Navigate the Isis Pharma ISIS Research Pages
- Introduction (valuation, financials, outlook, company catalysts)
- Antisense background (science, technology, intellectual property, manufacturing)
- KYNAMRO - mipomersen (lead program in regulatory review for familial hypercholesterolemia)
- Mipomersen Liver Safety (special feature pages with extensive notes on long-term safety and efficacy of the drug)
- Cardiovascuar Disease pipeline (addtional CV drugs besides mipomersen)
- Metabolic Disease pipeline (several drugs for type II diabetes, as well as obesity)
- Cancer pipeline (including partnered with Eli Lilly $LLY and OncoGenex $OGXI)
- Severe, Rare, and Neurodegenerative Disease pipeline (incude GSK collaboration)
- Licensing deals and Satellite/spin-off companies (key business development activities)
- Discontinued Programs (not everything works out when you have a large and broad biotech portfolio)
Antisense Technology
- RNase H mediated antisense: "Antisense technology interrupts the cell's protein production process by preventing the RNA instructions from reaching the ribosome, thus inhibiting the synthesis of the protein. The mRNA sequence of nucleotides that carries the information for protein production is called the "sense" strand. The complementary nucleotide chain that binds specifically to the sense strand is called the "antisense" strand. We use the information contained in mRNA to design chemical structures, called antisense oligonucleotides or antisense drugs, which resemble DNA and RNA and are the complement of mRNA. These potent antisense drugs inhibit the production of disease-causing proteins. Specifically, almost all of our antisense drugs in development cause a cellular enzyme called ribonuclease H1, or RNase H1, to degrade the target mRNA. The drug itself remains intact during this process, so it can remain active against additional target mRNA molecules and repeatedly trigger their degradation. Our antisense drugs can selectively bind to a mRNA that codes for a specific protein and will not bind to closely related RNAs, providing a level of specificity that is better than traditional drugs. As a result, we can design antisense drugs that selectively inhibit the disease-causing member of the group without interfering with those members of the group necessary for normal bodily functions. This unique specificity means that antisense drugs may be less toxic than traditional drugs because we can design them to minimize the impact on unintended targets." (2010 10k)
- Advantage vs small molecule drug discovery: "the design of antisense compounds is less complex, more rapid and more efficient than traditional drug design directed at protein targets. Traditional drug design requires companies to identify a small molecule that will interact with protein structures to affect the disease-causing process. Since predicting which small molecules will do this has proven to be difficult, traditional drug discovery involves testing hundreds of thousands of small molecules for their ability to interfere with protein function. As a result, traditional drug discovery is a labor intensive, low probability endeavor. In contrast, we design our antisense compounds to bind to mRNA through well understood processes. We can design prototype antisense drugs as soon as we identify the sequence for the target mRNA." (2010 10k)
