Navigate the Ligand Pharmaceuticals, Inc. ($LGND) Stock Research Pages
- Introduction (valuation, financials, ownership, guidance, helpful links)
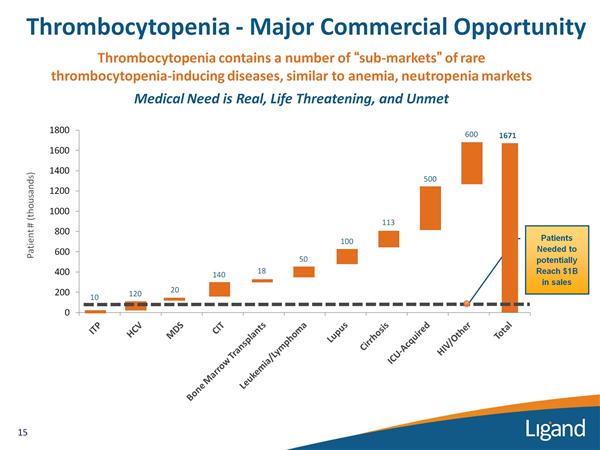
- Promacta (eltrombopag) (key asset licensed to GSK - platelet booster for many thrombocytopenia conditions)
- Kyprolis (carfilzomib) (key asset licensed to Amgen - developed by ONXX for multiple myeloma)
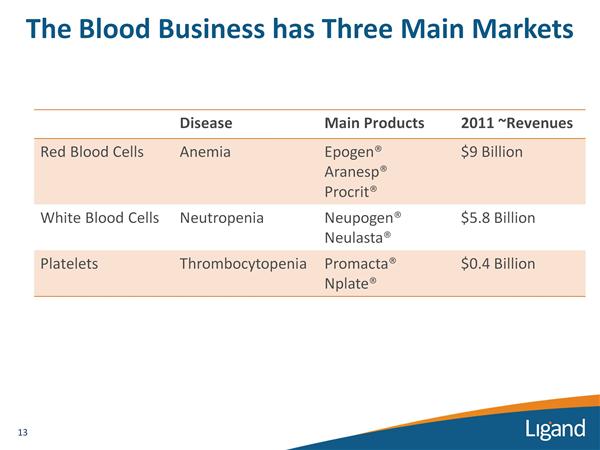
- Other Marketed Products (description of assets currently generating royalty revenue)
- Late-stage Pipeline Assets (generally partnered and developed at little to no cost to LGND and represent future royalty sources)
- Early-stage Development Programs (internal and partnered, future "shots on goal" or out-licensing opportunities)
- Acquisitions (Cydex, Metabasis ($MBRX), Pharmacopeia ($PCOP), Neurogen ($NRGN) - complete terms and details)
- Captisol-Cydex (additional info on the formulation technology acquired in 2011 and details on partnerships)
- Discontinued Programs (with >90 biotech and pharma assets in development, not everything works out!)
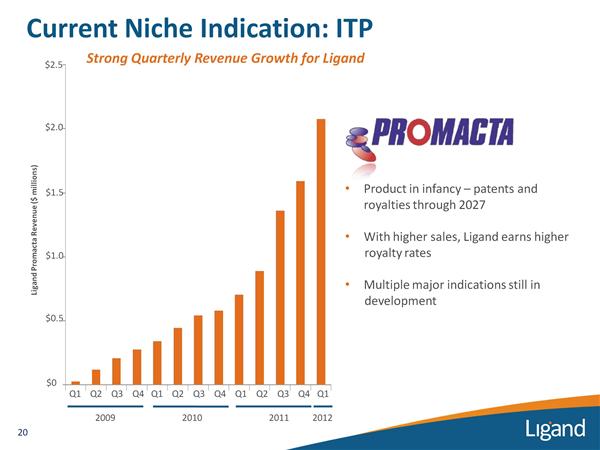
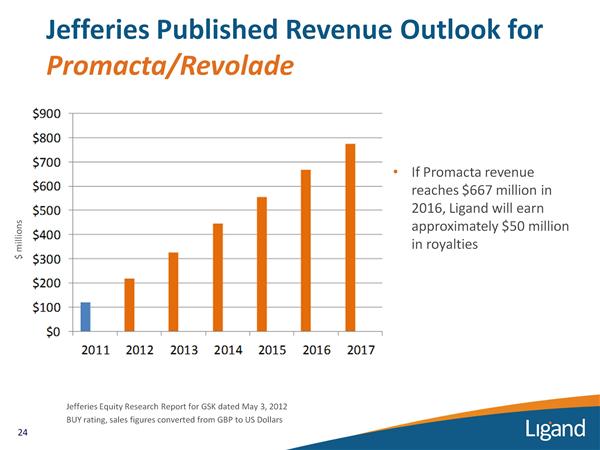
Promacta/Revolade (eltrombopag) for ITP
Status: Marketed and Royalty-producing, phase 2-3 for additional indications Source: Internal Ligand
- Once daily oral thrombopoietin (TPO) receptor agonist to boost platelets and release thrombocytopenia
- Discovered in 1995 in collaboration with GSK: Royalties [net of payment to Rockefeller] 4.7% (<$100m annual sales), 6.6% ($100-200m), 7.5% ($200-$400m), 9.4% ($400m-$1.5b), 9.3% (>$1.5b). Shared with Rockefeller (6-7% of this plus 50% of milestones).
