Navigate the Isis Pharma ISIS Research Pages
- Introduction (valuation, financials, outlook, company catalysts)
- Antisense background (science, technology, intellectual property, manufacturing)
- KYNAMRO - mipomersen (lead program in regulatory review for familial hypercholesterolemia)
- Mipomersen Liver Safety (special feature pages with extensive notes on long-term safety and efficacy of the drug)
- Cardiovascuar Disease pipeline (addtional CV drugs besides mipomersen)
- Metabolic Disease pipeline (several drugs for type II diabetes, as well as obesity)
- Cancer pipeline (including partnered with Eli Lilly $LLY and OncoGenex $OGXI)
- Severe, Rare, and Neurodegenerative Disease pipeline (incude GSK collaboration)
- Licensing deals and Satellite/spin-off companies (key business development activities)
- Discontinued Programs (not everything works out when you have a large and broad biotech portfolio)
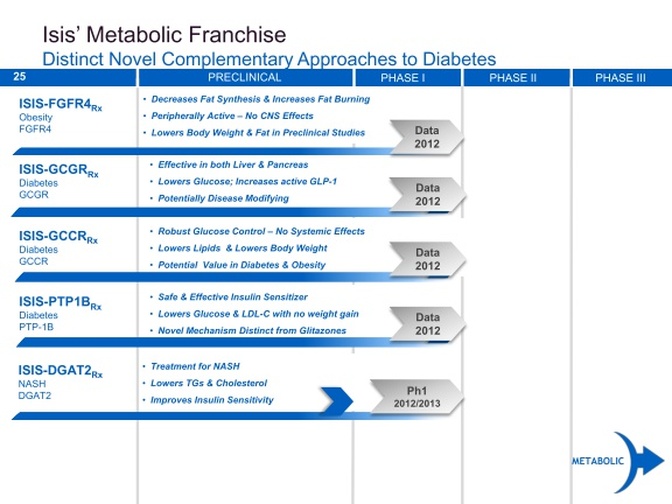
Metabolic Disease Franchise
- Diabetes affects 25m pts in US (8% of adults), 90-95% of these are type 2 diabetes
- Developing antisense drugs to add to existing diabetes and obesity drugs.
- Many targets for metabolic diseases are expressed in liver and fat cells - accumulate antisense drugs well even at low doses
- many targets undruggable b/c part of large protein families
- rapid clinical POC
- can add to existing therapies
- 4/2010: well along on selecting first of peripherally acting obesity targets
- 4/2010: each pgm has follow-on (related to generation 2.5 chemistry) in progress
- 6/2011: presented preclinical data at ADA meeting on two targets not yet in pipeline - click here for abstracts.
2) Phosphoenolypyruvate carboxykinase (cytosolic form, PCK1): Achieved 80% KD in rats, reduced fat deposits byt did not change glucose levels
Click here to download poster introduction to the metabolic franchise from 6/2011 Annual Meeting
PTP-1B Inhibitor
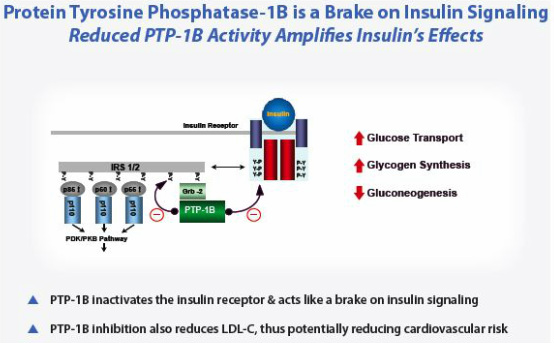
- Antisense targeting protein tyrosine phosphatase-1B (PTP-1B), a phosphatase that negatively regulates insulin receptor signaling. Reducing PTP-1B enhances insulin activity
- Long recognized as attractive target, but very difficult small molecule target because of close similarity to other proteins
- PTP1b inhibitors are insulin sensitizers. Antisense drugs more specific than current approved products
- Lowers blood glucose and LDL, may lower body weight.
- Former lead compound was ISIS-113715 which completed two phase 2 studies w/ no clinically significant adverse events
- 1) pts w/ uncontrolled blood sugar despite use of sulfonylureas. Data presented 6/2010 (76 pts, placebo and two doses- higher is same 200 mg subQ dose as mipo, similar reactions, but drs in this indication more used to this dosing). Stat sig reductions in short and intermediate term measures of glucose control and sig reduction in LDL, trend towards weight loss (associated with sig increase in adiponectin levels)
- 2) single agent in newly diagnosed type 2 diabetes. stat sig improvement in multiple measures of glucose control
- 4/2010: 26 wk tox should be well along by ye2010, should allow 26wk studies in 1q-2q2011, could give A1c data by ye2011- plan to complete these studies before partnering despite current interest.