- RNA interference (RNAi): "RNAi is an antisense mechanism that uses small interfering RNA, or siRNA, to target mRNA sequences. Most companies approach siRNA using double stranded oligonucleotides which exploit a cellular protein complex called the RNA-induced silencing complex, or RISC, to bind to the mRNA and to prevent the production of a disease-causing protein. We have a strong and growing intellectual property position in RNAi methodology and oligonucleotide chemistry for siRNA therapeutics. We have licensed these patents to Alnylam for double-stranded siRNA therapeutics." (2010 10k)
- single strand RNAi: "At present, the double-stranded siRNA drugs in development by others are either administered locally or, if administered systemically require complex formulations to achieve sufficient delivery. We have recently identified the critical drug design elements required to achieve RNAi activity with a single stranded RNAi drug. We have also begun to chemically optimize these design elements to ensure that they survive long enough under physiological conditions to produce the desired activity in animals. As a result, we have created single stranded RNAi compounds that, when administered systemically, distribute in a manner similar to our second generation RNase H antisense drugs, without requiring the complex formulation or delivery vehicle typically necessary for double stranded RNAi oligonucleotides. These new single stranded RNAi drug designs are an exciting advance in RNAi technology." (2010 10k) See below- this technology was previously partnered with Alnylam $ALNY but they terminated the deal in 2010
- Splicing: "Splicing is a normal cellular mechanism that the cell uses to produce many different, but closely related proteins from a single gene by varying the processing of the RNA. Scientists estimate that because of the approximately 25,000 genes in the human genome, 40 percent to 60 percent have alternative splice forms. In some cases, alternative splicing of proteins can produce altered proteins that are involved in disease. In other cases, alternative splicing can omit proteins that are critical for normal cellular function which can lead to disease. Using antisense technology, we can direct alternate splicing to produce a protein critical for normal cellular function, and potentially correct for a genetic defect. Examples of applications of antisense modulation of splicing to treat genetic disease include, SMA, thalessemia, cystic fibrosis and Duchenne's muscular dystrophy" (2010 10k)
- microRNA: "Our antisense technology is not limited to RNA sequences that translate into proteins, but rather we can apply the principals of our technology to develop drugs that target other RNAs, such as microRNAs. MicroRNAs are small, RNA molecules, typically 20 to 25 nucleotides in length, that do not encode proteins but instead work as natural antisense sequences that scientists believe regulate the expression of approximately one-third of all human genes. To date, scientists have identified more than 700 microRNAs in the human genome, and have shown that the absence or presence of specific microRNAs in various cells are associated with specific human diseases, including cancer, viral infection, metabolic disorders and inflammatory disease. MicroRNAs themselves may be drug targets. For instance, if a single microRNA can change the expression of a protein that may be involved in disease, then inhibiting this microRNA could provide a therapeutic benefit. Alternately, microRNAs could be used as drugs themselves, where increasing the cell concentration of a particular microRNA could modulate the expression of a particular protein." (2010 10k). See below- this technology is the basis for the formation of the Regulus JV with ALNY
