Navigate the Infinity Pharma $INFI Research Pages
Introduction - General corporate updates, financials, and resources
Saridegib (IPI-926) - Hedgehog inhibitor in multiple phase 2 cancer trials
Retaspimycin HCl (IPI-504) - HSP90 inhibitor in phase 2 for lung cancer
IPI-145 - PI3K inhibitor in phase 1 for oncology and inflammatory diseases
Pipeline - including IPI-940 pain drug candidate licensed to Purdue Pharma
Saridegib (IPI-926) - Hedgehog inhibitor in multiple phase 2 cancer trials
Retaspimycin HCl (IPI-504) - HSP90 inhibitor in phase 2 for lung cancer
IPI-145 - PI3K inhibitor in phase 1 for oncology and inflammatory diseases
Pipeline - including IPI-940 pain drug candidate licensed to Purdue Pharma
2012 Update: the INFI Saridegib clinical program has been terminated
Saridegib (IPI-926) Hedgehog Pathway inhibitor - Introduction
- Binds to and inhibits Smoothened receptor which is involved in the malignant activation of the Hedgehog pathway. "Inhibiting Smoothened with IPI-926 is believed to deplete the dense, fibrous stroma surrounding pancreatic tumors, leading to increased vascularity and facilitating the delivery of chemotherapy to the tumor."
- Foundation for this program was results in unique transgenic mouse model of pancreatic cancer that much better represents human disease vs existing xenografts. There is a dense fibrotic shell around pancreatic tumors, combination with IPI-926 allowed Gemzar to better penetrate tumor and doubled survival (4/2011 webcast; featured in Science, 21 May 2009)
- Click here for PDF file of info about pancreatic cancer and IPI-926. Pancreatic cancer: 35-40k annual case in US, roughly same number of deaths. Median PFS ~3 months and median overall survival <6 months. Gemcitabine is standard of care, based on 6 week improvement in OS.
- 6/2011 webcast had slide listing numerous pancreatic cancer trial failures- many have been 1x/2x/3x combined chemotherapy agents. Due to strong tumor stroma around these tumors, chemo doesn't penetrate thru the "dense shell". IPI-926 breaks down this shell
- Oral drug with plasma half-life, natural plant product required in production process.
- 6 patents on composition of matter (COM; and related compounds), formulation, etc expire 2025-2028.
- Was partnered w/ Medimmune, rights returned 12/2007 (no royalty obligation)
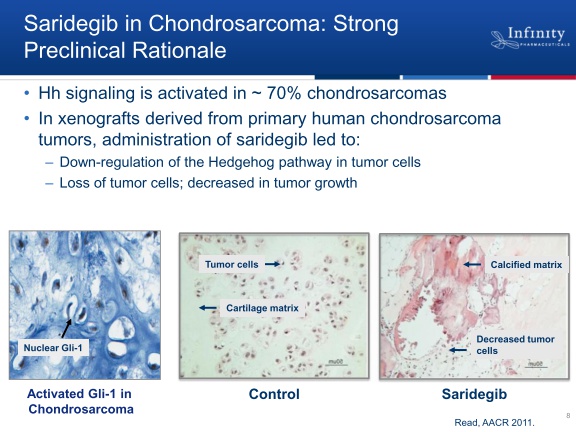
- 6/2011 granted orphan drug status for chondrosarcoma in EU
- Licensed to Mundipharma outside the US (see above for deal terms).
- Mundi must make next funding commitment by 30 days after end of p2 mtg for panc cancer or 11/30/13, whichever is first. If opt out, must pay $23.65m immed and up to $23.65m expenses for pgm in 2012 to complete any ongoing p2 trials. If continue at that time, return to Nov opt-out schedule.
- 2011 goals: Complete p2 pancreatic cancer combo trial enrollment by ye2011, initiate additional p2 development activity including broad investigator-sponsored trials in 2h2011. 6/2011 CC: including at least one more company-sponsored trial in 2011.
- Positive phase 2 survival data in advanced pancreatic cancer would allow aggressive development for earlier stage disease and in combo with other chemo agents (6/2011 Xconomy article)
- Competitors include: GDC-0449 (Genentech and Curis - click for more info), BMS833923 (BMY and EXEL), LDE225 and LEQ506 (NVS), PF-04449913 (PFE), TAK441 (Takeda/MLNM).
