Navigate the Ligand Pharmaceuticals, Inc. ($LGND) Stock Research Pages
- Introduction (valuation, financials, ownership, guidance, helpful links)
- Promacta (eltrombopag) (key asset licensed to GSK - platelet booster for many thrombocytopenia conditions)
- Kyprolis (carfilzomib) (key asset licensed to Amgen - developed by ONXX for multiple myeloma)
- Other Marketed Products (description of assets currently generating royalty revenue)
- Late-stage Pipeline Assets (generally partnered and developed at little to no cost to LGND and represent future royalty sources)
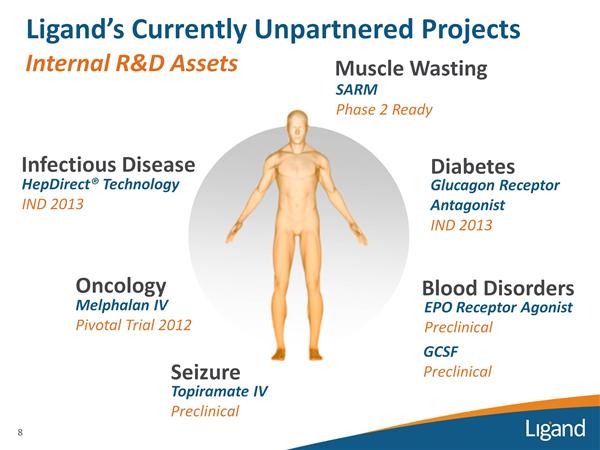
- Early-stage Development Programs (internal and partnered, future "shots on goal" or out-licensing opportunities)
- Acquisitions (Cydex, Metabasis ($MBRX), Pharmacopeia ($PCOP), Neurogen ($NRGN) - complete terms and details)
- Captisol-Cydex (additional info on the formulation technology acquired in 2011 and details on partnerships)
- Discontinued Programs (with >90 biotech and pharma assets in development, not everything works out!)
Ligand $LGND Preclinical and Phase 1 Drug Candidates
Please note indications of development status: Green (advancing), Red (discontinued/abandoned), Purple (uncertain)
LGD-4033
Status: Phase 1 complete- await update and partner Source: internal Ligand
- SARM w/ tissue selective properties-little prostate effect unlike testosterone. Also have large library of backup candidates.
- Program for specialty and long-term muscle wasting patient populations. Muscle health is a new target of interest for pharma research focus
- Goal is to be better drug that testosterone- spare adverse effects on prostate, breast, uterus
- Preclinical results: supportive animal toxicity data, unique AR binding and selective activity, muscle and bone building activity in animal models
- 2010 10k: PK consistent with qD dosing
- FDA has stated the required primary endpts for p2b will have to be total lean body mass and/or performance measures, NOT body weight.
- oral lead p1 for muscle wasting and fraility started 6/2009, single dose done 4q2009-potential for daily dosing (th 31 hrs)
- 6/2010: 90 day tox ongoing for smooth transition to p2.
- 2/2010 started multidose p1b (0.1, 0.3,1,3mg cohorts). 10/2010: in third cohort now, 4th won't start until early 2011. 0.3mg is expected p2/3 dose, 1mg better defines therapeutic window-potential partners waiting to see this data before paying large upfront, 3mg less impt, conclude 1q2011 and partner shortly thereafter.
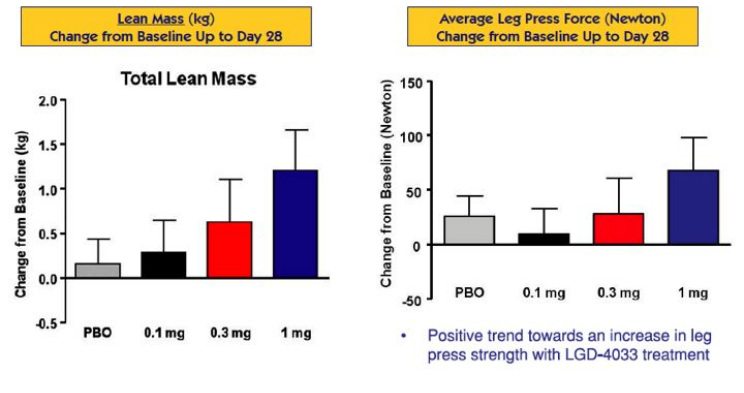
- 6/2011 presented phase 1 data at ENDO meeting. Click here for PR. Click here to download the poster. Key findings:
- LGD-4033 was safe (improvement vs anabolic steroids) and well tolerated at all doses following daily oral administration for three weeks in 21 young healthy males
- No clinically significant dose-related adverse events were reported
- No clinically significant changes in liver function tests, PSA, hematocrit or ECG were seen
- Positive dose-dependent trends in lean muscle mass increase were observed with drug-treated subjects
- Positive dose-dependent trends in functional exercise and strength measures were consistent with anabolic activity
- Previously company had guided for a partnership soon after p1 data (as recently as 2/15/11 webcast). But 6/2011 analyst day slides referenced a 4q2011 "SARM program update" - this was not spoken aloud and no further details were given
MB07133
Status: Phase 1/2 Source: Metabasis
- HepDirect araC prodrug for HCC
- completed multicycle p1/2 w/ "strong response rate."
