Navigate the Alnylam $ALNY Notes Pages
- Introduction (valuation, milestones, pipeline, general commentary)
- RNAi Background (science, delivery technologies, patents, litigation)
- ALN-TTR (lead 5x15 program for transthyretin amyloidosis)
- 5x15 Programs (details on 5x15 strategy and info on additional preclinical programs)
- Partnered Programs (partnered clinical and preclinical assets falling outside the 5x15 focus area)
- Alliances and Licensing (RNAi platform deals and other licensing agreements with pharma companies)
ALN-PCS
- RNAi therapeutic targeting the proprotein convertase subtilisin/kexin type 9 (PCSK9) protein for severe hypercholesterolemia. Program started in collaboration w/ UT-Southwestern
- Click here for a nice Reuters piece about PCSK9 and its discovery as a cardiovascular target.
- Systemically delivered RNAi utilizing 2nd generation LNP technology (MC3) - first with this technology (which is subject to legal dispute with TKMR) to enter clinic.
- Market Opportunity: “Coronary artery disease, or CAD, is the leading cause of mortality in the United States, responsible for 40% of all deaths annually. Hypercholesterolemia, defined as a high level of LDL-c, or bad cholesterol, in the blood, is one of the major risk factors for CAD. This condition occurs when excess LDL-c in the bloodstream is deposited in the walls of blood vessels. The abnormal buildup of LDL-c forms clumps, or plaque, that narrow and harden artery walls. As the clumps grow, they can clog the arteries and restrict the flow of blood to the heart. The buildup of plaque in coronary arteries increases a person's risk of having a heart attack. Although current therapies are effective in many patients, studies have shown that as many as 45% of high-risk patients with elevated LDL-c do not achieve adequate control of their high cholesterol level with existing treatments, which include drugs known as statins. Currently, in the United States, there are more than 500,000 patients with high cholesterol levels not controlled by the use of existing lipid lowering therapies. These patients are viewed as having severe hypercholesterolemia and constitute a potential target population for ALN-PCS.”
- 5/25/11 webcast: 500k pts with LDL >200 mg/dL - statins are inadequate, at risk for early/recurrent/fatal MI
- Current Treatments. “The current standard of care for patients with hypercholesterolemia includes the use of several agents. The first treatment often prescribed is a drug from the statin family. Commonly prescribed statins include Lipitor® (atorvastatin), Zocor® (simvastatin), Crestor® (rosuvastatin) and Pravachol® (pravastatin). A different type of drug, such as Zetia® (ezetimibe) and Vytorin® (ezetimibe/simvastatin), which reduces dietary cholesterol uptake from the gut, may also be used either on its own or in combination with a statin. Despite these therapies, there are many patients who have severe hypercholesterolemia and require more intensive treatment. In addition, some patients do not tolerate current treatments, with an estimate, based on extensive clinical study results, of at least five percent of those treated with a statin having to stop such treatment because of side-effects. In patients with very high uncontrolled cholesterol levels, a procedure called lipid apheresis is used, which effectively removes cholesterol from the blood using a machine specifically designed for this process. However, this procedure is inconvenient and uncomfortable, requiring regular weekly visits to a doctor's office.”
- PCSK9 “is involved in the regulation of LDLR levels on hepatocytes and the metabolism of LDL-c. PCSK9 is a widely acknowledged target for the treatment of hypercholesterolemia. PCSK9 is a protein that is produced by the liver and circulates in the bloodstream. The liver determines cholesterol levels, in part by taking up or absorbing LDL-c from the bloodstream. PCSK9 reduces the liver's capacity to absorb LDL-c. Published studies indicate that, if PCSK9 activity could be reduced, the liver's uptake of LDL-c should increase and blood cholesterol levels should decrease. In fact, published case reports have shown individuals with a genetic mutation in PCSK9 that lowers its activity and results in increased liver LDL-c uptake and decreased blood cholesterol levels. In turn, these individuals have been shown to have a dramatically reduced risk of CAD, including myocardial infarction or heart attack. In addition, studies have shown that PCSK9 levels are increased by statin therapy while LDL-c levels are decreased, suggesting that the introduction of a PCSK9 inhibitor to statin therapy may result in even further reductions in LDL-c levels.”
- Competitor: Isis Pharma, in collaboration with BMS, is developing an antisense therapeutic against PCSK9, currently in preclinical studies (Click here for more info on the Isis research page)
- Preclinical >50% reduction in LDL and reduced PCSK9 plasma levels in non-human primates. - these responses were durable for weeks after a single injection.
- 5/2011 (1q11 call and 5/25 webcast): expect IND 2q2011 and expect to report human POC data in 4q2011.
- 7/2011: announced filing of application to begin phase 1 trial in the UK (Click here for PR) - "Randomized, single-blind, placebo-controlled, single ascending dose study, enrolling approximately 32 healthy volunteer subjects with elevated baseline LDLc (>116mg/dL). The primary objective of the study is to evaluate the safety and tolerability of a single dose of ALN-PCS, with patients being enrolled into five sequential cohorts of increasing doses ranging from 0.015 to 0.25 mg/kg. Secondary objectives include characterization of plasma and urine pharmacokinetics of ALN-PCS, and assessment of pharmacodynamic effects of the drug on plasma PCSK9 protein and LDLc levels measured from serial blood samples prior to and following dosing"... expect to report safety, tolerability, and clinical activity data by ye2011 (very aggressive timeline!)
- Click here to read a blog post regarding this phase 1 trial initiation (not mine...)
