Navigate the Ligand Pharmaceuticals, Inc. ($LGND) Stock Research Pages
- Introduction (valuation, financials, ownership, guidance, helpful links)
- Promacta (eltrombopag) (key asset licensed to GSK - platelet booster for many thrombocytopenia conditions)
- Kyprolis (carfilzomib) (key asset licensed to Amgen - developed by ONXX for multiple myeloma)
- Other Marketed Products (description of assets currently generating royalty revenue)
- Late-stage Pipeline Assets (generally partnered and developed at little to no cost to LGND and represent future royalty sources)
- Early-stage Development Programs (internal and partnered, future "shots on goal" or out-licensing opportunities)
- Acquisitions (Cydex, Metabasis ($MBRX), Pharmacopeia ($PCOP), Neurogen ($NRGN) - complete terms and details)
- Captisol-Cydex (additional info on the formulation technology acquired in 2011 and details on partnerships)
- Discontinued Programs (with >90 biotech and pharma assets in development, not everything works out!)
Ligand LGND Captisol Partnerships (Non-marketed products)
Carbamazepine
Status: Phase 3 -appears on Lundbeck website by LGND never mentions Source: Cydex
- Developed by Lundbeck.
- Captisol-enabled novel IV formulation of commonly-used sodium channel blocker for epilepsy.
- For hospitalized/temp use when can't take oral.
- In p3 trial (NCT01128959) to show safety and bioequivalnce to higher oral doses of 13x 70% dose as15 min infusions over 4 days. 105 pts, started 6/2010, est completion Sept 2011.
- previously completed p1 trial (NCT01079351) w/ 98 pts to compare to oral
Undisclosed Merck Program
- 6/2011: Captisol clinical and commercial supply deal for undisclosed Merck drug candidate - click for PR.
- 6/2011 Analyst Day: program is currently in phase 2, potential 2013 launch
- Merck will make milestone payments for regulatory achievements and will purchase Captisol from LGND, but will not owe royalties. Deal will expire 12/31/2015 unless extended [MRK has one 5 yr option] or terminated earlier. Click here for 8k with details. Full redacted agreement will be filed with LGND 2q2011 10q.
- If approved, program would represent multiple metric tons per year of captisol material sales
- "Merck used Captisol to reformulate a drug in their portfolio and performed initial proof of concept under a research use agreement"
SAGE Therapeutics Platform Captisol deal
- 10/2011 license agreement with SAGE, a newly formed company focusing on CNS therapeutics
- SAGE was launched with $35m Series A by Third Rock Ventures - and will focus on allosteric receptor modulators - click here for Xconomy feature.
Rib-X
Phase 2 antibiotic entering phase 3 in 2012. Newly disclosed due to IPO filing
Cydex: Captisol info
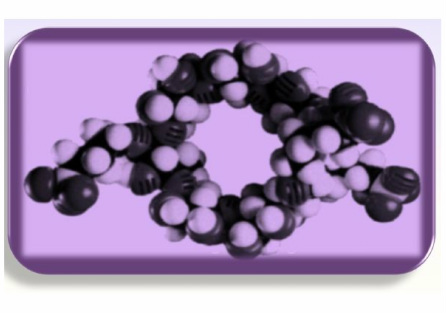
- Captisol is a chemically modified cyclodextrin that improves drug efficacy, solubility, stability, patent life.
- captisol is a "doughnut"- hydrophillic outside and hydrophobic inside (see figure below), 1/2 of API are hydrophobic- jump from one captisol to another in solution, but diluted in vivo and reach target
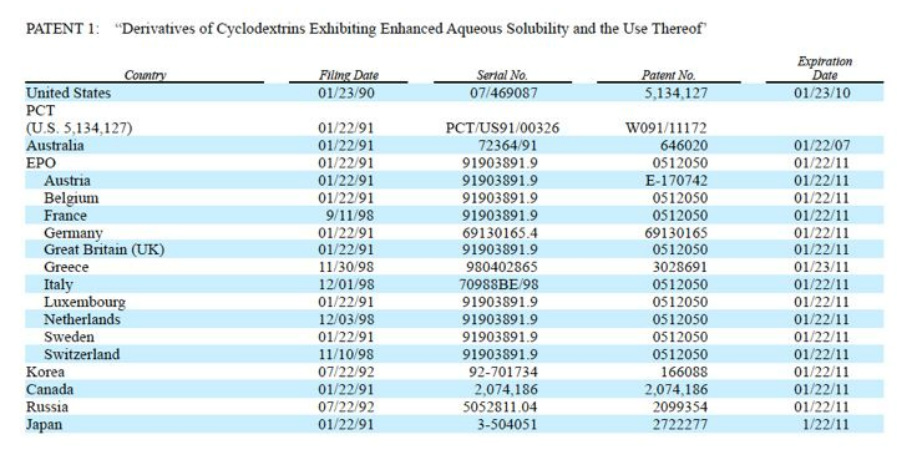
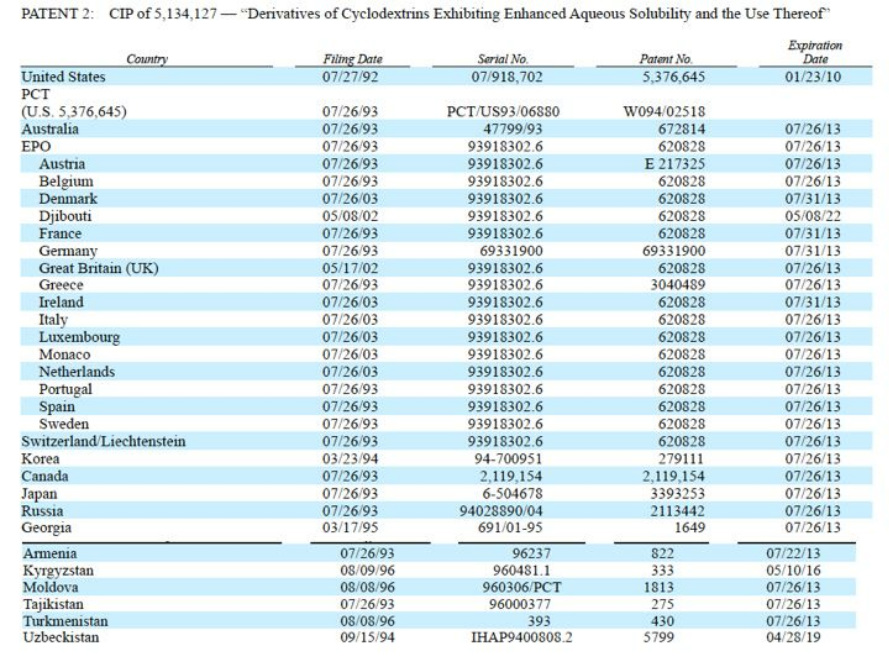
- US COM expired 2010, ROW exp 2011-2013 (one 2016).