- 9/2010: "gearing up for more studies"
- 11/2010: not mentioned
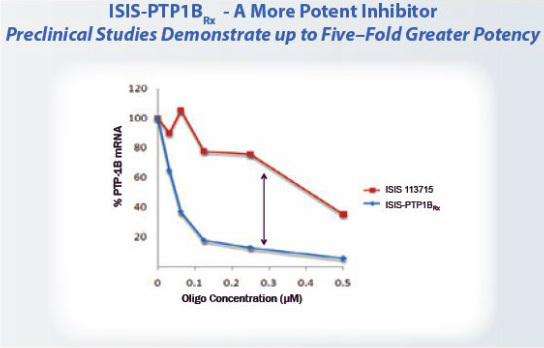
- 2010 10k: "We are currently completing long-term toxicology studies for our PTP-1B program, including evaluating a significantly more potent drug. Based on the significant potency difference we observed between the newer antisense drug and ISIS 113715, instead of developing ISIS 113715 we plan to begin clinical development on the more potent PTP-1B inhibitor in 2011."
- 7/2011: initiated phase 1 study with follow-on candidate - no details provided (PR)
- 6/2011 Annual meeting: backup has completed IND enabling studies, phase 1 in 2011 and phase 2 program in 2012 (combination studies with metfornin and insulin w/ 6 months treatment duration).
Glucagon Receptor (GCGR) Inhibitor
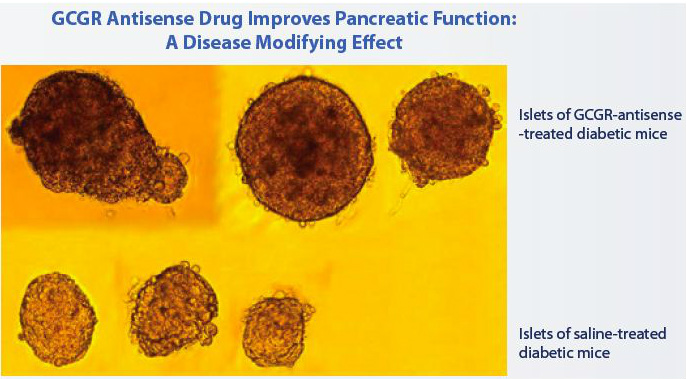
- "Glucagon is a hormone that opposes the action of insulin and stimulates the liver to produce glucose. In type 2 diabetes, unopposed action of glucagon can lead to increased blood glucose levels. Reducing the expression of GCGR using antisense inhibitors, and thereby reducing excessive liver glucose production, should lower blood glucose and help control type 2 diabetes." (2010 10k)...also increased levels of glucagon-like peptide (GLP-1)- a hormone that helps preserve pancreatic function
- No selective small molecules found to date for this target.
- Seen as complementary to PTP-1B program. Most important role in Type 2 Diabetes, so that is first targeted indication
- preclinical: "observed improved glucose control and reduced levels of blood triglycerides without producing hypoglycemia" (2010 10k)
- p1 data reported 6/2010: dose dependent, sig increase in glucagon-induced blood glucose levels in normal volunteers
- While p1 ongoing, found a more potent molecule and will now advance more potent follow-on candidate instead (2010 10k).
