Navigate the Ligand Pharmaceuticals, Inc. ($LGND) Stock Research Pages
- Introduction (valuation, financials, ownership, guidance, helpful links)
- Promacta (eltrombopag) (key asset licensed to GSK - platelet booster for many thrombocytopenia conditions)
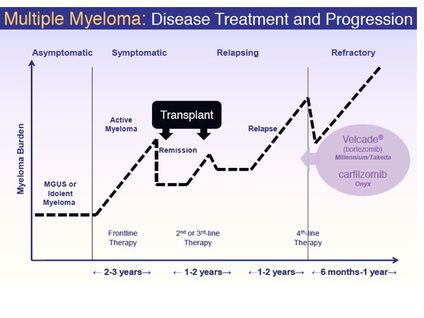
- Kyprolis (carfilzomib) (key asset licensed to Amgen - developed by ONXX for multiple myeloma)
- Other Marketed Products (description of assets currently generating royalty revenue)
- Late-stage Pipeline Assets (generally partnered and developed at little to no cost to LGND and represent future royalty sources)
- Early-stage Development Programs (internal and partnered, future "shots on goal" or out-licensing opportunities)
- Acquisitions (Cydex, Metabasis ($MBRX), Pharmacopeia ($PCOP), Neurogen ($NRGN) - complete terms and details)
- Captisol-Cydex (additional info on the formulation technology acquired in 2011 and details on partnerships)
- Discontinued Programs (with >90 biotech and pharma assets in development, not everything works out!)
Ligand $LGND Late-stage Clinical Assets
Please note indications of development status: Green (advancing), Red (discontinued/abandoned), Purple (uncertain)
Fablyn
Status: Approved in EU, PFE not developing Source: internal Ligand
- licensed to PFE for osteoporosis (3% flat royalty- already sold additional 3% stream for first 10 yrs)
- Member of the selective estrogen receptor modulator (SERM) class
- 11/2010 Bloomberg article (Click for link): PFE-sponsored study showed decreased risk of breast cancer (79% less) and fractures, but increased risk of blood clots. This new report is 5 year data from same study that PFE submitted 3 year data from in 2007
- 9/2005 rejected by FDA for osteoporosis, 1/2006 rejected by FDA for vaginal atrophy
- 1/2009 US CRL (9-3 positive FDA advisory committee that benefits outweigh risks in 9/2008)...due to increase # of all-cause deaths (38% higher at low 0.25mg dose)
- 2/2009 EU approval
- 5/2010: PFE withdrew NDA and is exploring options for out-license or sale of this asset
- 2q2011: PFE returned rights to LGND
- 10/2011: LGND licensed worldwide rights to Chiva for $4m upfront (due by 6/1/2012) plus milestones and royalties. Safety and efficacy data from over 10,000 patients in hand.
Aprela
Status: Awaiting NDA filing Source: internal Ligand
In SMART-5, endometrial safety was evaluated by the incidence of endometrial hyperplasia as measured by endometrial biopsy at one year. Results indicate that BZA 20 mg/CE 0.45 mg and BZA 20 mg/CE 0.625 mg demonstrated less than one percent incidence of endometrial hyperplasia over 12 months. Breast tenderness associated with BZA/CE was similar to that of placebo. Bleeding and spotting with BZA/CE was similar to placebo.
The effects of BZA/CE on breast density over 12 months were also evaluated in 940 postmenopausal women with a uterus. Subjects were randomized to receive two doses of BZA/CE [BZA 20 mg/CE 0.45 (n=231), BZA 20 mg/CE 0.625 mg (n=247)], BZA 20 mg (n=122) and CE 0.45 mg/MPA 1.5 mg (n=100), or placebo (n=240). Results of this analysis indicate that women treated with BZA 20 mg/CE 0.45 or BZA 20 mg/CE 0.625 mg for 12 months showed no statistically significant differences in breast density compared with those treated with placebo.
