Navigate the Antares Pharma $ATRS stock research pages
Introduction: financials, highlights, pipeline, upcoming events and catalysts
Injectables: Vibex, HgH, Teva collaborations, Pen injectors, autoinjectors, MTX program
Vibex injectors: Includes epi-pen and undisclosed drug partnered with Teva
Vibex MTX and QST: Internal programs for injectable methotrexate and testosterone
Pen Injectors: Two undisclosed programs partnered with Teva
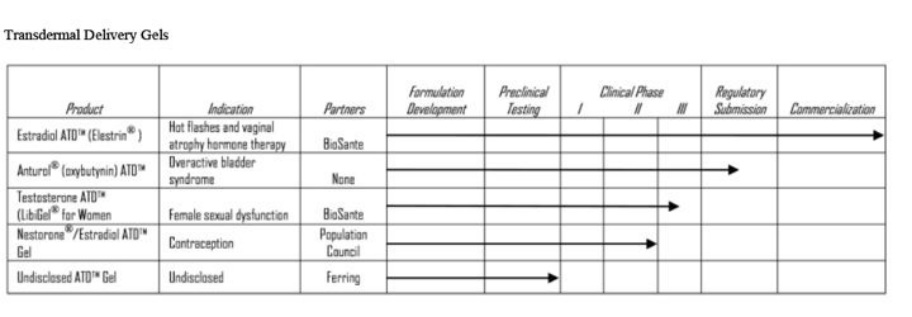
Gels: Transdermal technology, Elestrin, Nestragel, Libigel with BPAX
Anturol/Gelnique 3%: FDA-approved Overactive bladder gel licensed to Watson Pharma
Blog: Archive of all posts related to AIS/ATRS
Injectables: Vibex, HgH, Teva collaborations, Pen injectors, autoinjectors, MTX program
Vibex injectors: Includes epi-pen and undisclosed drug partnered with Teva
Vibex MTX and QST: Internal programs for injectable methotrexate and testosterone
Pen Injectors: Two undisclosed programs partnered with Teva
Gels: Transdermal technology, Elestrin, Nestragel, Libigel with BPAX
Anturol/Gelnique 3%: FDA-approved Overactive bladder gel licensed to Watson Pharma
Blog: Archive of all posts related to AIS/ATRS
Transdermal Gel Pump Technology Division
- ATD transdermal gel pumps a precise metered dose (can be titrated based on number of pumps used) that is applied to the skin and has a 24 hour release formulation for once daily use. Gel is applied to shoulder, upper arm, or abdomen and dries completely within 2 minutes (has been compared to Purel hand sanitizer consistency and absorption)
- 2010 10k: "We believe that our transdermal gels minimize first pass liver metabolism, gastrointestinal effects and skin erythema. Other advantages include cosmetic elegance and ease of application as compared to transdermal patches and have potential applications in such therapeutic markets as hormone replacement, overactive bladder, contraception, pain management and central nervous system therapies."
- 4/5/11 Needham: gel formulation contains proprietary permeation enhancers. 2010 10k: all components are GRAS certified substances (further discussion of GRAS on SNMX research page)
- In November 2009, AIS sold this division to Ferring, resulting in a significantly reduced cash burn for the company. AIS has said that they were satisfied with their existing pipeline and thought they got better return on investment in the injector products division.
- The sale does not affect current gel products in the pipeline, including Libigel, Elestrin, Anturol, Nestragel- AIS retains full ownership and financial gains.
- AIS received an undisclosed upfront payment and is entitled to milestones as Ferring develops new gel products (AIS said it received a "significant" development milestone in 2q2010
Elestrin: Marketed gel product
- Elestrin is a transdermal estrogen gel marketed by Jazz Pharma $JAZZ via their acquisition of Azur Pharma (sublicensed by BioSante Pharma BPAX) for the treatment of hot flashes (click here to visit product website)
- 3/15/11 at Roth: Azur is focused on women's health, based in Philadelphia, about 160 employees, mostly in sales
- 9/2011: Azur announces merger with Jazz Pharma. Merger presentation slides seemed to indicate ~$5m 2010 sales. Women's health sales force has 51 reps. Total women's health sales estimated at 10% of 2011 combined revenues, would correspond to $47.5m in first year of merged company. Slide shows Elestrin estimated to be ~20% of women's health sales in 2011.