- 9/2011 initiated Phase 1 trial - Click for PR - click for ClinicalTrials.gov listing.
- See below embedded slide deck from March 2010 scientific presentation on this program.
ALN-APC
- Click to download slides about this program (protected so I cannot extract images)
- RNAi therapeutic targeting Activated Protein C for hemophilia (announced 9/2011 as fourth 5x15 program)
- Hemophlias are X-linked recessive bleeding disorders: "A" type involves loss of function of Factor VIII (40,000 patients in US+EU); "B" type involves loss of function of Factor IX (9,500 patients in US+EU)
- Target market is severe Hemophilia A patients are called "Inhibitor" patients (6,500 in EU+US) and represent high medical cost and unmet need (average of >8 bleeds, >5 hospital days, and $300,000 costs per patient per year, with very poor quality of life)
- Only current therapies for Inhibitor patients are NovoSeven (recombinant Factor VIIa) and FEIBA - both lack optimal efficacy, have short half-lives, and require frequent dosing. <10% of Inhibitor patients receive chronic care. Next generation products shift half-life somewhat but unlikely to change treatment paradigm.
- Seek to provide treatments that provide extended protection from bleeds (prophylaxis or during high risk times)
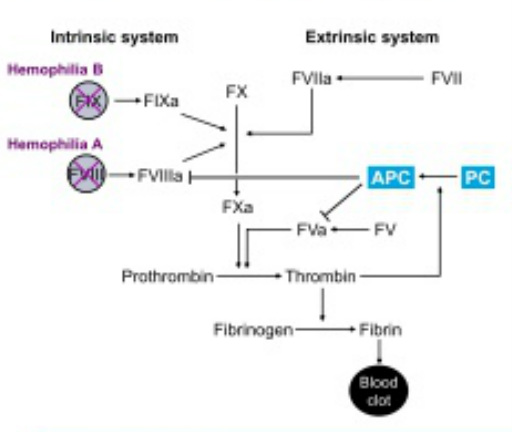
- Activated Protein C (APC) inactivates factors Va and VIIIa and attenuates thrombin generation. Expressed in Liver and circulates in plasma. Patients with Heterozygous PC deficiency have increased thrombin generation.
- APC resistance (Factor V-Leiden) patients - this co-inheritance is associated with milder bleeding.
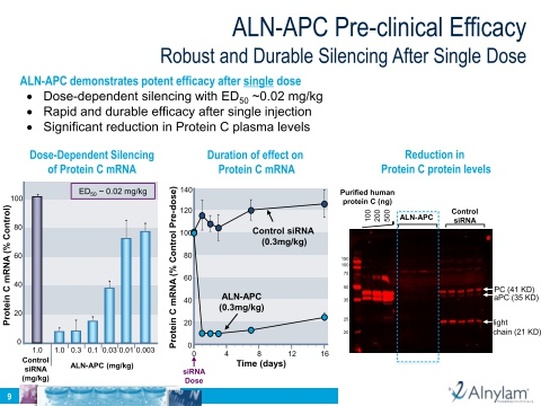
- "At the 7th Annual Meeting of the Oligonucleotides Therapeutics Society being held in Copenhagen, Denmark between September 8-10, 2011, Alnylam presented pre-clinical data from its new ALN-APC program. Specifically, ALN-APC showed dose-dependent silencing of the protein C mRNA with an ED50 of 0.02 mg/kg. When administered as a single intravenous dose of 0.3 mg/kg, the LNP-formulated siRNA achieved 90 percent silencing of protein C mRNA within 24 hours with effects lasting for over two weeks. Further, administration of the siRNA resulted in marked reductions in protein C plasma levels"
- 9/2011: program is in research to development transition, involving siRNA optimization, in vivo efficacy in preclinical animal models, and predict IND filing and phase 1 trial in 2013.
ALN-HPN
- RNAi therapeutic against the hepcidin protein (genetically validated gene involved in iron homeostasis) being developed for refractory anemia.
- 5/25/11 webcast: hepcidin is a central regulator of iron homeostasis that down-regulates ferroportin (reduced iron transport to bone marrow). can monitor serum iron levels in p1.
- Systemically delivered RNAi utilizing 2nd genertation LNP technology
- 5/2/11 and 5/25/11 webcasts: goal is IND filing in 2012
- Market Opportunity” “Anemia is the clinical manifestation of a decrease in circulating red blood cell mass and is usually detected by low blood hemoglobin concentrations. Symptoms include fatigue and dizziness, and generally have a significant impact on the patient's quality of life.
- Anemia of chronic disease, or ACD, occurs in patients with end-stage renal disease, or ESRD, cancer and chronic inflammatory disorders. There are also additional genetic causes, such as iron-refractory iron deficiency anemia. ACD patients who are refractory to erythropoiesis-stimulating agents, or ESAs, which stimulate red blood cell production, and intravenous iron, define a condition of refractory anemia for which there is a substantial unmet need. Currently in the United States, there are approximately 500,000 patients with ESRD and approximately 50,000 ESRD patients with refractory anemia.”
- Current Treatments: “There are several treatment options available for anemia, depending on its cause and severity, which may include oral or intravenous iron supplements, blood transfusions and ESAs. However, there are currently no approved therapies for the treatment of refractory anemia. Treatment for this condition is largely supportive, including blood transfusion in patients with symptomatic anemia.”
- “ Pre-clinical studies with an siRNA targeting hepcidin demonstrated the ability to silence the gene and increase serum iron levels.”