In another SMART-5 sub-study, the effects of BZA/CE on sleep parameters and health-related quality of life (HR-QoL) were evaluated in 459 postmenopausal non-hysterectomized women with bothersome vasomotor symptoms and sleep problems at baseline. Subjects were randomized to BZA 20 mg/CE 0.45 (n=115) or BZA 20 mg/CE 0.625 mg (n=123), BZA 20 mg (n=49), CE 0.45 mg/MPA 1.5 mg (n=56), or placebo (n=116) daily for 12 months. Using a validated quality of life measurement tool, women treated with BZA/CE had statistically significant improvement in sleep parameters and health-related quality of life at one year compared with placebo."
- Licensed to PFE
- Consists of viviant in combination w/ estrogen for menopause
- two positive p3 (80% reduced hot flashes)
- 7/2012 - announced EMA regulatory submission has been accepted, decision in 2013
- 1/2011: Expect filing of NDA in 2011 (timeline has slipped)
- 2/2011 BIO CEO summary: status listed as "NDA/BLA filed or in process"
- 6/2011 Analyst Day: listed as possible 2013 launch
- 11/2011 - Pfizer presented 1 year safety and efficacy data from SMART-5 phase 3 study at the NAMS meeting"
In SMART-5, endometrial safety was evaluated by the incidence of endometrial hyperplasia as measured by endometrial biopsy at one year. Results indicate that BZA 20 mg/CE 0.45 mg and BZA 20 mg/CE 0.625 mg demonstrated less than one percent incidence of endometrial hyperplasia over 12 months. Breast tenderness associated with BZA/CE was similar to that of placebo. Bleeding and spotting with BZA/CE was similar to placebo.
The effects of BZA/CE on breast density over 12 months were also evaluated in 940 postmenopausal women with a uterus. Subjects were randomized to receive two doses of BZA/CE [BZA 20 mg/CE 0.45 (n=231), BZA 20 mg/CE 0.625 mg (n=247)], BZA 20 mg (n=122) and CE 0.45 mg/MPA 1.5 mg (n=100), or placebo (n=240). Results of this analysis indicate that women treated with BZA 20 mg/CE 0.45 or BZA 20 mg/CE 0.625 mg for 12 months showed no statistically significant differences in breast density compared with those treated with placebo.
In another SMART-5 sub-study, the effects of BZA/CE on sleep parameters and health-related quality of life (HR-QoL) were evaluated in 459 postmenopausal non-hysterectomized women with bothersome vasomotor symptoms and sleep problems at baseline. Subjects were randomized to BZA 20 mg/CE 0.45 (n=115) or BZA 20 mg/CE 0.625 mg (n=123), BZA 20 mg (n=49), CE 0.45 mg/MPA 1.5 mg (n=56), or placebo (n=116) daily for 12 months. Using a validated quality of life measurement tool, women treated with BZA/CE had statistically significant improvement in sleep parameters and health-related quality of life at one year compared with placebo."
IV Clopidogrel
Status: Pivotal trial initiation pending Source: Cydex
- Same active ingredient as oral drug Plavix from $BMS $SNY is oral, water insoluble so there previously was no IV or oral liquid
- LGND version is Captisol-enabled IV formulation (blood levels 15 min vs 2-4 hrs- now doctors must wait before starting stenting procedure, perhaps achieve same results with lower dose).
- Previously partnered by Prism for atherosclerosis/prevention of clot formation, returned 10/2010.
- 6/2011: licensed to The Medicines Co $MDCO for $1.75m upfront payment, up to $22m in milestones, and up to double digit royalties. [this deal had been in the works and was specifically mentioned in the 8k related to the Cydex acquisition]. LGND will supply Captisol for clinical trials and if approved, will be the exclusive supplier of commercial grade Captisol. Click here for PR. This deal fits with MDCO's stated (4/27/11) strategy of targeted late-stage acquisitions within established areas of expertise and geography.
