Navigate the Isis Pharma ISIS Research Pages
- Introduction (valuation, financials, outlook, company catalysts)
- Antisense background (science, technology, intellectual property, manufacturing)
- KYNAMRO - mipomersen (lead program in regulatory review for familial hypercholesterolemia)
- Mipomersen Liver Safety (special feature pages with extensive notes on long-term safety and efficacy of the drug)
- Cardiovascuar Disease pipeline (addtional CV drugs besides mipomersen)
- Metabolic Disease pipeline (several drugs for type II diabetes, as well as obesity)
- Cancer pipeline (including partnered with Eli Lilly $LLY and OncoGenex $OGXI)
- Severe, Rare, and Neurodegenerative Disease pipeline (incude GSK collaboration)
- Licensing deals and Satellite/spin-off companies (key business development activities)
- Discontinued Programs (not everything works out when you have a large and broad biotech portfolio)
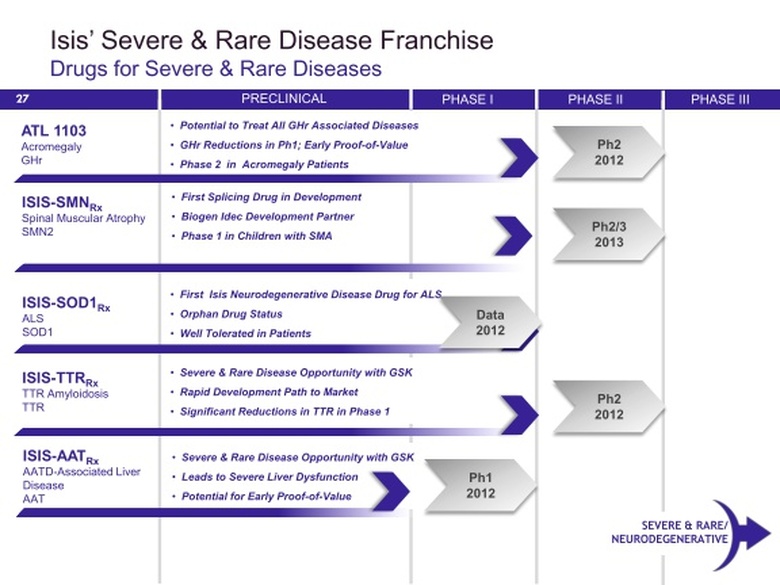
Neurodegenerative, Severe, and Rare Diseases Franchise
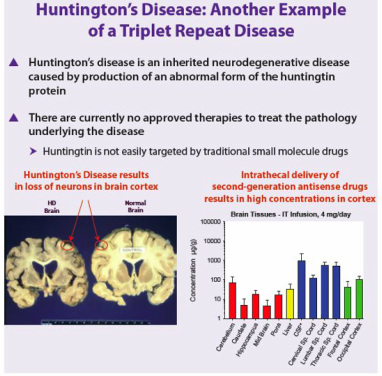
- Antisense drugs don't cross BBB, so intrathecal injection into CSF.
- Start w/ rare diseases- faster to market and balance portfolio. NIH says there are 5000-8000 of these.
- "Most of these diseases are genetic or have a genetic component. In some cases, the onset of disease is characterized by the presence of a protein that, through a genetic defect, cannot function properly in the cell. In order to treat these diseases, we can discover and develop antisense drugs that selectively inhibit the production of only the disease-causing protein. In other cases, alternative splicing can result in the omission of proteins that are critical for normal cellular function. Using antisense technology, we can direct alternate splicing to potentially correct for a genetic defect." (2010 10k)
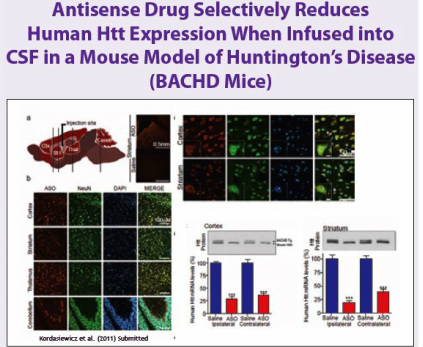
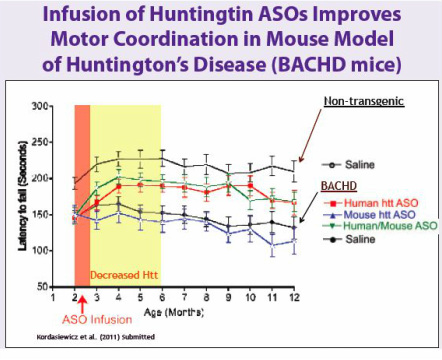
- Pgms in HD, PD, and muscular dystrophy ongoing (plan 4 new nominated pgsm in 1-2 yrs).
