Navigate the Infinity Pharma $INFI Research Pages
Introduction - General corporate updates, financials, and resources
Saridegib (IPI-926) - Hedgehog inhibitor in multiple phase 2 cancer trials
Retaspimycin HCl (IPI-504) - HSP90 inhibitor in phase 2 for lung cancer
IPI-145 - PI3K inhibitor in phase 1 for oncology and inflammatory diseases
Pipeline - including IPI-940 pain drug candidate licensed to Purdue Pharma
Saridegib (IPI-926) - Hedgehog inhibitor in multiple phase 2 cancer trials
Retaspimycin HCl (IPI-504) - HSP90 inhibitor in phase 2 for lung cancer
IPI-145 - PI3K inhibitor in phase 1 for oncology and inflammatory diseases
Pipeline - including IPI-940 pain drug candidate licensed to Purdue Pharma
Infinity Pharmaceuticals (INFI)Website link: www.infi.com
Archive of all INFI related blog posts and articles: 2010 10k, 4q2010 results - Click here for my CC notes 4/15/11 BioCentury webcast - Click here for my notes. 1q11 results 5/10/11 - click for PR - click for CC notes 6/1/11 ASCO/pipeline webcast - click for my notes 6/8/11 Jefferies webcast - click for my notes 8/9/11 2q2011 earnings - click here for my notes. Note: this INFI page is completely up-to-date as of 9/5/2011 9/7/2011 Stifel Nicolaus webcast - click here for my notes. 9/12/11 Rodman and Renshaw webcast - click for notes. 10/21/11 BioCentury webcast - click for notes. 2/2012 BIO CEO conference - click for my notes. 3/2012 Citi Healthcare conference - click for notes. 3/2012 Cowen Healthcare conference - click for notes. 5/2012 ASCO preview and partner updates - click for blog. 5/2012 ASCO abstract - click for blog. 6/2012 Jefferies Healthcare conference - click for notes. Upcoming events:
|
"We are building a fully integrated sustainable biotech company"
|
Links
- Click here for archive of all blog posts related to INFI.
- 6/2011 Xconomy article "Infinity Dares to Think Big Against Pancreatic Cancer"
- 3/2011 Bolt International thought leader summary re hedgehog inhibitors for pancreatic cancer.
General Info
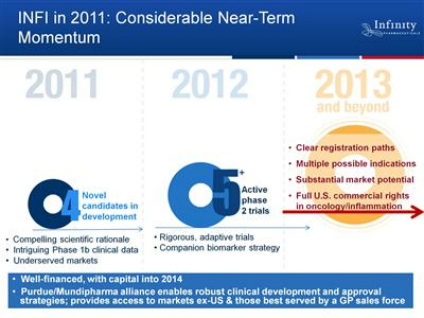
- 6/2011 webcast: in 2012 we will have 5 or more phase 2 trials underway. Use "rigorous adaptive trial designs"
- 8/2011 2q11 CC: Focus on unmet medical needs/difficult to treat disease: NSCLC, pancreatic cancer, chrondrosarcoma, myelofibrosis
- 6/2011 webcast: Expect to name new development candidate from discovery operations in 2011.
- 168 employees, 133 R&D (35% doctoral).
- 6/2011 CC: Added new CMO 11/2010 who has experience with Taxol (BMS), taxotere, tarceva, campath, yondelis (sarcoma)
- 6/2011 CC: Company added molecular pathology group- help translate scientific insights into clinic and vice versa. Company policy is that each trial has a molecular pathology /biomarker effort. Ask which patient is most likely to benefit?
Financials and Guidance
- March 2009 Purdue gave $50m line of credit -balloon note at prime, matures 2019 (non borrowed 12/31/10).
- 11/2008 Purdue bought 4m shares at $11.25 for $45m total.
- 1/2009 Purdue bought 2m shares at $15m for $30m plus 6m warrants at strokes from $15-40 (1m have expired and 5m outstanding 12/31/10: 2m expire 7/1/11 and are exercisable at range of $20-30 depending on when exercised. 3m purdue expire 7/2/12 that are exercisable at range $30-40 depending on time exercised..