- Aptamers: "Aptamers are oligonucleotide molecules that form a three-dimensional shape that specifically binds to a protein molecule of interest for disease treatment. Aptamers differ from antisense inhibitors because they do not bind to an RNA sequence to inhibit protein formation, but rather they modify the function of a protein by binding directly to the protein. However, our patented chemical toolbox can greatly improve the chance that an aptamer will succeed as a drug."
More information on Antisense
- Click here to download poster on second generation antisense drug pharmacodynamics and pharmacokinetics from the June 2011 ISIS pharma annual meeting.
- Click here to download poster summarizing antisense drugs' safety findings across many drugs/trials/indications/patient populations (including photos of injection site reactions - ISRs) from June 2011 annual meeting.
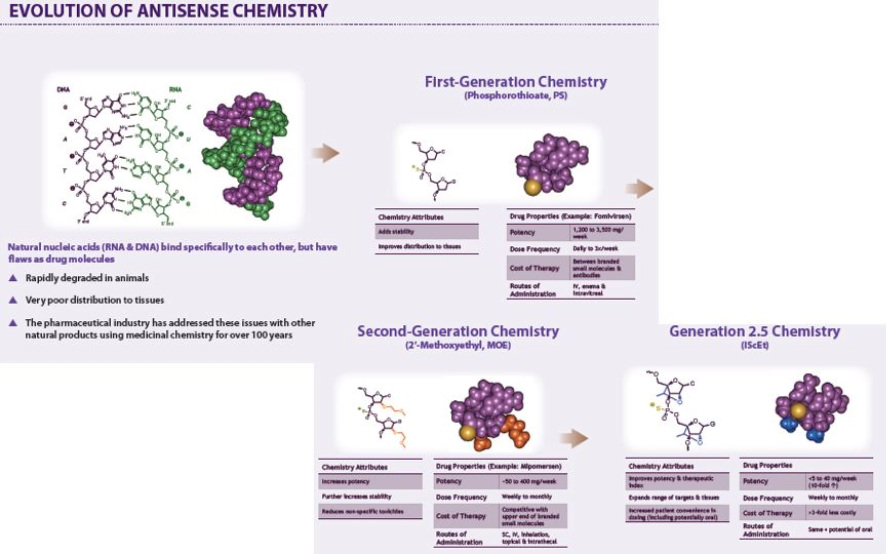
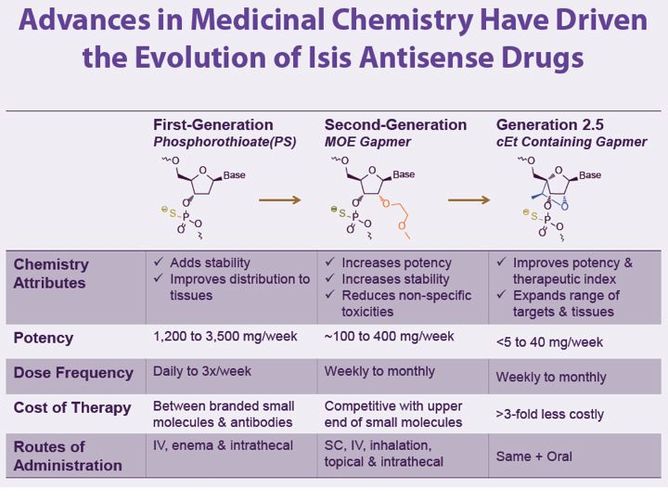
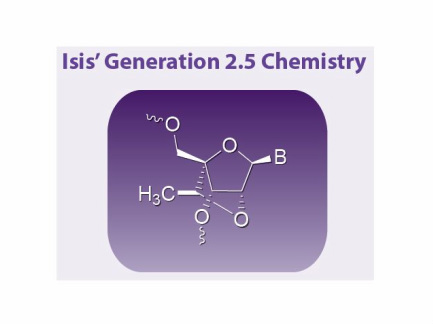
- Click here to download poster on antisense chemistry advances, focused on the newest generation 2.5 (see excerpted illustrations below) from June 2011 Annual Meeting..
Intellectual Property (IP)
- See this poster from 6/2011 Annual Meeting for extensive details on IP estate.
- Protected assets include:
- Chemical Modification of Oligonucleotides (sequence-independent)
- Antisense Drug Design Motifs (sequence-independent)
- Antisense sequences targeting genes of therapeutic interest (independent of oligo chemistry)
- Drug Products (combo of antisense sequence and drug design motif)
- As of 4/2011, ISIS had 1485 issued patents (838 in US) and 897 pending patent applications (288 in US)
- IP coverage of 2'MOE [or for you chemists: 2'-O-(2-Methoxyethyl)] drug designs includes:
- MOE gapmer antisense oligos (ASOs) until 3/ 2023 (Generation 2.0 Chemistry)
- Gap-widened MOE ASOs until 2025
- Shortmer MOE ASOx until 2027 (includes Generation 2.2 Chemistry)
- ssRNAi motifs protected until at least 2029
- Optimized Generation 2.5 ASOs until 2032
- Generation 2.5 chemistry is covered by several issued patents expiring 2027 or later: U.S. Patent Nos. 7,399,845, 7,547,684 & 7,666,854 and European Patent No. EP1984381 (pending in Japan, India, South Korea, China)
Manufacturing
- "Due to the growing numbers of our antisense drug development partners and the clinical successes of our antisense drugs, including mipomersen, in 2009 we increased our manufacturing capacity by upgrading and optimizing the efficiency of our manufacturing facility. In 2011, Genzyme plans to submit for marketing approval of mipomersen in the United States and Europe. If approved, the increased capacity of our manufacturing facility will provide the supply of drug substance we believe is necessary for the initial launch of mipomersen." (2010 10k)
- Learn more about the manufacturing process with a poster from the 6/2011 Annual Meeting.
- Is a "platform" manufacturing process. Have reduced costs to achieve typical pharmaceutical margins
- Isis has expanded capacity by a total of 18,400x per run versus original setup
- Isis site has two manufacturing suites (one can do small batches of multiple products, one does large batches of a single product) with an annual capacity of 500 kg
- 6/2011 Isis is currently manufacturing mipomersen product to support initial commercial launch