Navigate the Alnylam $ALNY Notes Pages
- Introduction (valuation, milestones, pipeline, general commentary)
- RNAi Background (science, delivery technologies, patents, litigation)
- ALN-TTR (lead 5x15 program for transthyretin amyloidosis)
- 5x15 Programs (details on 5x15 strategy and info on additional preclinical programs)
- Partnered Programs (partnered clinical and preclinical assets falling outside the 5x15 focus area)
- Alliances and Licensing (RNAi platform deals and other licensing agreements with pharma companies)
ALN-TTR01
- RNAi therapeutic targeting the transthyretin (TTR) protein being developeding for TTR-mediated amyloidosis (ATTR).
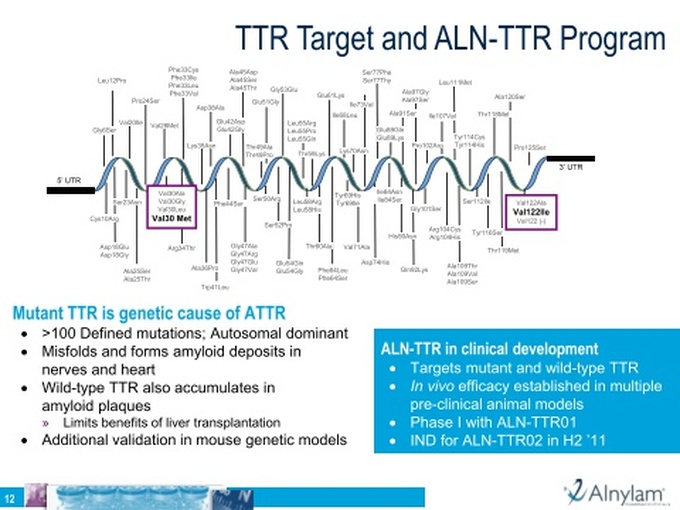
- Market Opportunity: “ATTR is a hereditary, systemic disease caused by a mutation in a protein predominantly made in the liver, known as TTR. Mutations in this protein result in the accumulation of toxic deposits of the wild-type and mutant protein in several tissues, including the peripheral nervous system, heart and/or gastrointestinal tract, which leads to FAP and/or familial amyloidotic cardiomyopathy, or FAC. FAP is associated with severe pain and loss of autonomic nervous system function, whereas FAC is associated with heart failure. Typical onset for ATTR occurs between the fourth and sixth decades of life, and the disease is often fatal within five to 15 years of onset. In its severest form, ATTR represents a significant unmet medical need with high rates of morbidity and mortality. ATTR is an orphan, or rare, disease, affecting approximately 50,000 people worldwide.”
- 5/25/11 webcast: Over 100 different mutations defined, but don't expect to need a companion diagnostic and expect to work with all mutations because the drug targets the highly-conserved 3' UTR region.
- Current Treatments: “There are no existing disease-modifying treatments for ATTR. Currently, liver transplantation is the only available treatment for FAP. However, less than 3,000 FAP patients qualify for this costly and invasive procedure and, even following liver transplantation, the disease continues to progress for many of these patients, presumably due to normal TTR being deposited into preexisting fibrils. Moreover, there is a shortage of donors to provide healthy livers for transplantation [5/25/11 webcast: only 3000 pts per year eligible]. The only currently available treatments for FAC are aimed at relief of symptoms, such as diuretics, or water pills, to treat the swelling of the ankles, one of the symptoms of FAC.”
- “TTR is a carrier for thyroid hormone and retinol binding protein and is produced almost exclusively in the liver. We believe TTR is a suitable target for an RNAi therapeutic formulated to maximize delivery to liver cells. ALN-TTR targets wild-type and all known mutant forms of TTR, including the predominant V30M mutation, which is the major mutation of ATTR, particularly in FAP, and therefore is a potential therapeutic for the treatment of all forms of ATTR, including FAP and FAC.”
