Navigate the Isis Pharma ISIS Research Pages
- Introduction (valuation, financials, outlook, company catalysts)
- Antisense background (science, technology, intellectual property, manufacturing)
- KYNAMRO - mipomersen (lead program in regulatory review for familial hypercholesterolemia)
- Mipomersen Liver Safety (special feature pages with extensive notes on long-term safety and efficacy of the drug)
- Cardiovascuar Disease pipeline (addtional CV drugs besides mipomersen)
- Metabolic Disease pipeline (several drugs for type II diabetes, as well as obesity)
- Cancer pipeline (including partnered with Eli Lilly $LLY and OncoGenex $OGXI)
- Severe, Rare, and Neurodegenerative Disease pipeline (incude GSK collaboration)
- Licensing deals and Satellite/spin-off companies (key business development activities)
- Discontinued Programs (not everything works out when you have a large and broad biotech portfolio)
PCSK9 Inhibitor
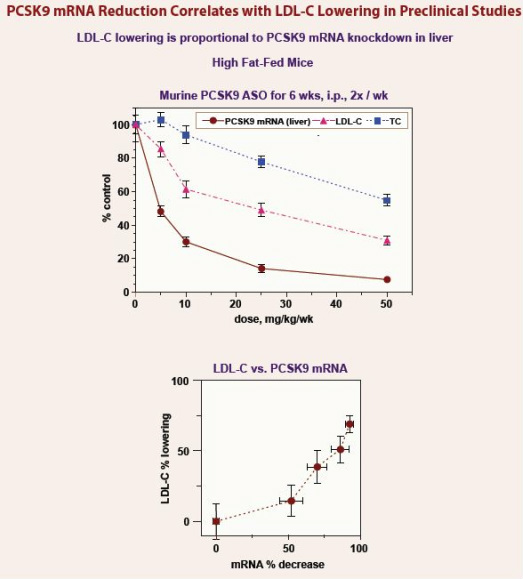
- Antisense targeting proprotein convertase subtilisin/kexin type 9 (PCSK9) - involved in the metabolism and cholesterol and LDL. PCSK9 breaks down the receptor that binds to and captures LDL particles...so inhibition results in more available receptors to eliminate LDL-C from the blood. Human levels of PCSK9 correlate with LDL levels.
- Click here for a nice Reuters piece on PCSK9 and its discovery as a cardiovascular target.
- Difficult drug target due to many closely-related family members
- Seen as a potential companion product w/ mipomersen. Potential commercial opportunity > 1 million pts
- The PCSK9 program was featured on this poster at the 6/2011 Annual Meeting.
- Partnered w/ BMS 5/2007 ($15m upfront and $9m research funding over 3 yrs, up to $170m milestones, undisclosed royalties) and collaboration extended 7/2010 for 2 years (expires 6/30/2012)
- Clinical candidate selected in 4/2008 (triggered $2m milestone), entered p1 3/2010 (triggered $6m milestone), but was terminated by BMS in 9/2010 (no safety issues) and will seek a more potent follow-on
- no fatty liver seen to date in animals.
- No add'l milestone when new candidate starts p1
- Competitor: ALNY has a RNAi program targeting PCSK9 (click here for more info)
AVI Pharma (formerly Ercole Biotech, acquired 5/2008)
- $AVII - click here to visit website
- 5/2003 cross-licensing deal involving alternative splicing of RNA and license of some ISIS chemistry patents. Each party could owe up to $21m milestones per drug plus royalties. 3/2008 $AVII acquired Ercole
- As of 12/31/2010 Isis did not have any drugs in development incorporating Ercole technology
- "Subject to the satisfaction of certain milestones triggering the obligation to make any such payments, we may be obligated to make milestone payments to Isis of up to $23.4 million in the aggregate for each product developed under a licensed patent under this agreement. As of December 31, 2010, we have not made, and are not under any current obligation to make, any such milestone payments, as the conditions triggering any such milestone payment obligations have not been satisfied. The range of percentage royalty payments required to be made by us under the terms of this agreement is from a fraction of a percent to mid single digits. We believe that our DMD, Ebola, Marburg and influenza programs [so what about Dengue and Junin virus programs-would they be covered?] will not fall under the scope of this agreement and therefore will not be subject to milestone or royalty obligations under its provisions" (AVII 2010 10k)
- ISIS has warrants to purchase 238k shares at $0.17 and 208k shares at $3.61 (stock quote $1.60 at 5/24/11)
Vitravene (fomivirsen)
- first generation antisense approved in 1998 by FDA for treatment of cytomegalogvirus (CMV) retinitis in AIDS patients.
- Launched in 11/1998 by Novartis $NVS. Due to increased survival achieved by newer HIV/AIDS medications, the incidence of such opportunistic infections has declined and NVS no longer markets the product
Altair Therapeutics (AIR645)
- private-spun out from ISIS 10/2007, ISIS owned ?? (<10% at wind-down).
- "We discovered AIR645 while evaluating targets involved in various inflammatory pathways. In our preclinical studies, AIR645 potently reduced target RNA and protein levels and demonstrated activity in mouse models of asthma. Altair evaluated AIR645 in patients with asthma in a Phase 2 study. In this study, treatment with AIR645 reduced its intended target and patients tolerated the drug well. However, reducing the target did not produce enough therapeutic benefit to warrant continued development, and Altair discontinued the program. While we were disappointed with these results, we gained significant insight from Altair's efforts with our first inhaled antisense drug." (2010 10k)
- AIR645 - 2nd gen antisense for inhaled therapies. Earlier pgm targets not disclosed.
- 12/2010 company shut down, ISIS reacquired assets associated with AIR645 and $0.4m cash