- AIS receives 25% of any sublicense income paid to BPAX, as well as mid-single digit royalties
- Head over the the BPAX research page for any recent news regarding this product
- November 2010 analyst presentation: soon will relaunch with a new container and image
- Cowen 3/7/11: Weekly prescriptions have increased from <100 to >700 since Azur began promoting the product in March 2009, think this could be a $30-40m product
- Roth 3/15/11: product was relaunched in Dec 2010 with a smaller 1 month dispenser (previously was 3 month supply), have seen improved sales since then. Needham 4/5/11: liked by pts (smaller size) and payers (more frequent copays)
- 5/9/11 CC: sales showing steady growth after relaunch
Nestragel: contraceptive gel
- Gel contains a combination of the progestin Nestorone® and a form of estrogen, called 17!-estradiol (E2), which is chemically identical to the naturally occurring estrogen. Nesterone desirable because has no androgenic effects but not orally bioavailable and therefore cannot be used in oral contraceptive products.
- 31% of women discontinue oral contraceptives within 6 months, 44% within one year
- Partnership with the Population Council, who has also licensed a vaginal ring product of this combo. 2010 10k: "We are responsible for research and development activities as they relate to ATD formulation and manufacturing and the Population Council will be responsible for clinical trial design development and management. In 2010, we announced with the Population Council successful results from a dose-finding Phase II trial for the contraceptive gel. Together, we expect to identify a worldwide or regional commercial development partner as clinical data becomes available."
- May 2009 and January 2010 announced positive phase 2 results: 3 doses for 21 days, 18 pts, primary endpt of ovulation suppression was met successfully and well tolerated at all doses
- August 2010: Pop Council in looking for a partner and has had preliminary discussions. November 2010 analyst presentation: in partnering discussions now, could find the resources to move forward but would prefer to partner. 3/14/11 4q2010 "looking for potential partner with Population Council"
- 3/7/11 at Cowen: product is "gearing up to go to next stage"
- 3/14/11 4q2010 CC: there is interest in nesterone pgm from a couple players, esp one international compny in reasonablly advanced discussions- but no guidance on timing. 5/9/11 CC: Continue to look for partner, have some initial interest
- 4/5/11 Needham: Anticipate that Nesterone contraceptive gel will now move into a phase 2b trial run by the Population Council (so no partner right now I guess)
Libigel: a potential blockbuster product
- Libigel is a transdermal testosterone gel being developed by BioSante Pharma (click for BPAX research page) for hyposexual desire disorder (HSDD) in menopausal women- check that page for updates on Libigel clinical development and news items
- BPAX management was also involved in the approval of Androgel, a male testosterone gel
- This product has been in the news a great deal lately as the phase 3 efficacy and safety trials approach completion- however, all of this newsflow is focused on BPAX, when it has equally important implications for AIS
- BPAX licensed the development rights to Libigel for US and a few select other countries only (Canada, China, Australia, New Zealand, South Africa, Israel, Mexico, Malaysia and Indonesia)...AIS owns the development and commercialization rights in the rest of the world, including the EU and Japan. AIS also owns worldwide manufacturing rights so would make money on this aspect for the US market as well. AIS would receive 4.5% royalties on Libigel sales by BPAX (or partner) and is due 25% of all sublicensing income/milestones (note that this includes premium paid for any equity sold)
- Click here to download a PDF of the redacted license agreement. A few key notes: AIS rec'd $1m upfront; AIS is due a different % (presumably lower) of sublicense income for the China, Hong Kong, Malaysia, Indonesia; AIS gets full and free access to all BPAX data to use in other territories
- 3/14/11 4q2010: first time any real ROW discussion by mgmt. Stated that BPAX is doing comprehensive safety and efficacy trials, would need to to talk to EU officials (haven't yet), but think would "get a good hearing" w/ the BPAX data. Also of interest in south america (countries accept NDA straight-up). Has always been the plan to wait for BPAX to complete clinical work- AIS owns rights to that data. BPAX discussions could be w/ someone that wants worldwide rights and could negotiate those w/ AIS, that partner could then control ROW actions
- 4/5/11 at Needham- finally AIS has switched to call the NDA a 2012 event, which has been obvious for a while now. Off-label sales of testosterone for women about $200-300m annually in US
- 5/9/11 CC: Question re is AIS involved in BPAX Libigel business development discussions? We are in touch regarding future of Libigel, working with BPAX to ensure both companies get a good deal. Hope that any potential partner would want worldwide rights, so then AIS would have to be involved. Hope for significant partner fairly soon.