- 11/2007 Collaboration with CHDI Foundation that provides funding (up to about $10m) for Huntington's Disease (HD) program. Upon sublicense, split proceeds up to point at which CHDI recovers investment with interest, Isis keeps the rest. 8/2011 renewed (previous deal had ended in 2010, may get reimbursed for $2m expenses between new and old deal) to conduct IND-enabling studies. Would expect to later collaborate for clinical development (PR).
- Genzyme has some rights to option drugs at p2 but no restrictions on other partnerships
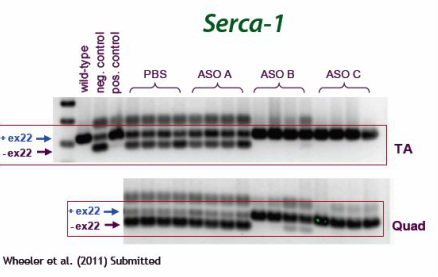
- Click here to download poster about this franchise from June 2011 Annual Meeting - see select data from this presentation below
SOD1 Inhibitor
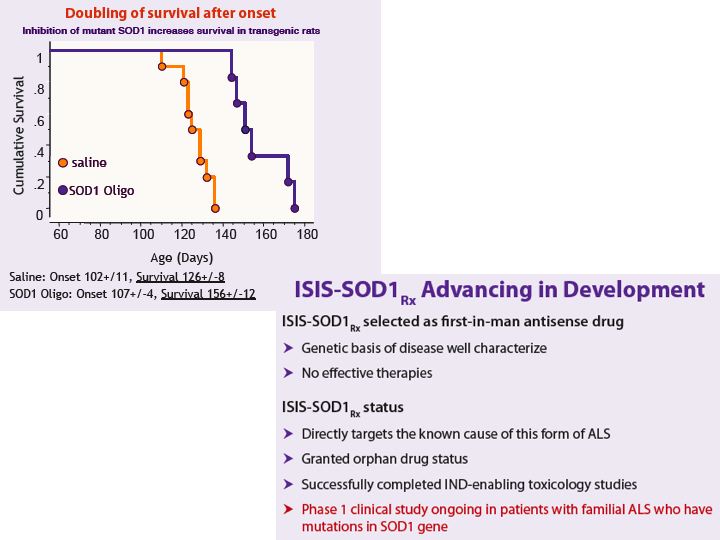
- Antisense targeting superoxide dismutase (SOD1). Deliver into CNS to reduce SOD1 expression in neurons and surrounding cells
- FDA Orphan Drug designation for ALS (Lou Gehrig's disease). 10% of patients have familial history, initially just 600 US pts w/ this mutation - but could have broader benefit too
- Preclinical (JCI publication): SOD1 antisense slowed ALS symptoms and prolonged life in rat model
- Funding provided by Ludwig Institute and Center for Neurological Studies for IND-enabling studies (would owe milestone payments and modest royalties)
- Funding from ALS Association and Muscular Dystrophy Association for p1 trial (apparently no royalties etc owed)
- Genzyme has right of first negotiation to license this product
- 2010 10k: double blind placebo controlled p1 ongoing in familial ALS patients- (date uncertain) said:) will be slow but next trial could be registrational
- 9/2010: p2 in 2011
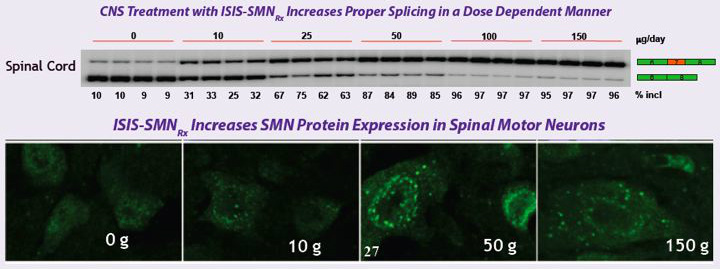
SMN2 Splicing Modulator for Spinal Muscular Atrophy (SMA)
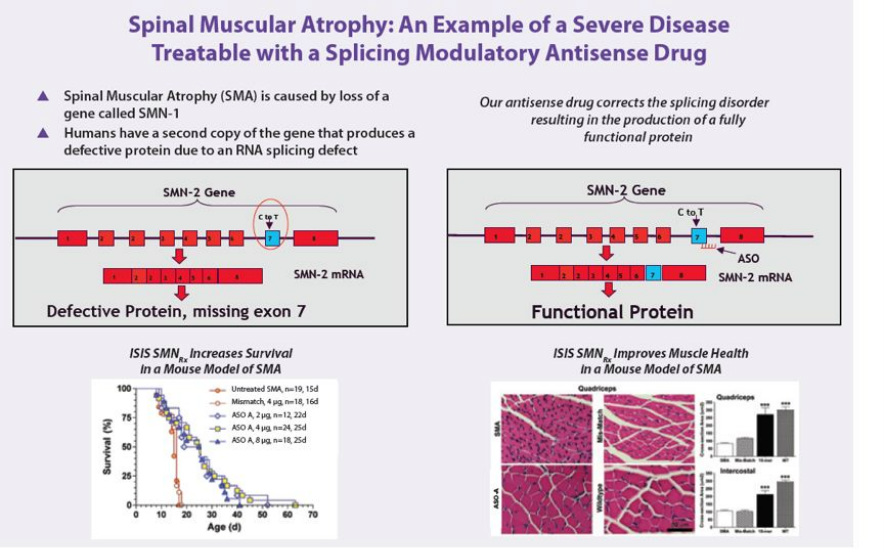
- First antisense candidate that alters splicing instead of inhibiting protein production (added to pipeline in 2009)
