Navigate the Synta Pharma SNTA Research Notes Pages
- Introduction (valuation, financials, outlooks, catalysts)
- Ganetespib - Background (HSP90, basic drug info, regulatory strategies and business development)
- Ganetespib - HSP90 inhibitor safety and toxicity (info on liver and ocular toxicities associated with this class of drugs)
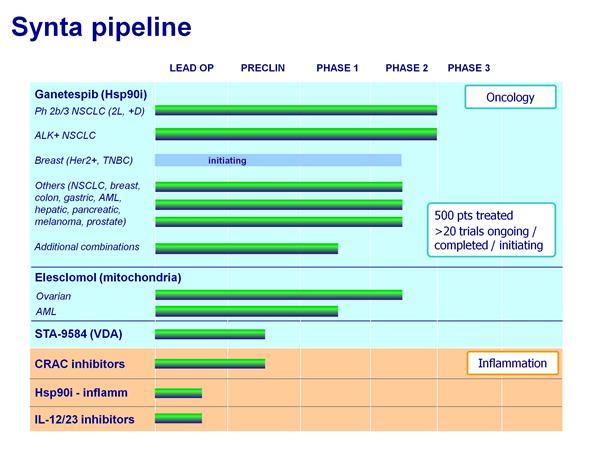
- Ganetespib - NSCLC (lead programs in lung cancer including ALK+ group and combo w/ docetaxel)
- Ganetespib - Clinical Trials (complete list of trials in other indications and links to data presentations)
- Elesclomol & Pipeline (information about other drug candidates in the SNTA pipeline)
- HSP90 Inhibitors - learn more about targeting heat shock protein 90 and all inhibitors in development
Synta Pharmaceuticals (SNTA)Website link: www.syntapharma.com
*** Click here for archive of all blog posts related to SNTA *** Archive of all SNTA related blog posts and articles: Ganetespib data presentation and clinical trial announcements 2/26/2011 4q10 conference call 3/11/11 Roth presentation 3/14/11 (click here for notes) Barclays presentation 3/16/11 Ganetespib NSCLC webcast 3/25/11 (click here for notes) 4/15/11 webcast (click here for notes) 1q2011 earnings 5/5/11 (click here for notes) 6/4/11 data presentation at ASCO (click here for abstracts/notes) 6/9/11 Jefferies webcast (click here for notes) 8/4/11 2q11 earnings (Click here for notes) 8/11/11 Canaccord Genuity webcast (Click here for notes) 9/13/11 Rodman and Renshaw webcast (Click here for notes) 11/15/11 Lazard webcast (click here for notes) 2/2012 BIO CEO conference (click here for notes) 3/2012 Citi Healthcare conference (click for notes) 3/2012 Cowen Healthcare conference (click for notes) 4/2012 Roth Captial Growth conference (click for notes) 5/2012 ASCO presentations (click for blog) 6/2012 Jefferies Healthcare conference (click for notes) 7/2012 JMP healthcare conference webcast (click for notes) Links
12/5/10: "Synta's lead program gaining momentum" 3/25/11: "Putting a price tag on Synta's lead agent" 7/3/11: "Top 10 Oncology Drugs available for partnering" |
|
Guidance and Outlook
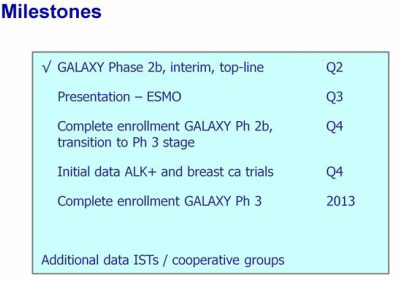
- 4/15/11 presentation: 2011 Events (see above)- Elescmol trial start date has slipped slightly, Predict one or more partnerships from above listed 3 lead programs in 2h2011
- 7/2011: signed unique CRO deal for ganetespib and elesclomol clinical trials with Ergomed. SNTA gets CRO services at reduced rates in return for a percentage of licensing and/or commercial revenues, up to a preset (undisclosed) cap.