- Agglomerated form "morphology" patent expires 2025 (2010 10k and 3/31/11 10q)
- 10/2011: captisol is cover by 2 orange book listable patents - benefit to partners
- 6 pending combo patents with APIs...one combo patent for an undisclosed early-stage program expires 3/2022 (2010 10k and 1q2011 10q)
- High purity form patent expires 2029 (2010 10k)...all partners except PFE are using this and this will dictate royalty terms
- Extensive drug master file (DMF) is key to brand value that can be used by partners (2/15/11 webcast: saves collaborators 3-4 yrs and $10-20m in expenses). May be used for LGND internal pgms (ie, topical JAK3).
- Exclusive manufacturing deal w/ Hovione thru 2019 (amended to reduce annual commitment but extend term). Total commitment of at least $15m over term of deal. 10/2011 - 50 metric ton capacity and ability to significantly expand this.
- Guidance for new licensing deal 2h2011. 5 internal pgms to eventually partner- S1 said move from screening to NDA in 18-36 months.
- Website- over 1000 evaluation agreements.
- 10/2011: >20 captisol-enabled drugs in development
- Undisclosed large pharma partnerships: currently get material sales for these products- includes injectables, topical, oral, some in late stage development. >20 current Use agreements (started offering in 2004, preclinical and p1- contract fees and material sales). >10 current P2 and beyond: License and Supply agreements (upfront, milestones, royalties). Website: partners include PFE, BMY, Daiichi, Allergan, AZN, GSK. S1 (2008) lists Critical Therapeutics, Daiichi Asubio, Proteolix, Sunesis, Taisho and TargeGen as partners
- All products are formulations of FDA-approved APIs, so should be 505b2 pathway- show bioequivalence only (FDA can ask for more trials).
- 80 of 200 API screened have characteristics suggesting improvement with captisol.
- PFE option deal 12/1993, exercised in 6/1996, all chemistry, toxicology, and pharmacology data go in DMF. 2010 10k: sounds like PFE doesn't actually by Captisol material from LGND (?)
- "Aware of third party patent related to Captisol" We have rights under two patent families, which include U.S. 5,134,127 and U.S. 5,376,645 and corresponding foreign patents and patent applications
Captisol - Intellectual Property (in addition to Drug Master File trade secrets)
Use these links to navigate to additional LGND stock research pages:
- Introduction (valuation, financials, ownership, guidance, helpful links)
- Marketed Products (description of assets currently generating royalty revenue)
- Promacta (key asset licensed to GSK - platelet booster for many thrombocytopenia conditions)
- Late-stage Pipeline Assets (Generally partnered and developed at no cost to LGND, represent future royalty sources)
- KYPROLIS-carfilzomib Oncology drug licensed to ONXX for multiple myeloma - July 2012 PDUFA date
- Early-stage Development Programs (internal and partnered, future "shots on goal" or out-licensing opportunities)
- Acquisitions (Cydex, Metabasis $MBRX, Pharmacopeia $PCOP, Neurogen $NRGN - complete terms and details)
- Captisol-Cydex - Additional info on the formulation technology acquired in 2011 and details on partnerships
- Discontinued Programs (with >50 biotech and pharma assets at any time, not everything works out!)