- Received orphan drug designation and accelerated approval in US 12/2008 and a few other countries for ITP ($20m US sales 2009). Filed in Japan 9/2009, approved 11/2010 and launched 1q2011. Approved EU 3/2010 and launched in major markets 4/2010 (req price agreement in each country).
- Initial approval was received based solely on 6 week clinical trial.
- 2/2011: Promacta has now received full approval from FDA- now included efficacy data from 6 month "RAISE" study in ITP and chronic liver data (ELEVATE trial) and 2 yr safety data in ITP. Click for PR. Long-term safety data collection was post-marketing requirement for GSK.
- Patents exp 12/2021 (2010 10k says OB patent exp 12/2024). 6/2011 Analyst day expect royalties thru July 2025 (certain countries maybe?)
- 6/2011: Australian government reverses decision and will start subsidizing Promacta use under the "Pharmaceutical Benefit Scheme (PBS) - click here for link.
- GSK also owns rights to follow-on GSK-5921 (see below).
Promacta (also marketed, see above)
Status: Phase 2, Phase 3 Source: Internal Ligand
- Two P3 ENABLE trials started 4q2007 for HCV (low platelet=cannot go on other therapies). Primary endpoint is sustained viral response (SVR) at 24 weeks after completion of therapy (48-72 weeks in duration depending on HCV genotype). ENABLE1 is in combination with Pegasys + ribovarin. ENABLE2 is in combination with PegIntron + ribovarin.
- 6/2011: first 750 pt phase 3 is complete according to clinicaltrials.gov. 9/2011: Second phase 3 trial is also complete.
- 7/2011: GSK announced on 2q2011 CC that ENABLE1 met its primary endpoint (Click for PR - Click for my notes)
- First phase 3 trial data are available now and will be presented at AASLD 11/2011. Click here for GSK press release.
- GSK 3q2011 CC: Second trial data "is in house and being reviewed"...also ready for phase 3 in Chronic Liver Disease - next steps plan will await final HCV data analysis.
- 4/2010 EASL: avoid infusion in 72% chronic liver disease vs 28% for placebo p2 w/ 292 pts, but 12/2009 halted by IDMC due to inc thrombotic events) are fully enrolled (combo w/ PEGasys or PEGintron and rib, 1500 pts, 200 worldwide sites, primary endpt SVR at 6 months. p2 65% finish tx vs 6%, no thromotic events or liver enzyme elevations)
- 4q2010: completion 3q2011, 2012 launch (concern: IFN causes the problem and this is going away from tx regimen in future).
- 6/2011: 11 ongoing phase 1 or 2 trials in cancer-related thrombocytopenia- solid tumors p2, p1/2 AML started 4/2010, aplastic anemia p2, MDS p2, AML after MDS p2, stem cell transplant p2.
- 1/2011: p2 oncology data 2h2011
GSK-2285921 (aka LGD4665)
Status: Phase 2 - investment depends on Promacta success in new indications Source: internal Ligand
- TPO/promactca follow on aka GSK-5921
- licensed to GSK 12/2008 ($5m upfront, $158m milestones, fully funded), 14.5% royalties 6.5% first year- owe 1.5% to Rockefeller).
- 10/2010 p2 in thrombocytopenia ongoing (and same status listed on 2/2011 GSK pipeline)
- 2/15/11 webcast: has 5-7 years longer patent life and higher royalty rate compared to Promacta