Navigate the Astex Pharmaceuticals ASTX Research Pages
- Introduction (background, summary, links, valuation, financials, outlook, upcoming catalysts)
- Supergen-Astex merger (details on transformative transaction in 2011)
- DACOGEN (aka decitabine, marketed product for myelodysplastic syndrome MDS by Eisai and JNJ)
- AT13387 (HSP90 inhibitor for cancer)
- SGI-110 (follow-on to Dacogen for MDS and AML)
- Amuvatinib (aka MP470 in development for small-cell lung cancer SCLC)
- AT7519 (CDK inhibitor for multiple myeloma)
- Fragment-based Drug Discovery Collaborations (via Astex Ltd side of merger)
- Pipeline (other assets in development)
- Montigen (proposed spin-out transaction info and assets)
Major Event: Supergen merges w/ privately-held Astex
Merger was completed July 20, 2011 - click here for press release.
Here are my notes from the conference call presentation by the two CEO's
Here are my additional notes from the 8-k filed with additional merger document- we are also waiting for the SEC proxy form that will be filed within a couple weeks
- Click here to read the press release announcing the SUPG-Astex deal
- Click here to access joint site with 4/7/11 conference call replay and slide deck
- Click here for letter to SUPG shareholders - Astex management estimates $75m milestone payments next 3 years.
- Key terms: SUPG will pay $25m upfront, issue shares to give Astex owners 35% stake (about 32.5m by my calculation), and pay $30m deferred compensation (SUPG's choice of stock, cash, or combo) over the 30 month period following closing (note these are not contingent on any particular event). Will change name to Astex and list on Nasdaq as $ASTX
- Click here for form 8k filed that has more details of the merger agreement
- The companies held a joint investor/analyst day webcast on April 12th (but only discussed independent pipelines, will not talk about integrated operations until after merger complete)
- June 2011: shareholders of both Astex and SUPG have approved the merger - click here for PR.
- Based on current SUPG price, see estimated pre- and post-merger valuation metrics above
Here are my notes from the conference call presentation by the two CEO's
- SUPG claims this deal will accelerate their business plan by 8 years (I have no idea what this means)
- Joint company will have 5 pharma partnerships w/ $2b aggregate potential earnings
- Discovery capabilities will offer "industry leading value creation" (I like the Astex platform. The SUPG CLIMB hasn't been very impressive to date)
- This is next step in Astex evolution- gain NASDAQ listing, SUPG's clinical and regulatory experience, and funding for its clinical programs
- The $25m upfront payment to Astex shareholders reflects the approximate cash on hand at Astex at ye2010
- $30m additional is deferred and paid over a 30 month period
- Expect to have ~$120m cash on hand at time of closing
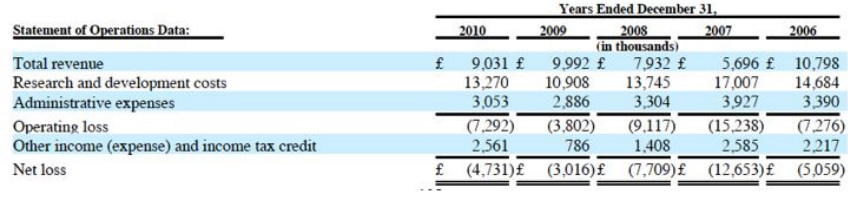
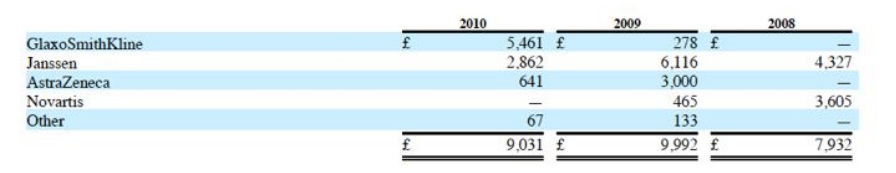
- Dacogen: awaiting info on p3 data. Favorable survival and trend and other supportive data. Now says that both JNJ and Eisai will file in EU/US in 2011 (had been claiming that Eisai would file by March 31, that clearly didn't happen)