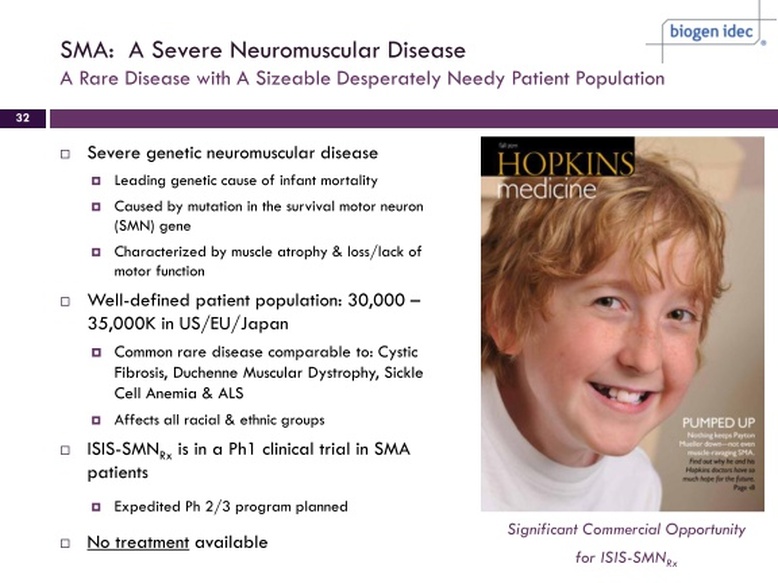
- for Spinal Muscular Atrophy (SMA, occurs in 1 of every 6000-10000 births). Most with Type 1 form die within two years. Genetic deletion of survival motor neuron 1 (SMN1) causes the disease.
- Drug alters splicing of SMN2, resulting in increased production of SMN protein that is needed for normal motor function
- preclinical- complete tox studies by 6/2011 and then start p1 in 2011
- 6/2011: plan to start clinical trials late 2011 or early 2012
- Discovered in collaboration with and licensed patents from Cold Soring Harbor (up to $0.9m milestones, plus sublicense fees and royalties).
- Utilizes technology in-licensed from Univ of Massachusetts (up to $0.65m total milestones, plus sublicense fees and royalties)
- 6/2011: presented data at SMA conference that "showed that systemic administration of ISIS-SMNRx produced therapeutic benefit from SMN restoration in peripheral tissue, suggesting that there may be therapeutic benefit for an increase in functional SMN protein in tissues outside of the central nervous system. In addition, researchers showed that ISIS-SMNRx produced sustained activity in mouse models of SMA and that target tissue concentrations were achieved in non-human primates following a single intrathecal injection. Isis and collaborators also outlined ongoing work to develop new animal models of disease that will help Isis and its collaborators further understand the role of SMN in the pathology of disease."
- Click here for 7/2011 article re updates from SMA conference.
DM1 - Myotonic Dystrophy
June 2012 - second partnership with Biogen.
GlaxoSmithKline $GSK Preferred Partner Deal
- 3/2010: collaboration for up to 6 programs in rare and serious diseases, including infectious diseases and some conditions causing blindness
- $35m upfront, ~$20m ($120m total) milestones per pgm thru p2 POC, up to $1.5B total milestones, up to double digit royalties.