- 7/2010 initiated p1 trial of systemically delivered RNAi (first generation lipid nanoparticle). Randomized, double-blind, placebo-controlled dose escalation in UK, Portgual, Sweden, France (cohorts from 0.01 to 0.4 mg/kg single IV dose). 5/2/11: received approval for additional cohorts up to 1.0 mg/kg, increasing total to 36 pts. 5/25/11 webcast: In addition to TTR levels, also measuring vitamin A, retinin protein binding, clinical features (but don't expect meaningful clinical outcomes)
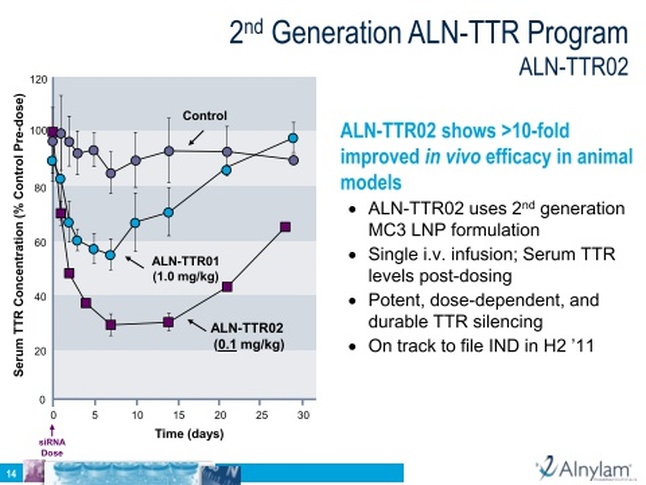
- 6/2011: on track to report placebo-controlled phase 1 data in 3q2011 (5/25/11webcast: blood levels of TTR before and after treatment)...What will be considered a good result? as little as 50% KD over time is sufficient for regression of amyloid deposition. Would hope to get doses giving 50-80% KD to advance into pivotal studies. Hope to achieve with TTR01, and certainly think it is achievable in TTR02 backup program with next generation LNPs (5/25/11 webcast)
- 1/2011 received EU orphan designation (10 yrs exclusivity and other benefits).
- 6/2011: presented data at Peripheral Nerve Society Meeting: "conducted a natural history study to measure serial TTR levels over a four week period in ATTR patients and gene carriers, and to determine the intra- and inter-patient variability in serum TTR. The study enrolled 26 patients and gene carriers with seven different amyloidogenic TTR mutations, the most common being the Val30Met mutation that is the primary cause of familial amyloidotic polyneuropathy (FAP). The data from this study showed that TTR levels were stable over time in patients and carriers. The average serum TTR concentration was approximately 200 micrograms/mL with 10 to 20% intra-subject variability over the 28-day observation period. Results showed that inter-subject TTR variability was approximately 25%...Alnylam scientists and collaborators also presented pre-clinical data from its ALN-TTR program, including new data demonstrating improved potency with ALN-TTR01 using a loading dose/maintenance dose regimen." - Click here for PR.
- Competitor: Isis Pharma, in partnership with GSK, is developing an antisense treatment for TTR (Click for details on the ISIS research page)
- 11/2011 - presented phase 1 data - click here to download PDF of slide deck.
Competitor: Tafamidis from Pfizer PFE (via FoldRx acquisition)
- In 2010, Pfizer (via purchase of FoldRx) “filed a marketing authorization application, or MAA, for tafamidis, an oral small molecule stabilizer of TTR, with the EMA. Tafamidis has orphan drug status in the European Union, or EU, for the treatment of FAP associated with ATTR.”
- 4/2011: Click here for details of phase 2/3 data results.
- Now approved and sold in Europe as VYNDAQEL
- Under review in US by FDA with June 2012 PDUFA date.
ALN-TTR02
- Backup to ALN-TTR01, utilizes 2nd generation LNP formulation and is being advanced in parallel.
- Preclinical- reduced mRNA levels in liver and circulating protein levels in transgenic mice, prevents deposits of mutant protein in multiple tissues
- 5/2/11: plan IND 2h2011