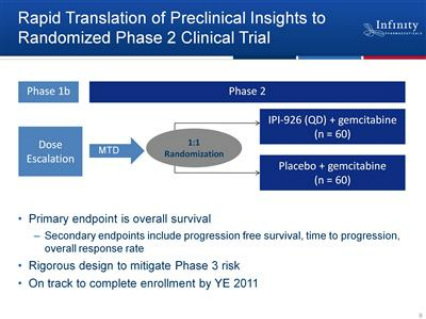
IPI-926 - Clinical Trials
- 2/2011 initiated randomized phase 2 trial in first-line metastatic pancreatic cancer
- --Expect to complete enrollment be year-end 2011 (6/2011 webcast: "enrolling beautifully") and have data by late 2012 (6/2011 Xconomy article)
- --No molecular stratification in this trial, but profiling patients for future info (6/2011 CC)
--Interim analysis: 3 of first 9 pts showed >50% shrinkage. Side effects included nausea, fatigue, and elevated liver enzymes
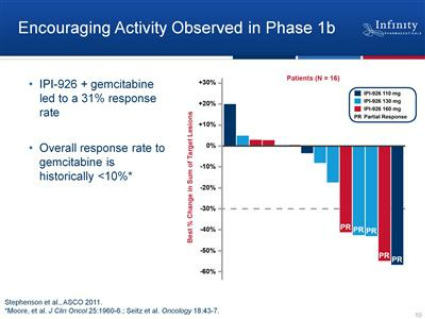
--ASCO 6/2011 presented data from phase 1b portion of trial: (Click here for PR - Click here for abstract) 5 of 16 pts experienced partial response (Gemcitabine historal response rate <10%). Doses ranged from 110 to 160 mg (at least 1 PR at each of 3 doses, doesn't really appear to be a dose response, but the doses are in a pretty narrow range). Median PFS > 5.5 months (not yet reached), OS ~6 months. No PK drug-drug interaction observed. No grade 4-5 adverse events, most common AEs were grade 1-2 fatigue, nausea, liver transaminase elevation
- --8/2011 2q11 CC: Continue to follow patients, will report again after mature overall survival data available
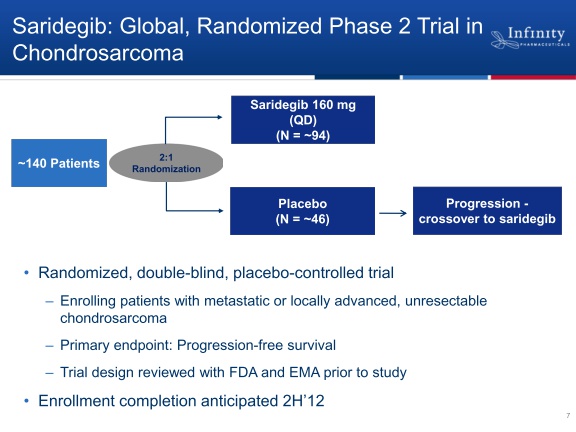
- 2/2011 initiated randomized single agent p2 in metastatic or locally-advanced inoperaable chrondrosarcoma
--Placebo-controlled (2:1 randomization), primary endpoint is PFS, placebo pts can crossover after progression. Trial design reviewed with FDA and EMA. Click here for Clinicaltrials.gov listing.
--No comment has ever been made about enrollment/data timeline. Database listing has estimated completion 2015.
- Phase 1 in advanced/metastatic solid tumors (including basal cell carcinoma)
--Preliminary data October 2010 (European Society of Medical Oncology, 60 pts including 24 w/ basal cell carcinoma (BCC; 17 naïve to hedgehog inhibitors), 4 PR among BCC pts, 3 SD >6mo among others)
--Follow-up data ASCO 6/2011 (Click here for abstract - Click to download poster): 79 patients, including 31 with BCC, 27 pts still on study. No grade 4-5 adverse events (grade 3 DLTs were liver enzyme related -33% at highest doses), no patient deaths on study. 6 clinical or radiographic PR is hedgehog inhibitor naive BCC patients (27%) I'm not sure why they haven't proceeded with another trial in this indication if they feel this data is strong
- 4/2011 initiated Erbitux (cetuximab) combo head and neck cancer investigator-sponsored trial (IST)
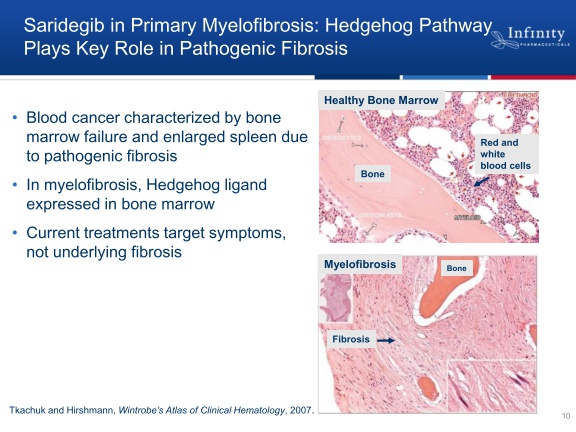
- 6/2011 announced plans for phase 2 trial in myelofibrosis to initiate 3q2011
--Primary endpoint of hematological response rate (international working group standard.
--8/2011 2q11 CC: Other treatment options like JAK2 inhibitors only treat symptoms and don't target malignant fibrosis.
- 8/2011 announced FOLFIRINOX combo pancreatic cancer IST: (Click here for ClinicalTrials.gov listing)