- Cydex CVR holders will receive 50% of upfront fee ($875k) and 50% of milestones after subtracting up to $5m owed to former partner Prism. Click here for 8k with details.
- MDCO has proposed an "efficient 505(b)(2) development plan"..this drug is highly complementary to their existing portfolio of acute and intensive care products
- MDCO slide (Jefferies 6/7/11): "complements global chest pain pathway"; "significant medical need for platelet inhibitor in NPO pts"; PK/PD study planned; 3 year data exclusivity anticipated plus additional patents
- estimated 505b2 NDA filing 3q2012
- BMS/SNY patent upheld thru 11/2011
Melphalan
Status: Phase 2a ongoing Source: Cydex
- Branded product w/ same active ingredient is Alkaran (launched in 1993 by GSK...but developed by Celgene?) is injectable, palliative therapy for those who can't use oral meds.
- 6/2011 Analyst Day: $85m sales in last 12 months, multiple myeloma (MM) indications is majority of use but is currently off-label and has limited 1) stability (product is two vials that are mixed together and must be used within 60 minutes); 2) absolute daily dosing; 3) infusion duration (again, 60 min stability); 4) safety (potential adverse reactions to propylene glycol co-solvent)
- LGND drug is Captisol-enabled high dose IV formulation (comes as single freeze-dried vial, use within 24 hrs after reconstitution with normal saline, no propylene glycol, administration flexibility).
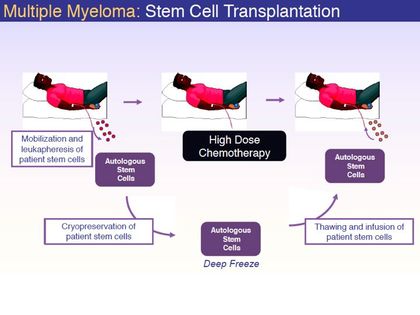
- Unpartnered program being developed for "high dose conditioning treatment prior to hematopoietic progenitor (stem) cell transplantation" for MM- 50,000 pts in US, 20,000 diagnosed yearly
- Patent filed 5/2009
- Received FDA orphan drug designation 11/2008 (grants 7 years market exclusivity and PDUFA fee waiver), IND 8/2009.
- 2/2010 initiated p2a (NCT00925782) for MM pts undergoing autologous stem cell transplant (24 pts, crossover). 6/2011: enrollment is complete, full data expected 4q2011
- 6/2011 presented interim p2a PK data at ASCO. Click here to download a copy of the poster. Key findings:
- All patients achieved myeloablation followed by successful engraftment.
- Except for expected grade 2-3 toxicities related to high-dose melphalan, no additional toxicities were reported.
- PK analyses revealed that the new propylene glycol-free melphalan met the requirements for bioequivalence to Alkeran.
- Captisol-enabled melphalan demonstrated a marginally greater melphalan systemic exposure (~110%) than was realized from Alkeran.
- Based on these preliminary results from the Phase II trial, the follow-on pivotal study will utilize a dosing regimen that appears to be comparable to Alkeran. This study will be designed to expose patients exclusively to the propylene glycol-free formulation and will further establish safety and efficacy measures for the new product.
- 6/2011: announced plans to continue development for 505b2 FDA filing anticipated in mid-2013. Pivotal trial will start early 2012 and enroll 60 pts. Click here for PR.
- 11/2011 - announced positive phase 2a trial results.
- This was one of several internal programs featured at 6/2011 Analyst Day. Reiterated ~2 yr timeline to NDA. May launch alone. Goals: safely achieve higher dose intensity to perhaps improve response rates
- 2008 branded sales of $81m, including off-label for conditioning prior to stem cell transplant (no plans to seek this higher dose indication).
