Antares Pharma (ATRS) - www.antarespharma.com
|
|
Company Financials
Proprietary financial modeling for AIS available upon request for Chimera Research Group subscribers.
Guidance and Outlook
- Cost of product sales has been around 50% in recent quarters
- Cash burn was significantly reduced from 2009 to 2010 and will be even lower in 2011 (3/15/11 Roth presentation)
- Company should be sustainably cash-flow profitable when the next product is launched
- Cowen 3/7/11: decreased cash burn upon completion of Anturol phase 3 trial, currently have 20 employees. Have "a lot more than one year's cash on hand"..."have a phenomenal relationship with Teva"...other internal projects behind MTX are in "concept" stage. 3/15/11 at Roth: "most likely more than 2 years cash on hand"
- As of 3/4/11, 21 employees, officers and directors hold 18% of fully diluted share count
- 3/14/11 4q2010 CC- 2011 Outlook and Milestones: increase total revenue y/y; announce vibex MTX data; Anturol approval and partner; advance Epi-pen and pipeline projects.
- 3/14/11 4q2010 CC- R&D costs inc'd ~$1m from 2009 to 2010. 2011 not giving precise guidance but Anturol trial spend done, about $1m scaleup costs will expense to R&D, about $2m for MTX- should see decrease overall
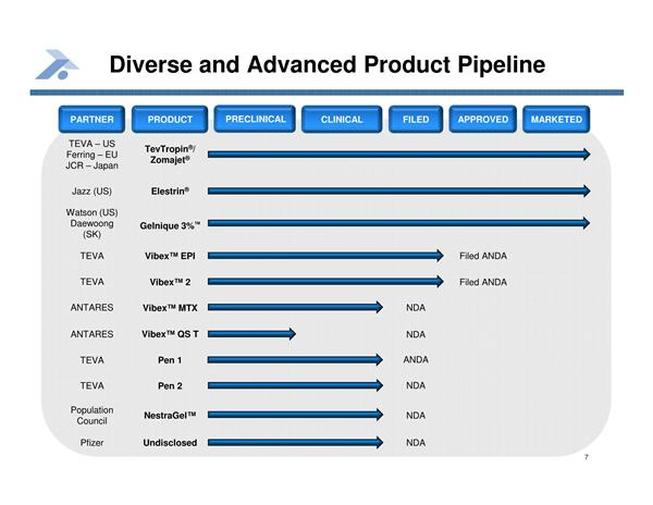
Antares Pharma Product Pipeline
|
Fundraising
- July 2009 raised $8.5m gross by selling 10.625m shares at $0.80 plus 4.25m warrants exercisable January 2010-January 2015 at $1.00- used to pay off debt
- September 2009 raised $2.9m net by selling 2.73m shares at $1.10 per share plus 1.09m warrants exercisable March 2010-March 2015 at $1.15
- Between Jan-Sept 2010, options and warrants for 1.8m shares were exercised, bringing in $2.0m cash
- 1q2011- about $5m worth of warrants were exercised at $1.50 (3/15/11 Roth: these were from a 2006 financing; 2010 10k: about 1/2 of these $1.50 warrants - another 3.5m shares - expired unexercised 3/2/11)
- FIrst 9 months of 2011 - 4.44m warrants exercised for $5.97m ($1.34/share average); 1.8m options exercised for $2.04m ($1.13/share average)
- 5/2011: sold 12.5m shares at $1.60 for $20m gross proceeds (no warrants)
- July 2012 - 4m warrants at $2.00 expire
FDA pathways for submission and Product approval
The distinctions between these pathways are important to keep in mind as AIS has or will utilize all three of these routes to approval for their product candidates
505(j) aka Abbreviated New Drug Application (ANDA)
505(j) aka Abbreviated New Drug Application (ANDA)
- Seeking generic approval of a product that is comparable to the branded, approval product that is already on the market (including formulation, dose, route of administration, and purity)
- Can be approved with designation to be automatically substitutable for the innovator product- ie, even though the doctor writes prescription for the innovator product, the pharmacy can give the generic drug instead
- Generally need to submit 3 month stability data, but no formal safety or efficacy data required
- "First to File" status can result in the generic company obtaining 6 months of exclusivity upon approval
- There is no "PDUFA date" like there is with NDAs, but the FDA did institute a policy of accelerating the review of high priority ANDAs in 2006. Instead of a "first-come, first-served" review policy, applications can go to the front of the line and get a goal of 6 month review time if it is the first generic application for a drug, if there is a shortage, or some other circumstances (click here for more info)
- Changes to dose form, strength, route of administration, or formulation
- Generally need 12 month stability data for new formulation
- A bridging study is performed to compare the systemic levels of the new product compared to approved product
- Sponsor may conduct some sort of safety or efficacy trials, but can also sometimes access the safety and efficacy data of the reference product
- Unlike ANDA pathway, these approval are not usually deemed substitutable
- This pathway is faster and less expensive than NDA pathway
- Approved product can be granted 3 years of Hatch-Waxman exclusivity if the sponsor conducted some clinical trials
- Unlike NDAs, approval can be held up due to patent protection of the already approved product
- New molecular entity
- Requires full clinical safety and efficacy studies