate 57% (24/42 pts), <3% ocular toxicity at MTD in >350 pts
- AUY-922 (NVS and Vernalis): p1 disease control rate 18% (18/101 pts), 89% ocular toxicity at 70mg/m2 dose. Does not appear on NVS pipeline submissions planned 2011-2015 but there are still numerous ongoing clinical trials of AUY922
- AT13387 (Astex): p1 disease control rate 33% (7/21 pts), >50% ocular toxicity at MTD of 120 mg/m2. Click here to see info at Astex pipeline website or check out SUPG research page now that they have proposed a merger
- SNX-5422 (PFE): no p1 data available, terminated due to ocular toxicity. PFE acquired this drug by buying Serenex for undisclosed price. There are no active clinical trials for SNX-5422 and I don't see any mention on PFE's latest pipeline chart (February 2011)
- Ocular toxicity has caused the abandonment (late 2010) of competitors including AUY-922 (9/10 at MTD had ocular toxicity, NVS) and SNXX5422 (PFE). These drugs (and 17dmag) caused apoptosis in the layer of photoreceptor cells, where protein folding and unfolding is critical. 3/14/11: Ganetespib (and 17-AAG) are less soluble than the above compounds and therefore do not accumulate in these cells to the same degree (see figure below on left).
- Additional competitors include: MPC-3100 (Myrexis, p1), IPI-504 and IPI-493 (Infinity Pharma), AUY922 and HSP990 (Novartis and Vernalis), KW-2478 (Kyowa Hakko Kirin), XL888 (Exelixis), Debio0932 (Curis and Debiopharma), ABI-010 (CELG)- click here for review article listing drugs in development
- 2010 10k: Oral and injectable backup compounds are in preclinical lead optimization stage
- Impressive preclinical results observed in naturally-occuring canine tumors (3/14/11: said publication would come out this year)
- As of 10/2010: four p1 trials are wrapping up, single agent CR+PR+SD >50%. 3/14/11: durable single-agent partial responses in patients with numerous tumor types
- As of 11/15/11, > 400 patients have been treated with ganetespib
Ganetespib: Clinical and Regulatory Path to Market
In a February 2011 investor presentation, SNTA outlined the opportunities and strategies available to develop the drug towards registration-enabling studies
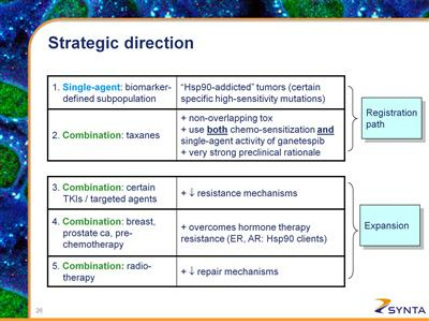
- The options presented include the following:
- Single agent trial in a biomarker-defined population (2010 10k: "Certain specific gene mutational profiles have been associated with "Hsp90 addicted" cancers, in which treatment with ganetespib as a single agent has potential to produce pronounced, durable tumor regression.")
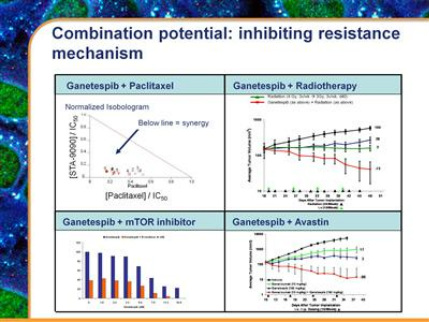
- Combination trial with taxanes-ie, docetaxel and paclitaxel, used in 500,000 US pts per year (2010 10k: "Preclinical evidence suggests pronounced synergy between the underlying anti-cancer effects of taxanes and Hsp90 inhibition. We believe that this synergy may be due to the ability of Hsp90 inhibition to sensitize cells to killing by taxanes, and to suppress certain mechanisms by which cells escape killing by taxanes.")
- Combination trial with certain tyrosine kinase, angiogenesis, or proteasome inhibitors (2010 10k: "Certain compounds with these mechanisms have shown promising preclinical evidence of synergy in combination with ganetespib. These include compounds that inhibit VEGF, such as Avastin; compounds that inhibit EGFR, such as Tarceva or Iressa; compounds that inhibit the IGF1R, PI3K/mTOR/AKT pathway, such as Torisel; and compounds that act via proteasome inhibition, such as Velcade.")
- Combination trial with hormonal therapy in breast and/or prostate cancer (2010 10k: "Because estrogen receptor and androgen receptor are client proteins of Hsp90, and because early stages of breast cancer and prostate cancer are believed to be due to aberrant signaling through these receptors, treatment with ganetespib may provide benefit in these earlier stages of breast cancer, before chemotherapy is used. Data presented at the San Antonio Breast Cancer Symposium in December 2010 showed that coadministration with ganetespib can restore sensitivity to and improve the anti-cancer activity of hormone receptor antagonist therapy in breast cancer.")
- As a sensitizer for radiation therapy (2010 10k: "Radiation therapy causes a rapid increase in stress inside cells and the unfolding and subsequent inactivation of proteins, leading to cell death. Hsp90 provides an important means by which cells repair protein unfolding and recover from this increased stress. By inhibiting Hsp90 function, we believe that ganetespib can suppress this repair mechanism. Preclinical experiments showed promising synergy of ganetespib in combination with radiotherapy.")
- Note that options #1 and #2 would be the initial registration-enabling trials, while other options #3-5 would be to expand the indication for gantespib
- On 2/26/11, SNTA announced that the first advanced, controlled trial would be "Door #2," a combination trial with docetaxel- see below for details on the trial design. This trial will initiate in 2q2011
- 3/14/11: Option #1 is comparable to PFE's ALK inhibitor Xalkori (crizotinib) and Roche/Plexxicon's PLX4032 and would be a population of about 3000 pts per year. The larger combination indication would be 100x larger.
- An indication for radiotherapy sensitization would be larger than all chemotherapy combined. April 2010 AACR data (click for link) suggests that ganetespib inhibits the repair mechanisms induced in response to radiation-induced DNA double stand breaks, further reinforcing the scientific basis of this combo. PR reiterated that a trial of ganetespib as a radiotherapy sensitizer would start in 2011
- 4q2011 conference call- reiterated that SNTA would pursue options #1 and #2 in parallel