BMS582949 (aka PS540446)
Status: Phase 2 Source: Pharmacopeia
- oral p38 kinase inhibitor (inhibits activity and activation of)
- licensed to Bristol Myers Squibb $BMY
- 4/2009 completed p2 in psoriasis and 9/2009 in RA, 4q2010: p2 ongoing in atherosclerosis.
- 10/2010: all three indications still listed on LGND website
- 11/2010 RA POC data presented (p2 120 pts mtx+/- 300 mg daily 582949 12 wks tx, ACR20 53% vs 33%, ACR50 15% vs 10%, decrease seen in CRP too).
- 4q2010: New trials to start 2011 (1/2011 said "in near term")
Dinaciclib (MK-7965)
Status: Phase 2 Source: Pharmacopeia
- CDK inhibitor, licensed to Merck
- 11/4/2011 MRK pipeline shown in phase 2 - not listed as filing in 2014 or sooner
- 8/2011 received orphan drug designation for CLL (link)
- phase 2 cancer trials ongoing in:
- advanced breast cancer
- AML/ALL (50mg [can inc to 70 cycle 2] infusion every 3 weeks, 20x AML randomized vs gentuzamab [crossover if no response], 6x ALL single arm)- data 12/10 ASH no objective responses but activity in 60%, folicular/mantle cell/bcell lymphona (12/10 ASH p1 data, 16 pts, 12 mg/m2, 1 PR)
- CLL (12/10 ASH updated CLL p1 data-weekly 3 of 4 weeks, was expansion of solid tumor p1, 33 pts, 16 at MTD of 14 mg, 8 PR of 23, 10 not yet evaluable)
- melanoma
- multiple myeloma
- 6/2010: trials will complete late 2010 thru 2012.
- 1/2011 "p2 data 2h2011". 4q2010 "complete various trials 2011 and 2012"
- 11/2011 listed in MRK R&D presentation as program with early clinical biomarker - simply said "histopathology"
- 2012 announced plans for phase 3 trial in CLL
CC-930 (fka PS873266)
Status: Phase 2 Source: Pharmacopeia
- JNK inhibitor licensed to CELG for fibrotic and inflammatory diseases
- phase 1 1q2008, 2% flat royalties.
- Ye2009: multi ascending dose done, this compound being advanced instead of the former lead pgm.
- 1/2011 p2 initiated in IPF (no mention of milestone received)
- 9/2011: US FDA orphan drug designation granted for IPF
- 10/2011 EU orphan drug designation granted for IPF
Pradefovir
Status: Phase 2 Source: Metabasis
- HepDirect PMEA prodrug (same metabolite as approved Hepsera) for HBV
- "strong activity" seen in phase 2 (11 trials >400 pts total), returned by Schering-Plough (now $MRK) due to preclinical toxicity issue.
- 10/2010 seeking to out-license, not in active development.
- 1/2011 licensed to Chiva for devel in China for HBV (8% of pop infected and gorwing, 30m need tx)
Tanaproget (aka NSP-989)
Status: Phase 2 (?) Source: internal Ligand
- tissue-selective, non-steroidal contraceptive progesterone receptor agonist for contraception w/ improved side effect profile.
- From legacy WYE partnership (earned milestone for entering p2 2003).
- 12/2010: PFE sublicensed to undisclosed multinational pharma, LGND received $1m upfront.
MB7803
Status: Phase 2a trial complete Source: Metabasis
- Fructose bisphosphatase (FBP) inhibitor - novel mechanism of action for diabetes
- Cinical POC data in hand
- Drug was discussed on 1q2008 MBRX CC (Click here to download): 1q2008 completed 28 day phase 2a trial in type 2 diabetes. Met primary endpoint of reducing fasting plasma glucose levels, 94% completion rate (how many patients?). Plan data presentation Barcelona 10/2008 at World Congress on Controversies to Consensus in Diabetes, Obesity and Hypertension and at the time planned p2b trial in 2009 in collaboration with a partner, also planned p1 combo w/ metformin
- Never seen any mention except at 6/2011 Analyst Day