- 1/2011 licensed to Chiva for development in China for HCC
Beta-secretase inhibitor (MK-8931)
Status: Phase 1 Source: Pharmacopeia
- B-secretase inhibitor (BACE): licensed to Merck for Alzheimers. Originally discovered from research collaboration between Schering-Plough $SGP and Pharmacopeia $PCOP.
- Single dose p1 done, multi dose ongoing
- 3q2009 Schering-Plough conference call: "We're also very excited about the progress of our early development program for BACE or beta secretase, a first and best-in-class treatment for Alzheimer's disease. Our preclinical data in primates which we shared with you last Fall was pretty exciting because it showed that BACE produced a greater than 90% reduction in A-beta 42 peptide upon continuous dosing. This Novel once a day treatment that can penetrate the blood-brain barrier is now in the clinic. Now we've seen in Phase I that with a single dose administration, we are observing a 58% reduction of A-beta peptide in the cerebral spinal fluid." .... "As far as the BACE program is concerned as I just reported we're in Phase I and we're moving into Phase II probably within the next three to six months. It's in the rising multiple dose phase of that study so it's early so I really can't offer too much more than that" - Click to download transcript.
- 2/2009: earned $1m milestone related to progress in the program (later described as for lead selection and phase 1 initiation) [link]
- 6/2010: expect p2 start late-2010.
- 10/2010: still in development, all disclosures thru MRK- they tend to be most tightlipped about exciting pgms, but LGND gets quarterly updates. Increased market opportunity b/c LLY abandoned gamma secretase pgm
- 11/2011: click for updates from Merck $MRK R&D day presentation
- 1/2012: MRK at JP Morgan conference "BACE is a potentially transformative program, moving into phase 2 soon"
Topical JAK3
Status: Preclinical Source: Pharmacopeia
- Janus Kinase 3 (JAK3) inhibitor program for topical/ocular treatment or prevention of skin and eye disorders
- "Specific inhibition of JAK3, which is selectively expressed in immune cells, should provide a lower potential for dose-limiting toxicity than currently available immunosuppressive drugs"
- Multiple potent compounds (IC50 = 0.1-8 nM), >10 fold selectivity vs JAK2 and other related kinases
- No safety issues observed in preliminary studies (i.e. genotoxicity, CYP inhibition, hERG, photocytotoxicity, skin irritation)
- licensed to Wyeth/PFE 12/2006 (up to $175m milestones and tiered double digit royalties), research period extended until 11/2010- received $3.1m payments. Now seen as backup to the JAK3 pgm PFE has in p3. 7/2010: sold rights to this pgm for $3m and terminate rest of deal, LGND maintains right to develop certain compounds topically for skin/eye diseases, elimated required r&d spend.
- 6/2011 this was one of featured internal programs at Analyst day. Developing topical and ocular formulations- targeting dermatology and ophthalmology . could be attractive for partnering
- Post Cydex acquisition: may try to apply captisol technology for these topical formulations
CXCR4 program
Status: Preclinical Source: Pharmacopeia
- CXCR4: rights to series of compound hits licensed to Promaxigen 9/2010 for CNS indications, $0.25m upfront plus milestones/royalties
MB11262
Status: Preclinical Source: Pharmacopeia
- Glucagon receptor (GCGR) antagonist pgm for diabetes - novel mechanism of action
- HepDirect liver targeting to minimize side effects
- preclinical lead optimization stage.
- 1q2011 10q, 2/2011 BIO CEO summary, 6/2011 Analyst Day: listed as internal development program in preclinical status
LG5460
Status: Preclinical Source: Neurogen
- Oral erythropoietin (EPO) program for anemia, complements existing internal pgm for oral EPO receptor agonists.
- 2/2011 BIO CEO summary listed as preclinical program
H3 antagonists
Status: Preclinical Source: Neurogen
- histamine H3 [common allergy meds hit H1 and H2] antagonist pgm
- preclinical- multiple clinical candidates. .
- 2/2011 BIO CEO summary lists as preclinical program for cognitive disorders
TR beta agonist program
Status: preclinical Source: Metabasis
- Thyroid receptor (TR) beta agonist for hyperlipidemia.
- Original lead compound MB07811: phase 1b trial completed (56 pts, dose dep dec in LDL).
- MB10866 is 2nd gen, liver-specific compound.
- 2/2011 BIO CEO summary listed as preclinical program for dyslipidemia
Topiramate
Status: preclinical Source: Cydex
- Topiramate (JNJ Topamax branded is oral for migraine prevention and epilepsy)
- IV formulation (faster onset, wider audience, branded not apporved for acute pts who may not be able to take oral form).
- 5/2008 pre-IND mtg, 2009 initiated p1 for epilepsy.
- 1q2011 10q and 2010 10k: listed as preclinical internal development program
- Not mentioned at 6/2011 Analyst day but mentioned as in development in 6/2011 PR announcing Clopidogrel deal.
IRAK4 inhibitor
Status: Preclinical Source: Internal Ligand
- preclinical for inflammation
- 11/2011: presented preclinical data at ACR meeting.
- 2010 10k: "other internal pgm awaiting devel by LGND or partner"
DGAT inhibitor
Status: Discovery Source: Internal Ligand
- preclinical for diabetes (and perhaps dual use for obesity too). Novel mechanism of action
- 2010 10k: "other internal pgm awaiting devel by LGND or partner"
- 6/2011 Analyst day mentioned as internal development program in preclinical
Glucokinase Activator
- Discovery program for diabetes - novel mechanism of action
- Ligand tissue targeting technology
- Never seen mentioned until 6/2011 Analyst Day