And based upon this idea, millions of dollars were spent on developing a unique sequencing-by-ligation method, with a unique method of library preparation and template preparation, involving small 'nanoballs' that adhere to microlithographically-etched patterned silicon substrates at high density. And with a sequencing services-only model (Complete Genomics, unlike Illumina $ILMN, Pacific Biosciences $PACB or Life Technologies $LIFE, does not sell any of their systems to researchers), they have a fee-for-service model with another unique dimension, which is the sequencing of only human whole genomes.
Only human whole genomes are sequenced - no human whole exomes, no mouse whole genomes, no plants for the agricultural segment, no RNA for expression experiments, no methylated DNA to look at epigenetics. Thus Complete Genomics, from its inception, looked at the next-generation sequencing market and was going to offer a single offering set to revolutionize medicine.
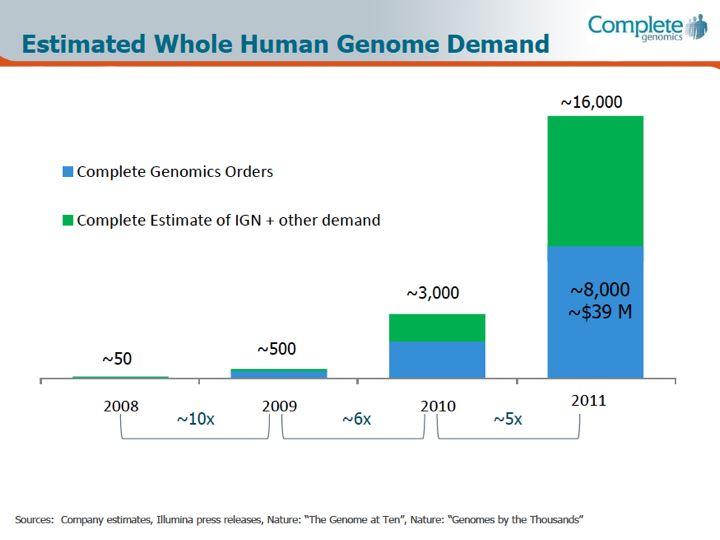
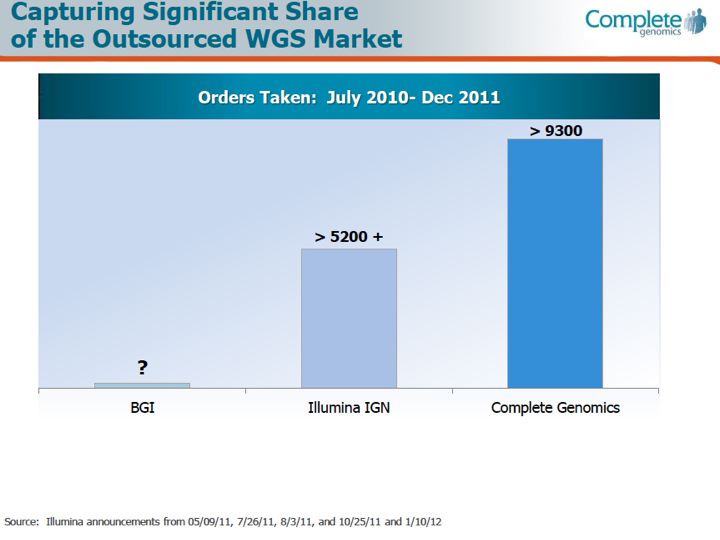
From what I've been able to gather (from various market research surveys), currently the segment of human Whole Genome Sequencing as a function of the overall market is on the order of about 15%. So by whatever metric one chooses for the overall size of the next-generation sequencing market as a whole (I use a $1B value for 2011, and at an estimated growth rate of about 20% puts the 2012 value of the overall NGS market at $1.2B), the whole-genome sequencing segment would represent about $150M of that. And of that $150M (doing some back-calculations and estimates here) $20M went to Complete Genomics in 2011, and perhaps $20M went to Illumina (these two have been locked into battle over the whole-genome sequencing services market space, and a 50/50 split may be just a guess, as Illumina does not break out their genome sequencing service business separately). Editors note: based on the March 2012 corporate slides reproduced below, GNOM appears to be capturing at least half of the market:
Looking back historically, the first individual whole genome, Craig Venter's, was first sequenced via CE Sanger sequencing only in 2007, followed shortly thereafter by the publication of James Watson's genome via Roche / 454. Only in the span of four years, going from 2 individuals to 40,000 is a truly remarkable achievement, a reflection of the pace of innovation and market-force driven competition so that the volumes of these samples are actually affordable, as well as the frontiers of genomics and genetics being expanded. Looking at costs, it was estimated to be 'several million dollars' for the Venter genome, and in the 'several hundred thousand dollars' for the Watson genome; now, as a service from Complete Genomics the price per sample is on the order of $5,000 or less depending upon the volume. (As a point of comparison, the list price of Illumina's reagents for a HiSeq 2000 is on the order of $3,300, but does not include the depreciation of the instrument - a significant expense on a $690,000 device - yearly maintenance, overhead, labor etc.)
On that note, assuming a 3-year straight depreciation schedule and including service over those three years, equates to about $23,000 per month, and given that a HiSeq 2000 could do about six human whole genome samples in 11 days, that's about 175 samples per year. Doing some math, the $3,300 in reagents on a fully-loaded HiSeq (assuming an 'uptime' of 90% utilization over the entire year), now magically turns into a $4,880 per sample price.
As I mentioned before, a Whole Genome Sequence compared to a Whole Exome Sequence is a debate that I've commented on before - there are pluses and minuses to argue for each side, and Complete Genomics only offers WGS. Yet for many prospective customers they rule Complete out as they simply do not make that offering, and prefer exomes as they can get more 'bang for the buck'.
What are the other barriers to the clinical application of WGS to drive incredible demand now? Manifold reasons - from federal regulation (i.e. FDA in the U.S.), to what a 'clinical genome' actually means in terms of data quality, to agreement in nomenclature between variant databases, to reimbursement issues (again a U.S.-specific situation). And in terms of its usefulness, at the Medical College of Wisconsin Liz Worthey said at a conference earlier in the year that of 13 human genomes sequenced (understand that these were selected for having an undiagnosed disease with a suspected genetic cause), only 3 of the 13 resulted in a definitive diagnosis. While it is certainly a nice addition (three pediatic patients and their families now know have a diagnosis), the 20%+ 'success rate' matches well with what Dr. William Gahl has said for the NIH Undiagnosed Diseases program. (He was on the US television show '60 Minutes' recently; the 12-minute interview is worth watching.)
So these efforts are to be heralded and celebrated - we are witnessing a revolution in health care, one undiagnosed case at a time. However, it does not follow that there is a successful service business to be built around that - Complete Genomics is buffeted by dropping sequencing costs (and when the $LIFE Ion Torrent Proton comes out, in particular the Proton II chip next spring, it will drop again), and a limited market segment (remember that 15% number), the true value of the company is its informatics pipeline. So don't be surprised should they decide to change their business model.
 RSS Feed
RSS Feed