- 5/2011: deal expanded to include a 6th program, GSK paid ISIS $3m additional upfront fee (click here for PR)
- ISIS is responsible for development thru phase 2 proof of concept, at which time GSK has the option to license the product
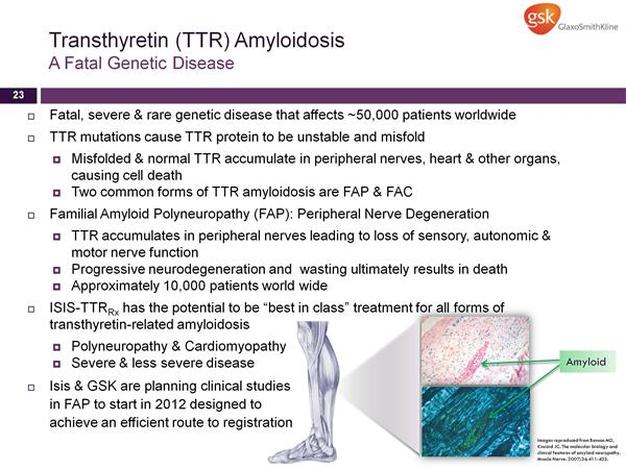
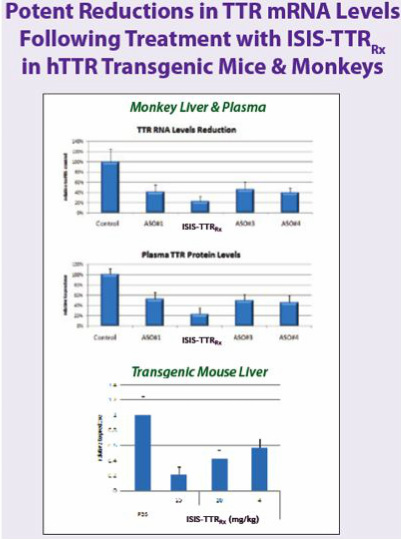
TTR Inhibitor (fka GSK1)
- "ISIS-TTRRx is designed to treat transthyretin amyloidosis [TTRA], a severe and rare genetic disease characterized by slow degeneration of peripheral nerve and/or heart tissues...a fatal disease that affects one in 100,000 people or about 50,000 people worldwide. ISIS-TTRRx will initially be developed for patients with familial amyloid polyneuropathy, or FAP, who have TTR build up in their peripheral nerves and experience the loss of motor functions, such as walking. Although these patients experience debilitating nerve damage throughout their body, they typically die from wasting due to the accumulation of TTR in the intestinal tract, which prevents the proper absorption of nutrients. Currently there are no approved drugs to treat transthyretin amyloidosis, and the only approved available option is liver transplant. Unfortunately, availability of donor livers is very limited and only a fraction of patients are eligible for this very expensive and invasive procedure." - see the ALNY research page for more details on TTRA
- 10/2011 - click here for slide deck on TTR antisense program.
- 8/2011 - Fierce Biotech article about TTR and new research at Scripps identifying potential new stabilizers of TTR
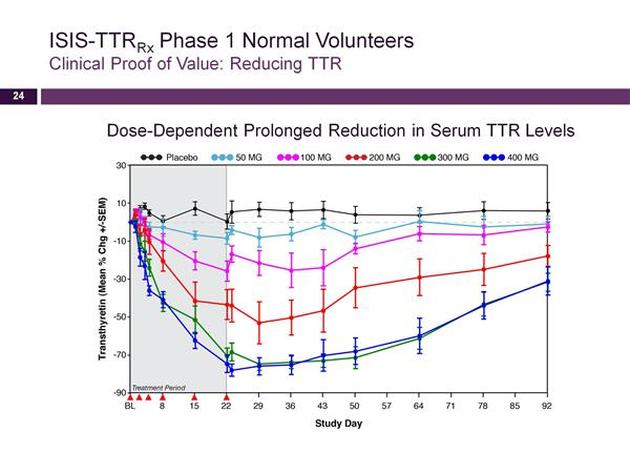
- 5/2011: Phase 1 trial initiated, earning ISIS $5m milestone. Click here for PR. No other details on trial design provided.
- 7/2010: earned $5m milestone for first nominated development candidate (aka GSK1 at first before target disclosed)
- 2010 10k: expect phase 1 in 2011. (timeline slipped from 9/2010 forecast of p1 late 2010 or 2011)
- ALNY has competing RNAi-based TTR program.
TTR Competitor: Tafamidis from Pfizer PFE (via FoldRx acquisition)
- In 2010, Pfizer (via purchase of FoldRx) “filed a marketing authorization application, or MAA, for tafamidis, an oral small molecule stabilizer of TTR, with the EMA. Tafamidis has orphan drug status in the European Union, or EU, for the treatment of FAP associated with ATTR.”
- 4/2011: Click here for details of phase 2/3 data results.
- Now approved and sold in Europe as VYNDAQEL
- Under review in US by FDA with June 2012 PDUFA date.