Navigate the Infinity Pharma $INFI Research Pages
Introduction - General corporate updates, financials, and resources
Saridegib (IPI-926) - Hedgehog inhibitor in multiple phase 2 cancer trials
Retaspimycin HCl (IPI-504) - HSP90 inhibitor in phase 2 for lung cancer
IPI-145 - PI3K inhibitor in phase 1 for oncology and inflammatory diseases
Pipeline - including IPI-940 pain drug candidate licensed to Purdue Pharma
Saridegib (IPI-926) - Hedgehog inhibitor in multiple phase 2 cancer trials
Retaspimycin HCl (IPI-504) - HSP90 inhibitor in phase 2 for lung cancer
IPI-145 - PI3K inhibitor in phase 1 for oncology and inflammatory diseases
Pipeline - including IPI-940 pain drug candidate licensed to Purdue Pharma
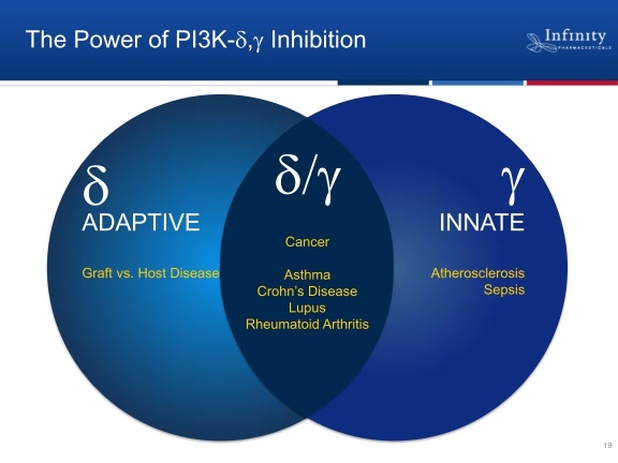
IPI-145 PI3K inhibitor
- oral, small molecule, dual inhibitor of delta and gamma isoforms (restricted to immune system cells).
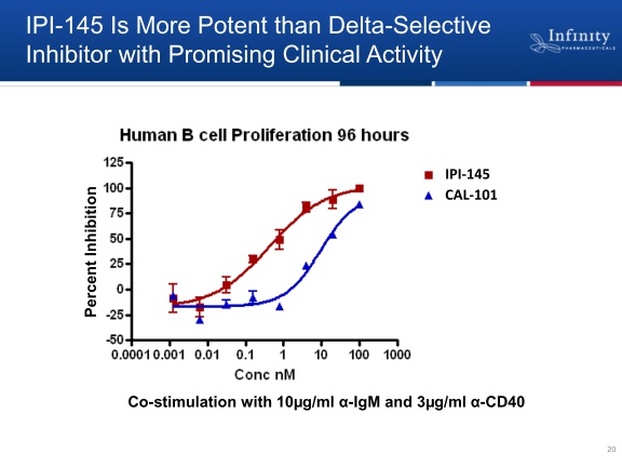
- 4/2011 webcast: Calistoga's delta specific PI3k inhibitor showed "stunning data." IPI-145 is 10-20x more potent for delta isoform and adds gamma inhibition too. Lots of crosstalk between these isoforms. Gamma isoform plays role in trafficking to site of inflammation
- In-licensed pgm and portfolio of inhibitors (global rights) in July 2010 from Intellikine ($13.5m upfront, plus 2 yrs res funding, up to $25m devel and $450m approval/commercialization milestones for 2 candidates, tiered royalties from single digits to mid teens, Intel can pay option fee after phase 2 to copromote if for an oncology candidate).
- Click for for the full redacted license agreement filed to the SEC
- Activity in preclinical models of inflammation. 6/2011 CC also mentioned interest for oncology indications.
- Mundipharma has rights outside US
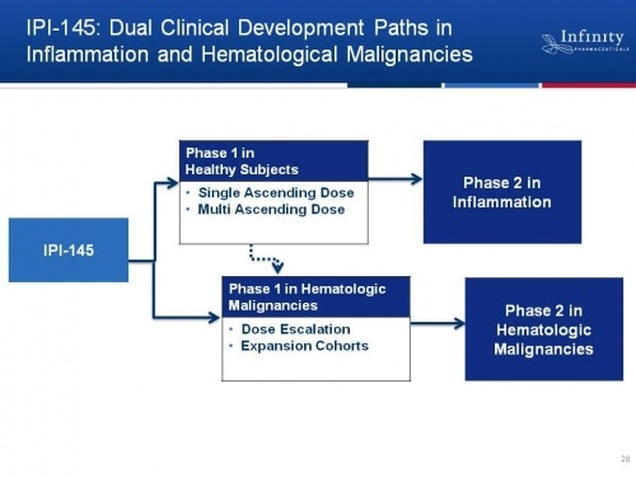
- 8/2011 2q11 CC: Recently filed US FDA IND and CTA in EU, will provide additional phase 1 details later this year (consistent with prior timeline guidance).
- See below for summary of competition from 2010 10k: