Navigate the Isis Pharma ISIS Research Pages
- Introduction (valuation, financials, outlook, company catalysts)
- Antisense background (science, technology, intellectual property, manufacturing)
- KYNAMRO - mipomersen (lead program in regulatory review for familial hypercholesterolemia)
- Mipomersen Liver Safety (special feature pages with extensive notes on long-term safety and efficacy of the drug)
- Cardiovascuar Disease pipeline (addtional CV drugs besides mipomersen)
- Metabolic Disease pipeline (several drugs for type II diabetes, as well as obesity)
- Cancer pipeline (including partnered with Eli Lilly $LLY and OncoGenex $OGXI)
- Severe, Rare, and Neurodegenerative Disease pipeline (incude GSK collaboration)
- Licensing deals and Satellite/spin-off companies (key business development activities)
- Discontinued Programs (not everything works out when you have a large and broad biotech portfolio)
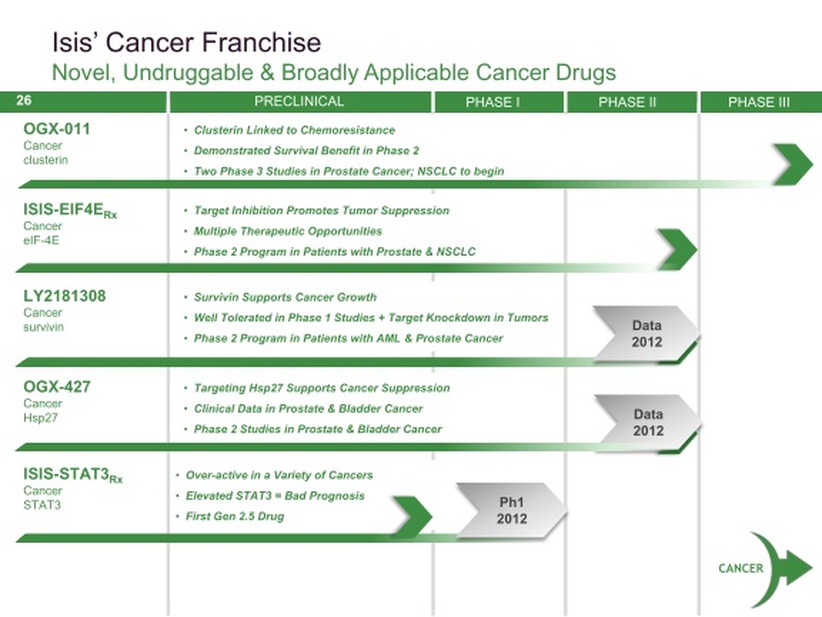
Cancer Franchise
- Tradeoffs: longer POC trials/endpts (liquid better than solid tumors for fast POC), but simpler regulatory path vs CV/metab.
- Positive p2 data from LLY and OGXI were gating factor for commiting more resources in this area.
- Noncoding RNAs (includes microRNA) are new target for cancer therapy.
- Leaky/diffuse blood vessels in cancer accumulate more drug than many tissues.
- 4 drugs in clinic all stopped at 1000-1200 mg/wk w/o reaching MTD/DLT.
- Gly2, Stat5, MCL1 also potential targets for future drug candidates
- FXI and CRP possible cancer indications too.
Custirsen (OGX-011)
- Target is clusterin, "a secreted protein that acts as a cell-survival protein and is over-expressed in response to anti-cancer agents"....Used as adjunct to chemotherapy to enhance effectiveness and combat treatment resistance
- Fast Track designation by FDA for combination with 1st and 2nd line docetaxel for progressive metastatic prostate cancer
- 11/2001 Licensed to and developed by Oncogenex (Click here for $OGXI research page --- Click here for Oncogenex homepage), who in 12/2009 sublicensed worldwide rights to TEVA--at that time ISIS received $10m of upfront and will receive 30% of up to $370m in non-royalty milestones (disagree as to whether this includes sales based milestones), 5.5-7% royalties, newly added $20m milestone upon change in control of OGXI or upon first US commercial sale (if that amount has not paid cumulatively in milestones to ISIS previously)
- OGXI ran five p2 trials, including prostate, breast, and non small cell lung cancer
- Randomized p2 trial in advanced metastatic hormone-refractory prostate cancer (HRPC): OGX-011 plus docetaxel had median surival of 23.8m vs 16.9m for docetaxel alone. HR=0.61 (39% reduced rate of death) and combo was well tolerated
- p1/2 trial in combo w/ first-line chemo for NSCLC data reported 2/2009: median survival 14.1m, one year survival rate 54%, two year survival rate was 30%
- OGX-011 p1 trial: drug given for prostate cancer before surgery so could study resected tissue to help inform p2- expect similar rationale for other candidates
- 2010: SATURN phase 3 initiated- combo w/ second-line chemo in HRPC - pain palliation endpoint
- 2010: SYNERGY phase 3 initiated-combo w/ first-line chemo in metastatic HRPC - OS endpoint w/ SPA
- 2011: intend to initiate p3 study in first-line advanced, inoperable NSCLC
- (see OGXI research page for complete details of OGX-011 clinical trials, etc)
LY2181308 (Survivin)
- Target is Survivin- expressed almost exclusively in cancer cells, "plays a role in cancer cell death and is one of the most commonly over expressed proteins in cancers"
- Licensed to Eli Lilly ($LLY) via deal 8/2001, LLY is responsible for preclinical and clinical development. Broad collaboration and partnership (not sure of upfront terms. Had $100m loan that converted in 2.5m shares at $40/share in 8/2005 upon deal extension). 12/31/10: have rec'd $4.3m milestones to date and up to $25m more plus royalties
- Completed p1 shows that drug penetrates tumor tissue and reduces survivin mRNA and protein levels in tumor cells
- As of 7/2011 LLY pipeline listed as phase 2 - per 3q2011 LLY CC, not entering phase 3 in 2011
- 2010 10k: phase 2 combo trial ongoing in 2nd line advanced, metastatic NSCLC pts
- 2010 10k: phase 2 ongoing in combo w/ first-line docetaxel for HRPC
- 2010 10k: phase 2a complete in refractory AML
- 9/2010: data late 2010, p3 possible in 2011
OGX-427
- Target is Heat Shock protein 27 (HSP27) "a cell survival protein that is over-produced in response to many cancer treatments, including hormone ablation therapy, chemotherapy and radiation therapy. Studies have shown that increased Hsp27 production is prevalent in many human cancers...Studies have also linked increased Hsp27 production to faster rates of cancer progression, treatment resistance and shorter survival duration."
- 1/2005 licensed to and 4/2005 selected for development by Oncogenex (Click here for $OGXI research page --- Click here for Oncogenex webpage)- up to $4.2m milestones plus royalties.
- 6/2010 p1 results: well tolerated alone and in combo w/ docetaxel. Single agent caused decline in circulating tumor cells
- p2 initiated 3q2010 (72 chemo naïve pts) in prostate cancer- triggered $0.75m milestone to ISIS 1/2011
- p1 ongoing in bladder cancer and OGXI plans p2 in first-line metastatic bladder cancer in 2011
- (see OGXI research page for complete clinical trial info, etc)
eIF-4E Inhibitor (fka LY2275796)
- Antisense targeting eukaryotic initiation factor-4E (eIF-4E), which is involved in many cellular processes critical to tumor cell growth and survival and is overexpressed in many cancers
- Preclinical: showed broad anti-tumor activity and demonstrated that tumor cells were more sensitive to eIF-4E inhibition that normal cells
- LLY completed a p1 trial, safe up to 1200 mg/wk, achieved active conc in tumor cells (known as LY2275796)
- 12/2009 reacquired from LLY ("to accelerate its development")..but 4/2011 still appears in LLY pipeline page
- LLY has right to acquire the rights to drug at phase 2 POC on predefined terms. If publically disclose data and LLY does not opt-in, can partner with another company
- 7/2011: still listed as phase 2 asset in LLY 2q2011 slides. Nothing mentioned for 2011 milestone/event.
- 1/2011: initiated p2 in HPRC and NSCLC in combo w/ first-line tx (randomized, controlled, ~100 pts each), will expand to other indications
Stat3 Inhibitor
- Target is signal transducer and activator of transcription 3 (STAT3). Overactive in many cancers, promotes tumor cell growth and prevents cell death
- Preclinical: broad antitumor activity with good safety profile
- 2010 10k: will study in multiple myeloma and liver cancer
- Added to pipeline 1/2011.
- 2010 10k: plan to initiate IND-enabling studies in 2011