Navigate the Isis Pharma ISIS Research Pages
- Introduction (valuation, financials, outlook, company catalysts)
- Antisense background (science, technology, intellectual property, manufacturing)
- KYNAMRO - mipomersen (lead program in regulatory review for familial hypercholesterolemia)
- Mipomersen Liver Safety (special feature pages with extensive notes on long-term safety and efficacy of the drug)
- Cardiovascuar Disease pipeline (addtional CV drugs besides mipomersen)
- Metabolic Disease pipeline (several drugs for type II diabetes, as well as obesity)
- Cancer pipeline (including partnered with Eli Lilly $LLY and OncoGenex $OGXI)
- Severe, Rare, and Neurodegenerative Disease pipeline (incude GSK collaboration)
- Licensing deals and Satellite/spin-off companies (key business development activities)
- Discontinued Programs (not everything works out when you have a large and broad biotech portfolio)
Kynamro (mipomersen, fka ISIS-301012) - Regulatory and Commercial Info
- Antisense targeting apolipoprotein-B (apo-B) - see next section for more details
- Partnered w/ Genzyme in 1/2008 in auction process following successful p2 data. $175m upfront, $150 in stock (5m shares at $30, with limitations on sales and additional purchases) and up to $1.5b in milestones, 30-50% royalties on mipomersen and follow-on compounds. ISIS pays the first $125m in development costs, which will be achieved in 2011, then will split equally. GENZ also gets right of first negotiation for SOD1 drug (see below)
- GENZ has rights to 3-5x more potent backup and potential generation 2.5 chemistry follow-on.
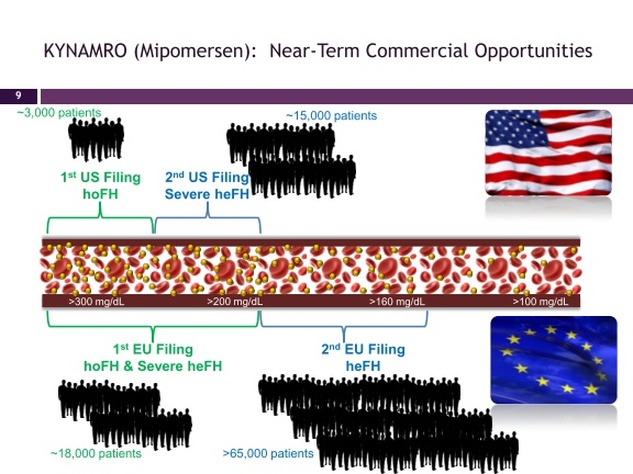
- first NDA and EU filings 2011 (35000 intitial severe hyperchol pop in US/EU/Japan). Seek initial indication for homozygous familial hypercholesterolemia (HoFH) in the US and HoFH and (probably) severe heterozygous FH (HeFH) in the EU.
- The FDA will require an additional study for the severe HeFH indication in which safety and efficacy are measured for 12 month exposure to the drug. An outcome study is not required in this indication (not feasible due to low pt population).
- Filing is for 200mg weekly, also successful prelim test of 30 mg daily or 70 mg 3x/wk.
- 1/2011 regulatory update: HoFH data package is sufficient for filing, but NDA may shift to 2h2011.
- 7/2011: Genzyme submits application for European approval in HoFH and severe HeFH indications, discloses trade name Kynamro (PR). 9/2011: SNY investor webcast guides for Kynamro launch by March 2012.
- 2/2011 initiated PK bridging study in healthy Japanese volunteers- 50,100, 200 mg single doses
- US patents expire 2021 (7,407,943) and 2024 (7,511,131)
- ISIS will manufacture initial launch quantities of drug substance, GENZ (SNY) is responsible for fill/finish of vials and prefilled syringes and must secure a long-term supplier of drug substance
- Commercial roll-out planned (per 6/2011 Annual Meeting manufacturing poster)
2nd Wave: Canada, Spain, Belgium, France, Italy, Portugal, Mexico, Venezuela, Colombia, Australia, Taiwan
3rd Wave: Japan, South Korea, New Zealand, South Africa, China, India
Mipomersen: Scientific and Clinical Info
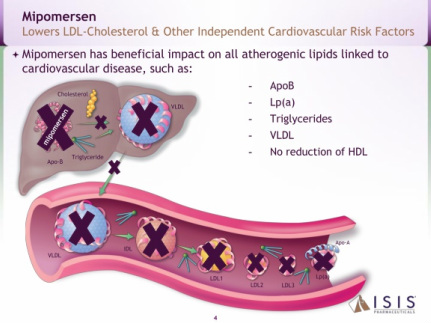
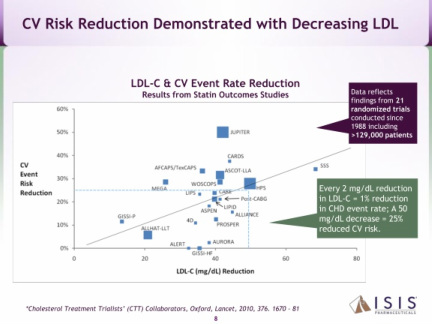
- Antisense targeting apolipoprotein-B (apo-B) with a 30 day half-life. "Apo-B provides the structural core for all atherogenic lipids, including LDL-C, which carry cholesterol through the bloodstream. Mipomersen reduces LDL-C and other atherogenic lipids linked to cardiovascular disease by preventing their formation." (2010 10k)
- Click here for 8/2011 RNAi Therapeutics blog post re ApoB target.