- 2/2011: Two late-stage oncology candidates into late-stage trials 2011, each w/ biomarkers to enable patient population enrichment strategies
---Elescomol p2b trial in NSCLC to begin 2q11
- Partnering: 2/2011: In many discussions about deals with a variety of structures (single or multiple drugs, regional, etc), highest focus is still on ex-US and EU deal for Ganetespib. 2/26/11 press release confident in one or more deals this year 3/14/11 presentation perhaps even more bullish, slide language was changed to one or more deals in 1h2011
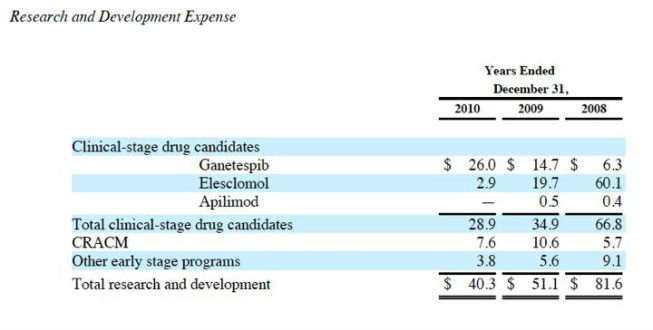
- March 2011: NSCLC is only ganetespib trial SNTA is paying for in 2011, external costs <$10m this year
- 2010 10k: proprietary 100,000x library of small molecules (not from combinatorial chemistry) and natural extracts
- 2010 10k: "As of December 31, 2010, we had 112 full time employees, including a total of 46 employees who hold M.D. or Ph.D. degrees. 82 of our employees are primarily engaged in research and development activities, and 30 are primarily engaged in general and administrative activities."
- 2010 10k: "Our directors, executive officers and principal stockholders, together with their affiliates and related persons, beneficially own, in the aggregate, approximately 45% of our outstanding common stock."
Financials
- 5/5/11 and 1q2011 10q: $75m cash on hand sufficient into 2h2012
- 8/2011: filed new $150m shelf registration to replace expiring one. This includes the $35m equity standby with Azimuth
- 4/15/11: sold 7.2m shares at $4.89 for net proceeds of $35m (click here for press release). Comment at webcast: offering was insider driven, self managed, did not use a placement agent or underwriting company... Board of Directors (3 individual members according to this afternoon's SEC filing bought 1.5m of the 7.2m shares sold) felt company was attractively valued in advance of upcoming events- including increased visibility and momentum as enter large pivotal program and numerous additional trials "as well as some partnerships that we expect to come in the 2nd half of the year." A number of institutional investors came in alongside the BoD. 70% of shares were bought by long-only mutual funds. This strengthens balance sheet as go into partnerhsip discussions, expected later this year
- 2010 10k: "In October 2010, we entered into a common share purchase agreement, or Purchase Agreement, with Azimuth Opportunity Ltd., or Azimuth, pursuant to which we obtained a committed equity line of credit facility, or Facility, under which we may sell up to a maximum of $35 million or 8,106,329 shares of our common stock, whichever is fewer, over the 18-month term of the agreement, subject to certain conditions and limitations." No shares sold as of 6/30/11
- 2010 10k: "On September 30, 2010, we entered into the $15 million GECC Term Loan. Interest on the borrowings under the GECC Term Loan accrues at an annual rate of 9.75%. We will make interest-only payments through June 2011, followed by 27 equal monthly payments of principal plus accrued interest on the outstanding balance. In addition to the interest payable under the GECC Term Loan, we paid origination fees in the amount of $150,000 and are obligated to pay an exit fee of $450,000 at the time of the final payment of the outstanding principal. These amounts are being amortized and accreted, respectively, to interest expense over the term of the GECC Term Loan. We also paid approximately $177,000 of legal fees and expenses in connection with the GECC Term Loan. These expenses have been deferred and, together with the $150,000 origination fees, are included in other assets, and will be expensed over the term of the GECC Term Loan. In the year ended December 31, 2010, we recognized approximately $453,000 in interest expense related to the GECC Term Loan." Payments will total $4.7m in 2011, $7.5m in 2012, and $5.6m in 2013
- 7/2011: GE loan amended again. If SNTA raises $30m cash by 12/30/2011 via partnering or stock sale, then SNTA will repay principal of loan in 30 equal installments starting 2/1/2012 - maturity would then be 7/1/2014. Click here for details.