- Astex HSP90 inhibitor is about to enter p2 for GIST. This is synthetic, fragment derived compound, not a natural poduct geldenamycin derivative. In responding to analyst question- the Astex CEO admitted that they do see some occular toxicity with their drug, but say it "is different fromother people's reports" and "don't view it as a hurdle at this time" (their drug is water soluble, with fits with info on my SNTA research page regarding which hsp90 inhibitors have this toxicity- this is obviously potentially a big deal regardless of what they say)
- CDK inhibitor- working with MMRF in US, phase 2 has started. NVS has an option on this program- can't disclose any more details (but sounded like royalty rate would vary depending on where in development compound is when optioned)
- 9238- aurora/jak2 inhibitor- starting MM p2 in Canada
- LEE011- first in class CDK4 inhibitor- NVS is developing this in phase 1
- AZD5363- AZN recently presented data at AACR
- Amuvantinib- "being prepared for phase 2", presentation at ASCO June 2011
- SGI-110 is proceeding satisfactorily in phase 1
- FGFR- have delivered compound to JNJ, moving towards preclinical development
- Beta secretase pgm (Alzheimer's) - AZN has selected for formal preclinical development
- Ongoing Astex discovery effort with JNJ for 2 undisclosed targets- developing new chemical entities to take into lead optimization
- Again described SUPG-GSK epigenetics program as working on "DNMT" targets
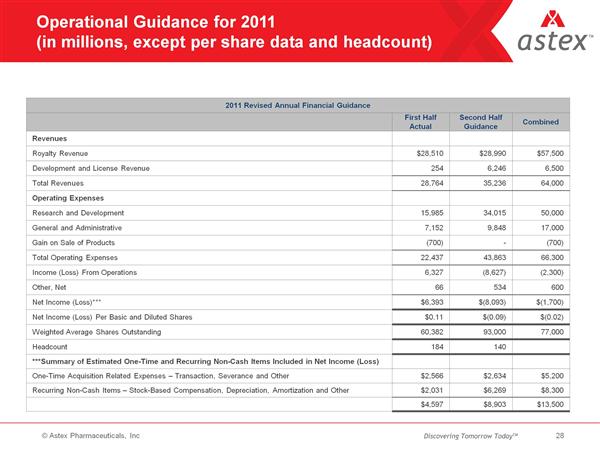
- Astex earned L4.3m milestones in 1q2011, expect L10.8m for full-year 2011, L16m for 2012, L20m for 2013
- In the near-term, maintain focus on operating at or near cash flow neutrality
- Question and answer session:
- Will evaluate all programs after merger, will be conservative with spending and may seek to monetize those that fall outside of oncology focus area
- Have not given Dacogen royalty estimate for EU, but said it could largely supplant US royalties after it goes generic
- No comment on combined synergies or burn rate
- Combined company may selectively decide to take a program later in development by itself (SUPG policy has been to plan to partner after phase 2)
- Astex has 2 years cash on hand based on current approx L10m per year burn rate net of milestones
- Say that yes they have the resources to fund 4 ongoing phase 2 programs
- There is not significant overlap in the two GSK collaborations
Here are my additional notes from the 8-k filed with additional merger document- we are also waiting for the SEC proxy form that will be filed within a couple weeks
- Confidentiality agreement dates back to February of 2009!
- Termination fee in $6m
- Deferred payment schedule is considered in 6 month intervals. The first L5m of milestones received (I believe only for Astex legacy partnerships) is paid to Astex holders. If greater amounts, 50% goes to Astex holders. SUPG may pay this in cash, stock, or a combination at their discretion. Within the first 18 months, a minimum of $15m must be paid. Within 30 months after