- FH patients cannot properly metabolize LDL-C and therefore have very high LDL-C levels. "HoFH patients can have LDL-C levels greater than 600 mg/dL and are at very high risk for early coronary events and sudden death. Severe heFH patients comprise a small subset of heterozygous FH patients, a more common form of the disorder. Severe heFH patients have LDL-C levels greater than 200 mg/dL with coronary artery disease or more than 300 mg/dL without coronary artery disease despite maintaining a regimen of maximally tolerated lipid-lowering therapy. Patients with untreated FH have a 50 percent mortality rate by age 60
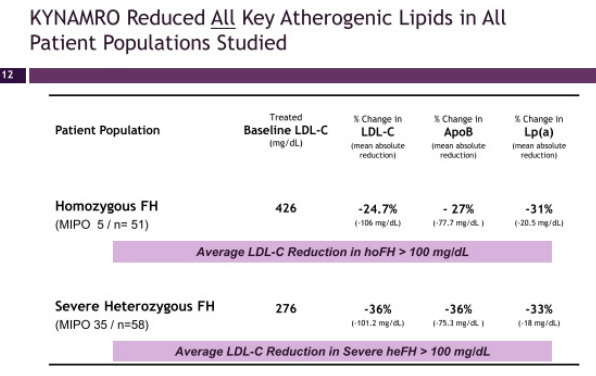
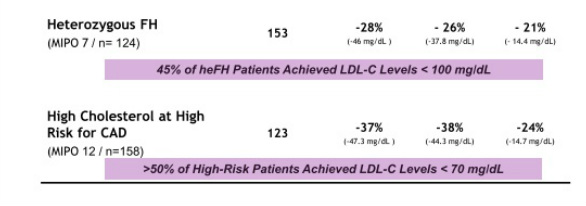
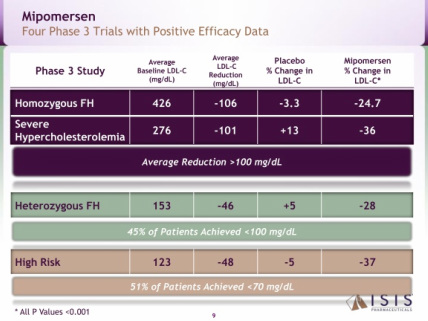
- Four p3 trials successfully completed in combination with maximally tolerated lipid lowering therapy (met all primary, secondary, and tertiary endpoints) - see summary chart of the data below. Mipomersen also significantly reduces apo-B, Lp(a), triglycerides, and VLDL. Common side effects include injection site reactions, flu-like symptoms and elevations in liver transaminases
- Average reductions of LDL levels of >100 mg/dL were achieved in the two most severe patient populations. 45% of patients achieved <100 mg/dL level in the HeFH trial. 51% of patients achieved <70 mg/dL in the high-risk patient trial.
- 6/2011: made two data presentations at the European Atherosclerosis Society (EAS) Congress - Click here for PR - click here for complete abstracts.
2) "In the phase 3 trial in HeFH patients with CAD, an additional analysis revealed that mipomersen reduced the percentage of patients with LDL-C levels ≥ 160 mg/dL by 95 percent (from 39 percent to 2 percent); with LDL-C levels ≥ 130 mg/dL by 74 percent (from 62 percent to 16 percent); and with LDL-C levels ≥ 100 mg/dL by 45 percent (from 98 percent to 54 percent). The reductions observed were in addition to those achieved with the patients’ existing therapeutic regimens"
Mipomersen competitor: Lomitapide from AEGR
- Lomitapide is a microsomal triglyercide transfer protein [MTP] inhibitor being developed by Aegerion Pharma $AEGR
- "Aegerion is initially developing lomitapide as an oral, once-a-day treatment for patients with homozygous FH. Aegerion is currently evaluating lomitapide in a Phase 3 study in 29 patients with homozygous FH. In earlier studies evaluating lomitapide, patients discontinued use of lomitapide at a high rate due to gastrointestinal adverse events, such as diarrhea, nausea and vomiting. In addition, some patients experienced elevations in liver enzymes and increased mean levels of fat in the liver, or hepatic fat, both of which Aegerion states it has observed in its ongoing Phase 3 clinical trial of lomitapide. Aegerion also states that patients in its ongoing Phase 3 trial have also experienced adverse gastrointestinal events. Aegerion states that before they submit an NDA for lomitapide to the FDA in 2011 they must complete additional studies to assess various other aspects of lomitapide. Aegerion plans to follow with an MAA submission in Europe in 2012." (2010 10k)