- program was returned by former partner Ortho-McNeil-Janssen (JNJ)...9/2007 deal for metabolic diseases including type 2 diabetes ($45m upfront total). Collaboration ended and rights returned to ISIS
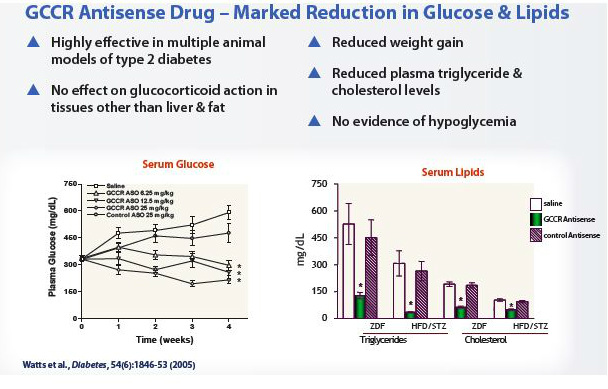
Glucocortocoid Receptor (GCCR) Inhibitor
- "Glucocorticoid hormones have a variety of effects throughout the body, including promoting liver glucose production and fat storage. Although scientists have long recognized inhibiting GCCR as an attractive strategy for developing therapeutics for type 2 diabetes, the side effects associated with systemic GCCR inhibition have challenged developers of traditional drugs. Antisense inhibitors of GCCR take advantage of the unique tissue distribution of oligonucleotides that allows the antisense drugs to antagonize glucocortocoid action primarily in liver and fat tissue. Notably, antisense drugs delivered systemically do not reduce GCCR expression in the central nervous system, or CNS, or adrenal glands, which could lead to systemic side effects." (2010 10k)...the side effects of decreased hormone level in the brain has prevented small molecule inhibitor development for this target
- preclinical: reduced blood glucose levels, dramatic and favorable effect on lipid levels (cholesterol and triglycerides), reduced body fat
- Plan to start phase 1 development in 2011 (2010 10k. Slipped from 4/2010 guidance of p1 late 2010 or early 2011)
- program was returned by former partner Ortho-McNeil-Janssen (JNJ)...9/2007 deal for metabolic diseases including type 2 diabetes ($45m upfront total). Collaboration ended and rights returned to ISIS
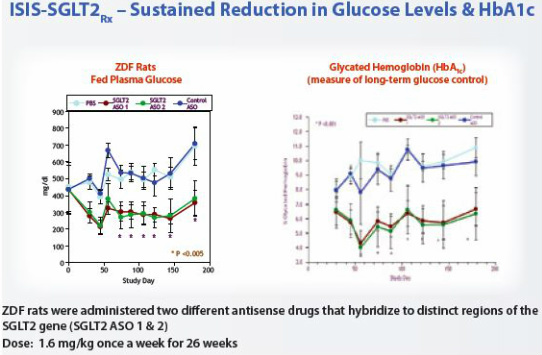
SGLT2 Inhibitor (ISIS-388626)
- "antisense drug that targets sodium glucose co-transporter type 2, or SGLT2, which is the major transporter for blood sugar re-absorption in the kidney. By specifically blocking the production of SGLT2 in the kidney tissue, we can promote blood sugar excretion and reduce blood sugar levels, without negatively affecting a related gene product, SGLT1." (2010 10k)
- Human subjects with mutated SGLT2 are otherwise healthy but have increased urine glucose levels
- Exception to company's general strategy- there are more advanced small molecule competitors- think can better reduce receptor levels and definitely not hit SGLT1 (note that Lexicon Pharma $LXRX is alone in the field in believing that it is an advantage that their molecule targets both SGLT1 and SGLT2 - Click here for an LXRX poster on LX4211 competitor)
- This is the first ISIS first candidate to hit a target in kidney. Oligo is generation 2.2 and is shorter than other candidates (12 vs 18-21 nt)
- preclinical: among most potent they have seen; reduced blood glucose and HbA1C (hemoglobin) levels, without causing hypoglycemia (low blood sugar)
- 4/2010: in 13 wk multi dose portion of p1 (started p1 2/09)
- 2010 10k: currently in p1 trial in healthy volunteers to measure glucose excretion in urine. Plan to complete this trial and initiate p2 trial in 2011 (timeline has slipped from 9/2010 forecast of data late 2010).
FGFR4 Inhibitor
- antisense targeting fibroblast growth factor receptor 4 (FGFR4) in liver and fat but not in CNS or heart
- treat obesity by increasing metabolism to cause fat and lipid burning
- preclinical studies "lowered body weight when administered as a single agent and in the presence or absence of a calorie-restricted diet. Additionally, inhibition of FGFR4 produced a decrease in body weight when administered in combination with an appetite-suppressing drug. In addition to the reduction in body weight, inhibiting FGFR 4 demonstrated an improvement in insulin sensitivity. ISIS-FGFR4Rx was safe and well tolerated in multiple species." (2010 10k)
- Added to pipeline 1/2011, plan to begin IND-enabling studies 1h2011.
- 8/2009 licensed IP from Verva (click to visit homepage)- undisclosed upfront, up to $6.1m milestones (can reduce this if convert to nonexclusive license), royalties
- 6/2011: data at ADA (click here for abstract): achieved 70-80% KD in mice and primates, reduced body weight and body fat