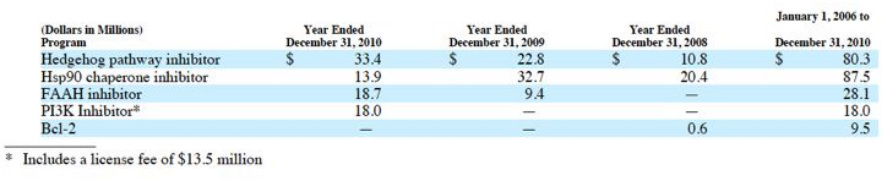
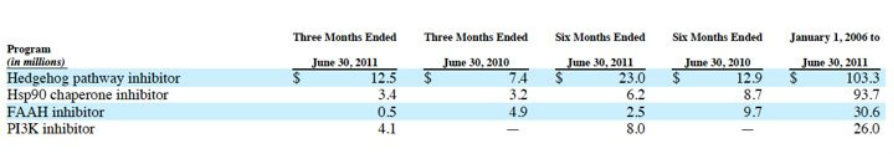
- R&D expenses $99.2m in 2010 ($67m of this was reimbursed), $77.9m in 2009 ($46.5m reimbursed), $47.5m in 2008 ($20.1m reimbursed).
- 2011 R&D expense reimbursement capped at $85m, transition expenses for FAAH not capped.
- 8/2011 2q11 CC: Now will draw $50m loan in 2011 and will have $110-120m cash at ye-2011.
- 8/2011 2q11 CC: reaffirm cash burn and net loss of $30-40m for 2011 (related to HSP90 program spending)
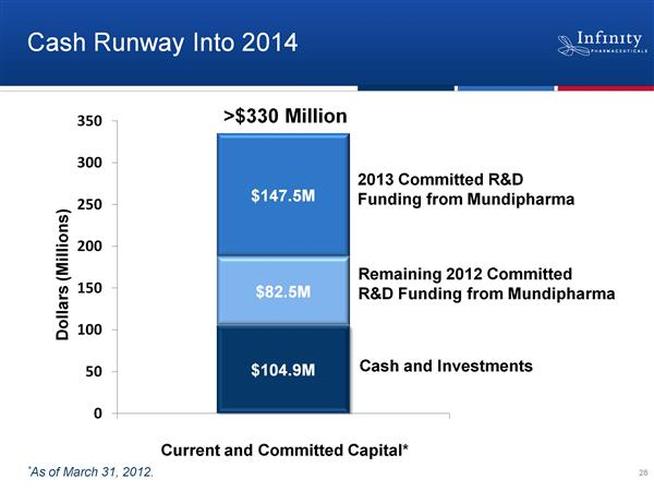
- Cash and committed research funding sufficient into 2014. Can drive programs thru value inflection points and generate "meaningful data" before raising cash (4/2011 webcast and 6/2011 CC)
Partnerships with Mundipharma and Purdue Pharma
- November 2008: agreements w/ Purdue (FAAH in US) and Mundipharma (ex-US rights to Hedgehog, FAAH, PI3K, and any discovery pgms during the funding period but excludes HSP90 pgm).
- Dec 2010, Mundipharma extended research period to 12/31/12 (one addl yr option remains).
- Mundi and Purdue will each owe 10-20% tiered royalties until patent expiration.
- INFI has exclusive control of discovery and early development (no joint steering committees).
- Mundipharma pays 100% r&d costs until "transition date"-the later of 12/31/13 or start of 1st p3 clinical trial of each such product candidate- then share costs equally.
- R&D budget was $50m 2009, $65m 2010, $85m 2011, and $110m 2012. Exceeded this amt at own expense in 2010 (PI3K license) and will again in 2011.
- Mundi has opt-out clause every Nov (but would have to pay full funding for addl yr after that) for each pgm except Hedgehog pgm which is now is milestone-driven opt-out clause (would owe 1-5% royalty to mundi depending on when opt out [after transition date=5%]).
- Each party can opt out at transition date (but have to pay 50% of costs for one yr).
- INFI retains US rights to all pgms except FAAH. Mundi can gain ex-US rights to any new inlicensed pgm (if tox studies started) by reimbursing up to 60% of upfront fee and funding r&d as for other pgms (no royalties pd b/t parties). Earlier stage assets (ie, PI3K) are just added to exisitng deal and keep same budget.
- INFI owes 5% royalty until repay devel funding for that pgm thru transition date, then owe 1-3% of US sales.
- Partners can terminate on change in control of INFI
- See above for related equity investment by Purdue Pharma